Figures & data
Table 1. Advantages of using topical NSAIDs in preference to oral NSAIDs in osteoarthritis.
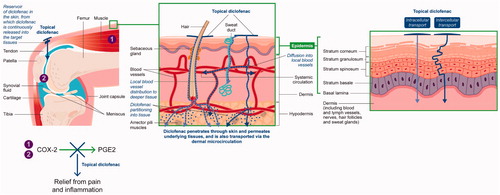
Table 2. Overview of the factors affecting the ability of a topical drug to optimally penetrate skin and permeate through the underlying tissues.
Table 3. Ratios of average plasma to tissue concentrations for topical versus oral diclofenac after administration over 3 days (data adapted from Brunner et al.Citation83,Citation90).
Table 4. Minimal effective therapeutic concentrations of diclofenac in target tissues, as reported in the literature.