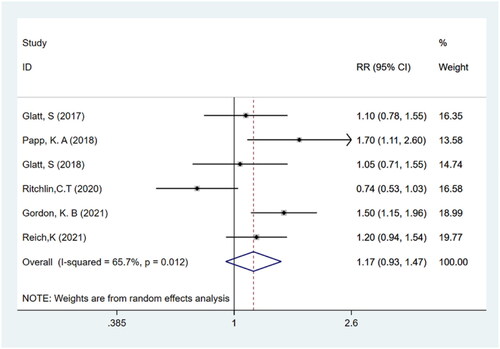
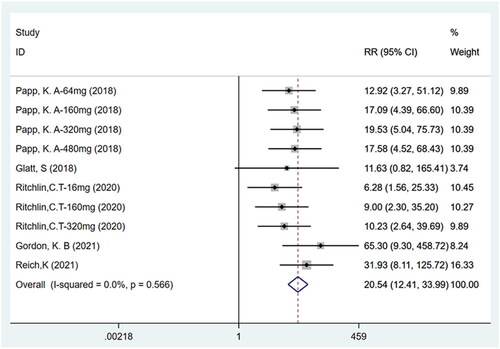
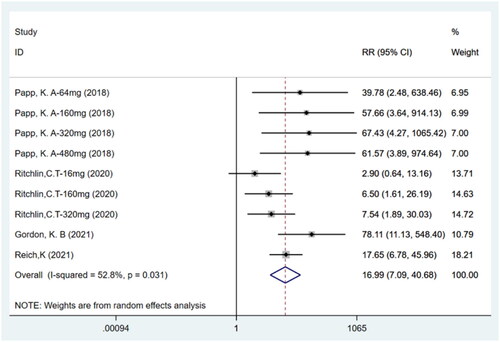
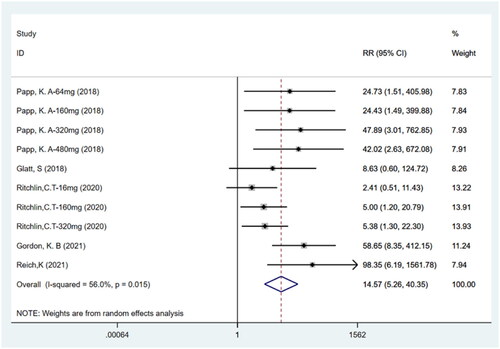
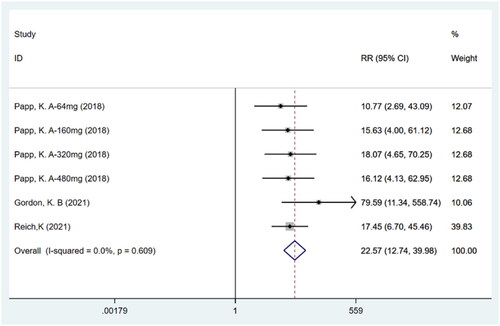
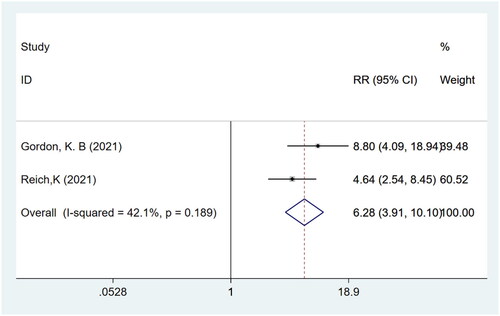
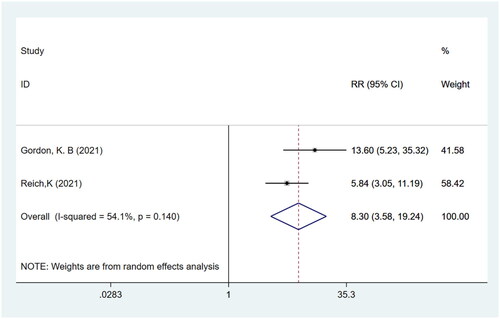
Figures & data
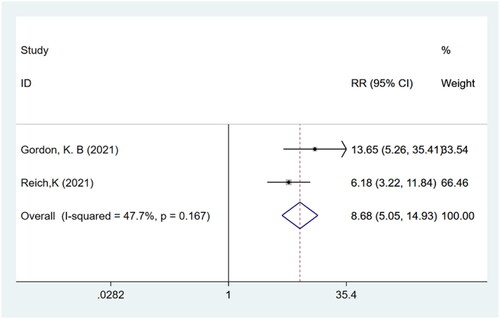
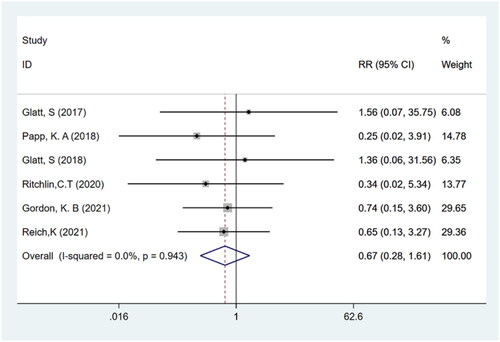
Table 1. Summary of clinical studies included in the meta-analysis.
Data availability statement
Data sharing is not applicable to this article as no datasets were generated or analyzed during the current study.