Figures & data
Table 1. Summary of the 5-grass pollen tablet clinical trials included in the pooled safety analysis.
Table 2. Demographics and baseline characteristics of the safety population (n = 2512).
Table 3. Extent of exposure to study treatment in the safety population (n = 2512), overall and in children and adolescents.
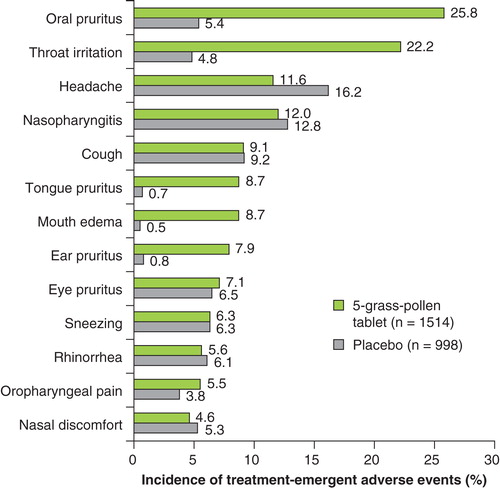
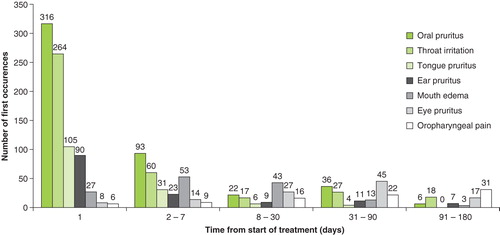
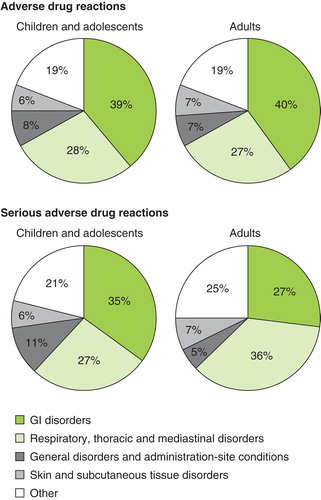
Table 4. TEAEs occurring in the safety population (n = 2512), overall and by age and treatment groups.
Table 5. TEAEs occurring in the safety population (n = 2512), stratified by sensitization and asthma status.
Table 6. PASS conducted in Germany: main characteristics.