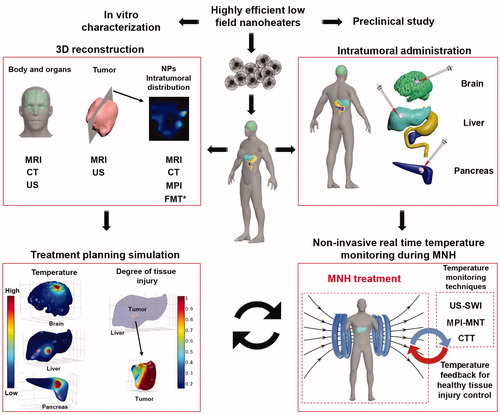
Figures & data
Table 1. Potential thermometry techniques compatible with in vivo MNH, limitations and advantages.
Table 2. Preclinical studies that show the effect of MNH (alone) in the treatment of solid tumors.
Table 3. Some mono-core and multicore nano-heaters (from 2008 to 2019), targeted for better SLP performance with in vivo MNH.
Table 4. Computer simulations for in vivo MNH.
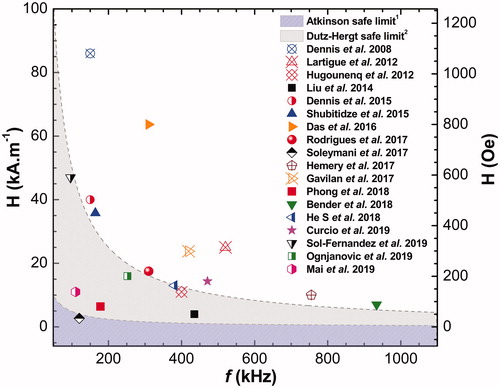
(in kHz) and amplitude
(in kA.m−1 and Oe) of the AMF for the reports presented in .
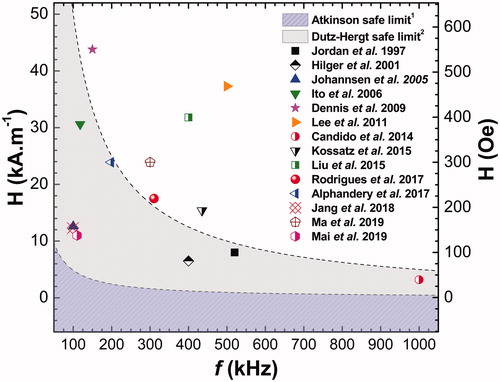
(in kHz) and amplitude
(in kA.m−1 and Oe) of the AMF for each work presented in .