Figures & data
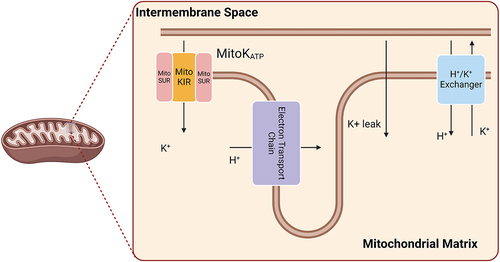
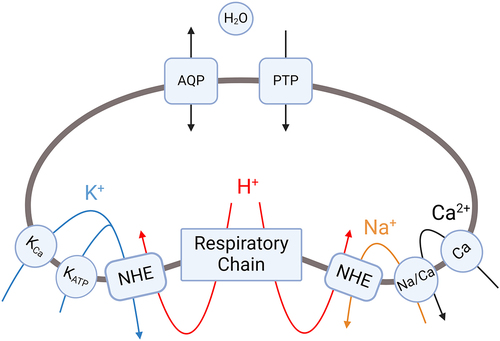
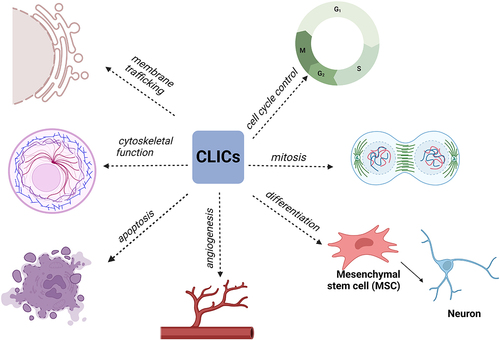
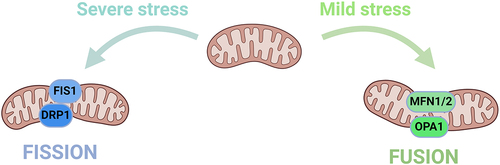
Table 1. Mitochondrial ion channels described in this article.
Paucek P, Mironova G, Mahdi F, et al. Reconstitution and partial purification of the glibenclamide-sensitive, ATP-dependent K+ channel from rat liver and beef heart mitochondria. J Biol Chem. 1992;267(36):26062–26069. doi: 10.1016/S0021-9258(18)35717-X Wojtovich AP, Brookes PS. The endogenous mitochondrial complex II inhibitor malonate regulates mitochondrial ATP-sensitive potassium channels: implications for ischemic preconditioning. Biochim Biophys Acta Bioenerg. 2008;1777(7–8):882–889. doi: 10.1016/j.bbabio.2008.03.025 Pereira O Jr., Kowaltowski AJ. Mitochondrial K(+) transport: modulation and functional consequences. Molecules. 2021;26(10):2935. doi: 10.3390/molecules26102935 Garlid KD. On the mechanism of regulation of the mitochondrial K+/H+ exchanger. J Biol Chem. 1980;255(23):11273–11279. doi: 10.1016/S0021-9258(19)70286-5 Bernardi P, Azzone GF. Electroneutral H±K+ exchange in liver mitochondria. Regulation by membrane potential. Biochim Biophys Acta. 1983;724(2):212–223. doi: 10.1016/0005-2728(83)90140-8 Fan M, Zhang J, Tsai CW, et al. Structure and mechanism of the mitochondrial Ca(2+) uniporter holocomplex. Nature. 2020;582(7810):129–133. doi: 10.1038/s41586-020-2309-6 Di Lisa F, Menabò R, Canton M, et al. Opening of the mitochondrial permeability transition pore causes depletion of mitochondrial and cytosolic NAD+ and is a causative event in the death of myocytes in postischemic reperfusion of the heart. J Biol Chem. 2001;276(4):2571–2575. doi: 10.1074/jbc.M006825200 Xu W, Liu Y, Wang S, et al. Cytoprotective role of Ca 2+ - activated K + channels in the cardiac inner mitochondrial membrane. Science. 2002;298(5595):1029–1033. doi: 10.1126/science.1074360 Ponnalagu D, Singh H. Anion Channels of Mitochondria. Handb Exp Pharmacol. 2017;240:71–101. Ashley RH. Challenging accepted ion channel biology: p64 and the CLIC family of putative intracellular anion channel proteins (review). Mol Membr Biol. 2003;20(1):1–11. doi: 10.1080/09687680210042746