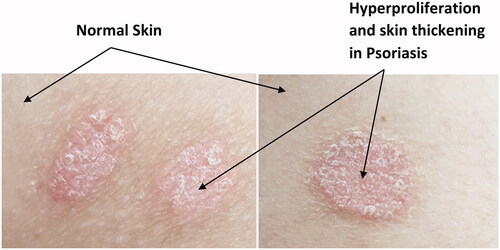
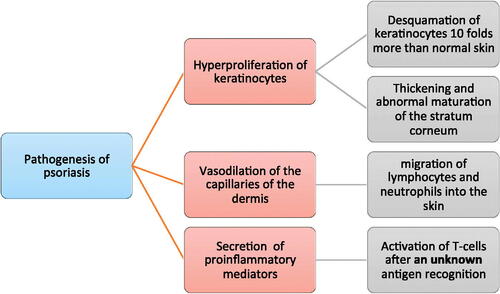
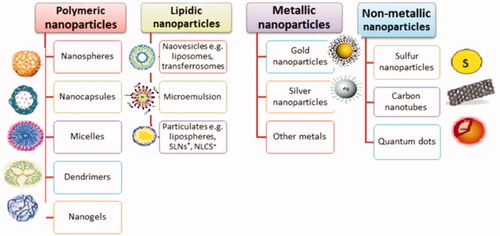
Figures & data
Table 1. A summary of polymers that are commonly used for nano drug delivery targets.
Table 2. A summary of recent approaches carried out to formulate drug delivery systems for topical management of psoriasis using polymeric nanoparticles.
Table 3. Recent approaches utilizing metallic nanoparticles in management of chronic psoriasis.
Table 4. A summary of lipidic nanosystems for topical treatment of psoriasis in the past few years.
Table 5. A summary of vesicular lipid drug delivery systems that have been invented by formulators in the past years.