Figures & data
Figure 1 PICO chart detailing inclusion in systematic review.
Abbreviations: AATD, alpha-1 antitrypsin deficiency; AAT, alpha-1 antitrypsin; Pi, protease inhibitor; COPD, chronic obstructive pulmonary disease; PICO, population, interventions, comparators, and outcomes.
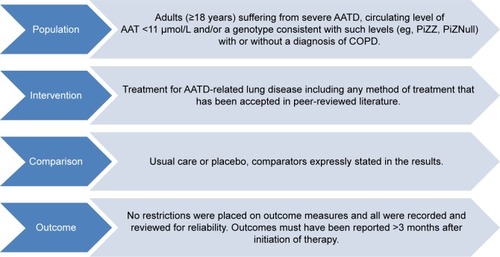
Figure 2 PRISMA flow diagram.
Abbreviation: COPD, chronic obstructive pulmonary disease.
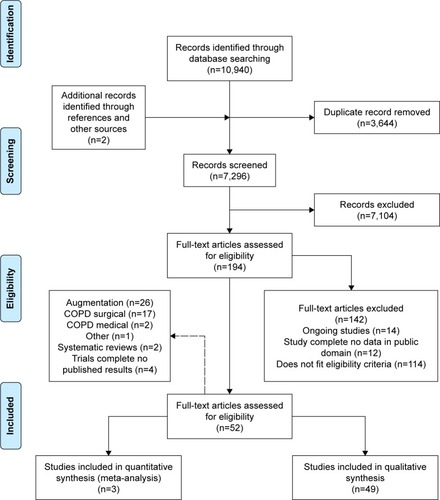
Figure 3 Forest plots of the objective results from meta-analysis of augmentation trials.
Notes: (A) Mean annual change in lung density. (B) Mean FEV1 % predicted. (C) Standardized mean difference in DLCO. Differences in units used for DLCO (mmol/min/kPa and mL/mmHg/min) and the use of percentage change from baseline in RAPID, but annual change in the other studies required the use of a standardized mean difference plot.
Abbreviations: FEV1, forced expiratory volume in one second (L); DLCO, diffusing capacity of the lungs for carbon monoxide; mmol/min/kPa, millimole per minute per kilopascal; mL/mmHg/min, milliliter per millimeter of mercury per minute; SD, standard deviation; CI, confidence interval; df, degrees of freedom; Std, standard; IV, inverse variance.
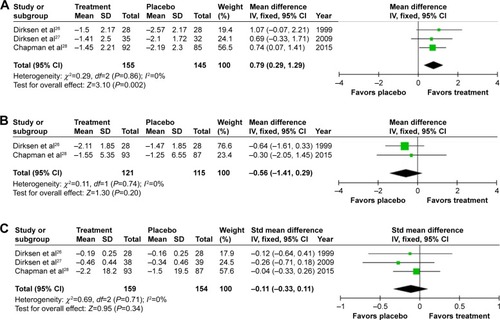
Figure 4 Forest plots for patient-reported outcomes.
Abbreviations: SGRQ, St Georges Respiratory Questionnaire; SD, standard deviation; CI, confidence interval; df, degrees of freedom; IV, inverse variance.
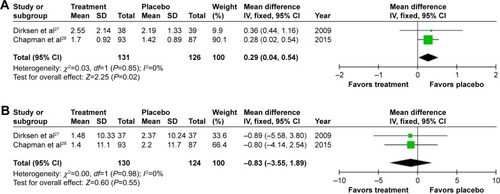
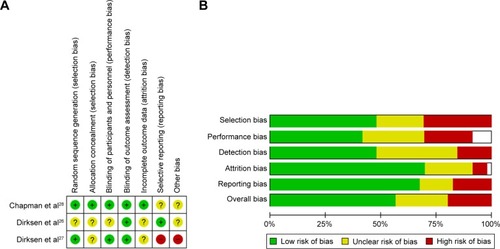
Figure 5 Risk of bias in included studies.
Abbreviation: RCT, randomized controlled trial.