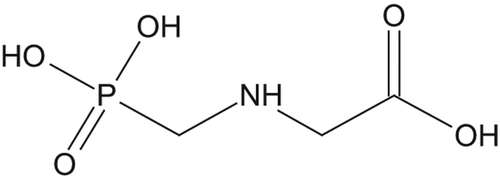
Figures & data
Table 1. Summary of toxicological endpoints for glyphosate (CitationGermany Rapporteur Member State 2015c).
Table 2. Summary of one-year toxicity studies with glyphosate.
Table 3. Study 1–26-month feeding study of glyphosate in rats (CitationMonsanto 1981).
Table 4. Study 1 – Pituitary tumor findings.
Table 5. Study 1 - Interstitial cell tumor findings in the testes.
Table 6. Study 1 – Summary of the contemporary historical control data for interstitial cell tumors in the testes of rats in chronic toxicity studies.
Table 7. Study 2 – Two-year feeding study of glyphosate in rats (CitationMonsanto 1990).
Table 8. Study 3 – Two-year feeding study of glyphosate in rats (CitationCheminova 1993a).
Table 9. Study 4 – Two-year feeding study of glyphosate in rats (CitationFeinchemie Schwebda 1996).
Table 10. Study 5 – Two-year feeding study of glyphosate in rats (CitationExcel 1997).
Table 11. Study 6 – Two-year feeding study of glyphosate in rats (CitationArysta Life Sciences 1997b).
Table 12. Study 7 – Two-year feeding study of glyphosate in rats (CitationSyngenta 2001).
Table 13. Study 8 – Two-year feeding study of glyphosate in rats (CitationNufarm 2009b).
Table 14. Publication, Study 9 – Two-year drinking water study in rats with 13.85% glyphosate ammonium salt (CitationChruscielska et al. 2000a).
Table 15. Study 10 – Two-year feeding study with glyphosate in mice (CitationMonsanto 1983).
Table 16. Study 11 – Two-year feeding study with glyphosate in mice (CitationCheminova 1993b).
Table 17. Study 12 – Two-year feeding study with glyphosate in mice (CitationArysta Life Sciences 1997a).
Table 18. Study 13–18-Month feeding study with glyphosate in mice (CitationFeinchemie Schwebda 2001).
Table 19. Study 14–18-Month feeding study with glyphosate in mice (CitationNufarm 2009a).
Table 20. Summary of select neoplasms in male rats (Studies 1–8).
Table 21. Summary of select neoplasms in female rats (Studies 1–8).
Table 22. Summary of select neoplasms in male mice (Studies 10–14).
Table 23. Summary of select neoplasms in female mice (Studies 10–14).
Figure 2. Likelihood of glyphosate carcinogenicity based on experimental and epidemiological data; a causal inference grid as proposed by CitationAdami et al. (2011) to utilize both toxicological and epidemiological data.