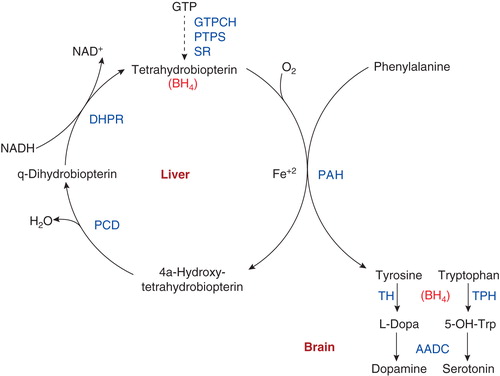
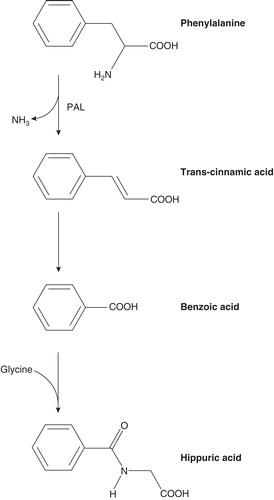
Figures & data
Table 1. Summary of clinical trials and selected other studies assessing treatment with sapropterin dihydrochloride in patients with elevated blood phenylalanine concentrations.
Table 2. Summary of clinical trials for rAvPAL–PEG (from Clinicaltrials.gov).