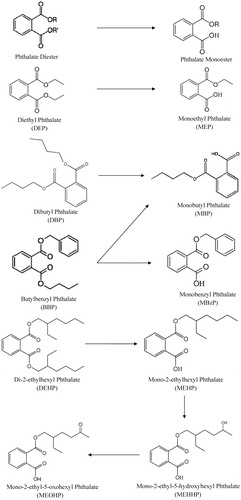
Figures & data
Table 1. Summary of the epidemiological literature exploring the link between phthalate exposure and adverse reproductive health outcomes in women.
Table 2. Summary of the NOAELs and LOAELs in mg/kg/d as well as the direction of change for each outcome, respectively, found in animal studies for each individual phthalate.