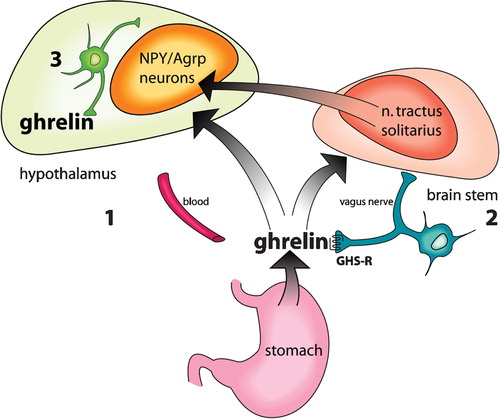
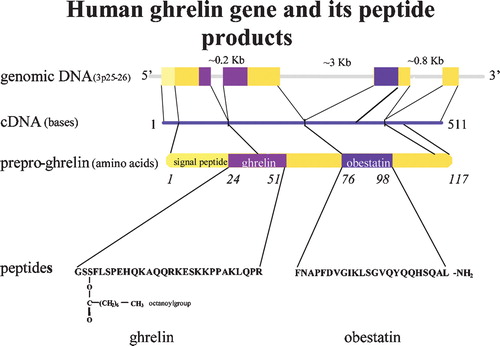
Figures & data
Table I. A summary of the effects of ghrelin (original reports or reviews with further details are indicated in brackets).
Table II. Transgenic and knockout models for ghrelin and GHS‐R.
Table III. Summary of the findings of studies published between January 2005 and July 2006 with reference to the subject of bariatric surgery and its effects on ghrelin levels. (For previous studies, refer to Citation205).