Figures & data
Table 1. Overview of inactivation methods for the development of killed virus vaccines.
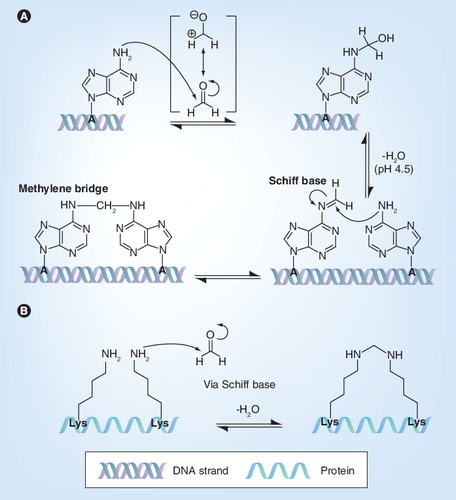
Table 2. Effect of formaldehyde on different viruses: inactivation, viral protein modification, induction of virus-specific and virus-neutralization antibodies and protection on challenge (reduction of viremia and fewer clinical signs).
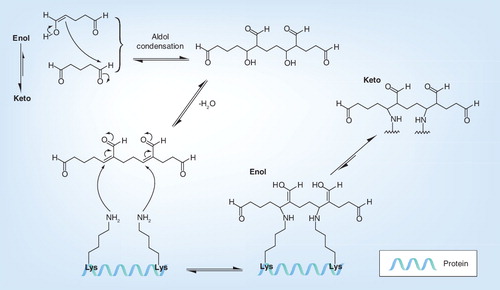
Table 3. Effect of glutaraldehyde on different viruses: inactivation, viral protein modification, induction of virus-specific and virus-neutralization antibodies and protection on challenge (reduction of viremia and fewer clinical signs).
Table 4. Effect of 2,2’-dithiodipyridine on different viruses: inactivation, viral protein modification, induction of virus-specific and virus-neutralization antibodies and protection on challenge (reduction of viremia and fewer clinical signs).
Table 5. Effect of β-propiolactone on different viruses: inactivation, viral protein modification, induction of virus-specific and virus-neutralization antibodies and protection on challenge (reduction of viremia and fewer clinical signs).
Table 6. Effect of binary ethylene imine on different viruses: inactivation, viral protein modification, induction of virus-specific and virus-neutralization antibodies and protection on challenge (reduction of viremia and fewer clinical signs).
Table 7. Effect of denaturing agents on different viruses: inactivation, viral protein modification, induction of virus-specific and virus-neutralization antibodies and protection on challenge (reduction of viremia and fewer clinical signs).
Table 8. Effect of irradiation on different viruses: inactivation, viral protein modification, induction of virus-specific and virus-neutralization antibodies and protection on challenge (reduction of viremia and fewer clinical signs).