Figures & data
Figure 1. Proposed AOP for cytotoxicity-mediated SI cancer in mice. (A) AOP diagram. See text for discussion regarding (M)IE. Future evolution of this AOP may better define the (M)IE associated with villous cytotoxicity. (B) Structure of intestinal mucosa. Villi are finger- or leaf-like projections into the lumen that are predominantly covered with mature, absorptive enterocytes, along with occasional mucus-secreting goblet cells. These cells live only for a few days, then die and slough into the lumen. The crypts, or glands of Lieberkühn, are tubular invaginations of the epithelium, lined largely with younger epithelial cells, which serve as a source of enterocytes to multiple villi. At the base of the crypts are stem cells, which divide continually and function as the source of all the epithelial cells in the crypts and on the villi. Adapted from O'Brien et al. (Citation2013).
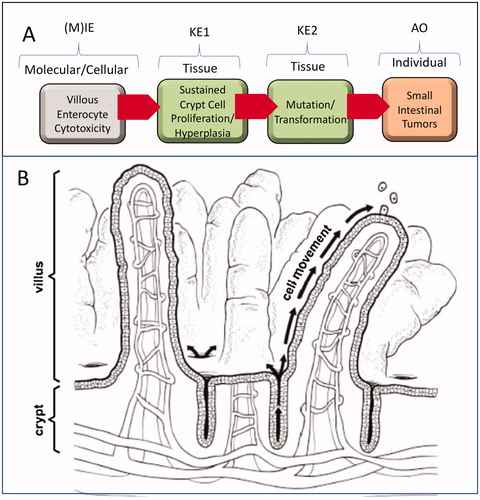
Figure 2. Synchrotron-based XRF microscopy of Cr in Swiss roll preparations of a duodenum from a mouse exposed to 180 ppm Cr(VI) for 7 days. (A) XRF for calcium (Ca) indicates presence throughout the intestinal mucosa. (B) XRF for chromium (Cr) indicates presence in villi but not crypts (white dotted circles). Adapted from Thompson, Wolf, et al. (Citation2015).
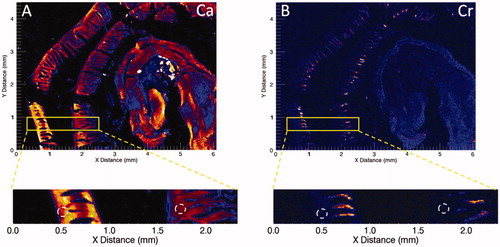
Table 1. Incidence of Cr(VI)-induced crypt hyperplasia in mice.
Table 2. Incidence of folpet- and captan-induced crypt hyperplasia in mice*.
Figure 3. Qualitative and quantitative evidence for sustained crypt hyperplasia following exposure to Cr(VI). Full transverse sections indicate the extent of villous blunting in mice exposed to 180 ppm Cr(VI) for 90 days (B) relative to controls (A). Higher magnification demonstrates the extent of crypt elongation in exposed mice (D) relative to controls (C). The arrows demonstrate blunting of villi, and the brackets indicate crypt expansion. (E) Cells per crypt and the incidence of crypt hyperplasia in mice exposed to various concentrations of Cr(VI) for 7 days. (F) Cells per crypt and the incidence of crypt hyperplasia in mice exposed to various concentrations of Cr(VI) for 90 days. Adapted from Thompson, Seiter, et al. (Citation2015); Thompson, Wolf, et al. (Citation2015); Thompson, Suh, et al. (Citation2017).
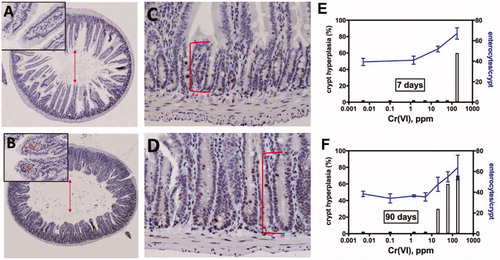
Figure 4. Qualitative evidence for sustained crypt hyperplasia following exposure to captan for 28 days. (A) Representative transverse section of control mouse SI. (B) Representative transverse section of SI in a mouse exposed to 6000 ppm folpet in diet. Folpet-induced short term cytotoxic and proliferative changes in the mouse duodenum, Gordon et al., Toxicology Mechanisms and Methods, reprinted by permission of Informa UK Limited, trading as Taylor & Francis Group, www.tandfonline.com
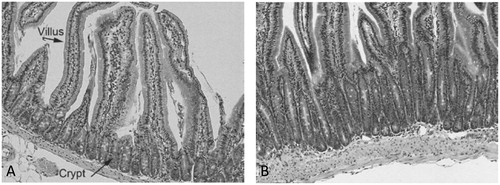
Table 3. Summary of in vivo duodenal genotoxicity assays for Cr(VI), captan, and folpet.
Table 4. Support for essentiality of key events (KEs).
Table 5. Support for biological plausibility of key event relationships (KERs).
Table 6. Empirical support for key event relationships (KERs).
Table 7. Evolved Bradford Hill causal considerations, defining questions, and body of evidence per OECD (Citation2018).
