Figures & data
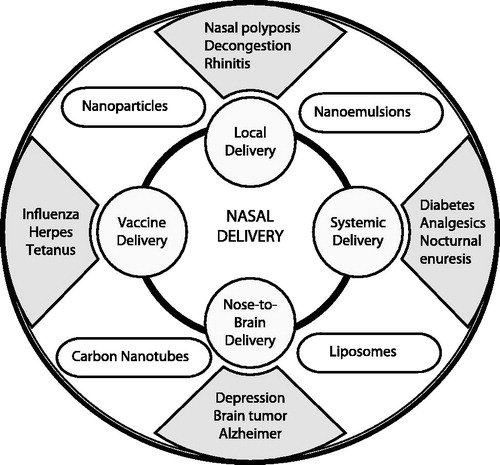
Table 1. Investigational target diseases/pathological conditions for nasal delivery.
Table 2. Marketed nasal products for topical/local delivery.
Table 3. Marketed nasal products for systemic delivery.
Table 4. Drugs and nanotechnology-based systems under investigation for nose-to-brain delivery.
Table 5. Marketed/Under clinical trial nasal products for vaccination.
Table 6. Nanotechnology-based systems under investigation for IN vaccination.
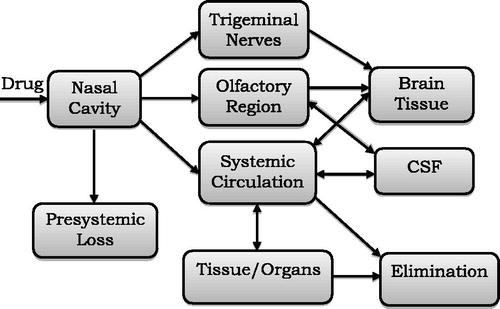
Table 7. Pathological conditions and their impact on nasal mucociliary clearance (Soane et al., Citation2001; Illum, Citation2006).