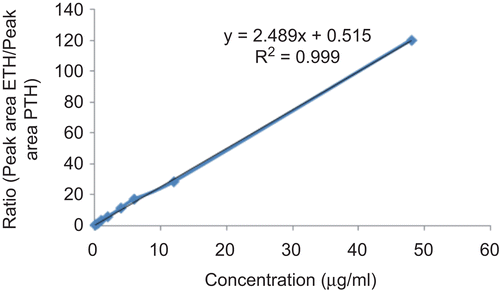
Figures & data
Table 1. Accuracy and precision data of ethionamide in spiked mice plasma.
Table 2. Recovery of ethionamide in spiked mice plasma.
Table 3. Stability of ethionamide in spiked mice plasma.
Table 4. Characterization of formulation before and after 90 days of storage at 4–8°C.
Table 5. Pharmacokinetic parameters of free ethionamide (ETH) and ethionamide-loaded nanoparticles (ETH NPs) upon oral administration in the mice.
Figure 2. In-vivo plasma concentration–time profile of orally-administered free ethionamide (ETH) and ethionamide-loaded nanoparticles (ETH NPs) equivalent to 130 mg/kg and 260 mg/kg of ethionamide in mice (n = 3–4 mice at each time point). Insert show an expanded view of 0–24 h plasma profile.
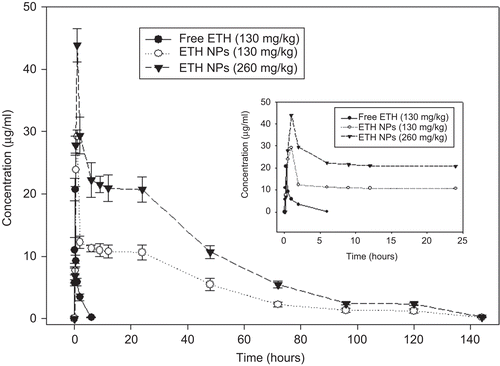
Table 6. Tissues drug levels following single oral administration of ethionamide-loaded PLGA nanoparticles (ETH NPs) at two doses in mice.
Table 7. Calculated values for AUC above MIC, Cmax above MIC, and time above MIC for free ethionamide (ETH) and ethionamide-loaded nanoparticles (ETH NPs) at a MIC90 (0.6 µg/ml).