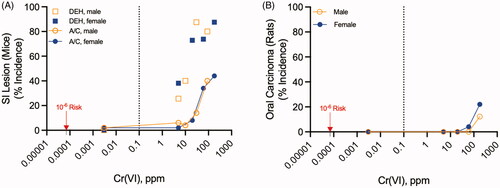
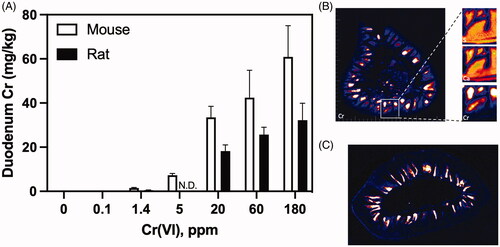
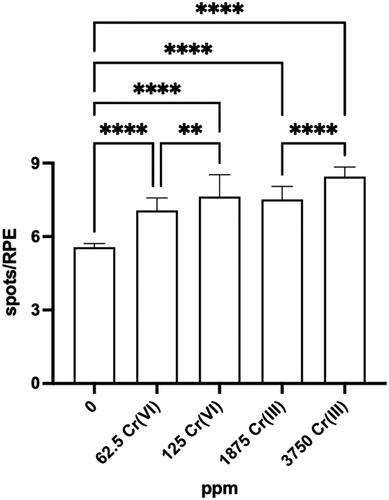
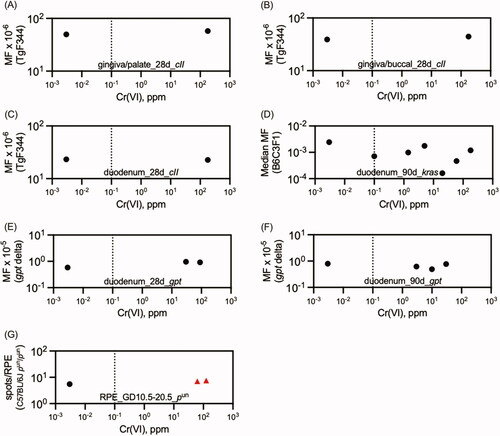
Figures & data
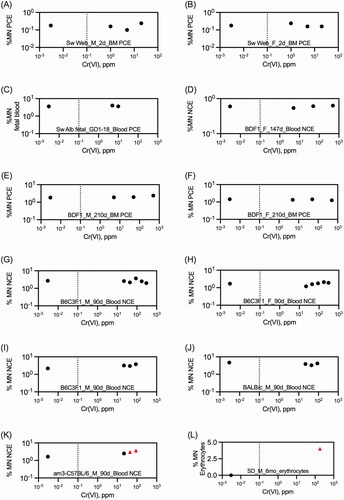
Table 1. Summary of in vivo mutation assays on Cr(VI).
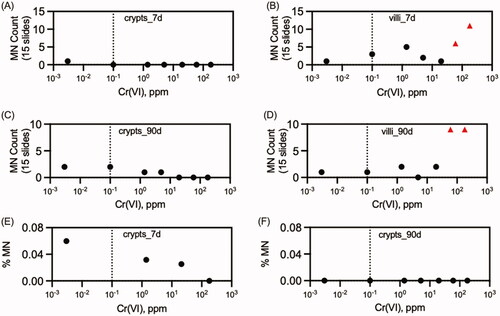
Table 2. Summary of in vivo chromosomal damage assays on Cr(VI).
Table 3. Summary of in vivo DNA damage assays on Cr(VI).