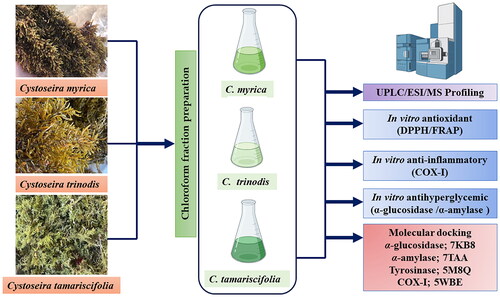
Figures & data
Table 1. Metabolite profiling of C. myrica, C. trinodis and C. tamariscifolia Chloroform fraction using UPLC-ESI-MS in the negative ion mode.
Table 2. Total phenolic and flavonoid content of C. myrica, C. trinodis and C. tamariscifolia chloroform fraction.
Table 3. Antioxidant activity (IC50) of C. myrica, C. trinodis and C. tamariscifolia chloroform fraction using DPPH and FRAP assays.
Table 4. Anti-hyperglycaemic activity and Anti-inflammatory activity % (IC50) of C. myrica, C. trinodis and C. tamariscifolia chloroform fraction.
Table 5. The docking scores achieved by the major identified compounds towards various enzymes.