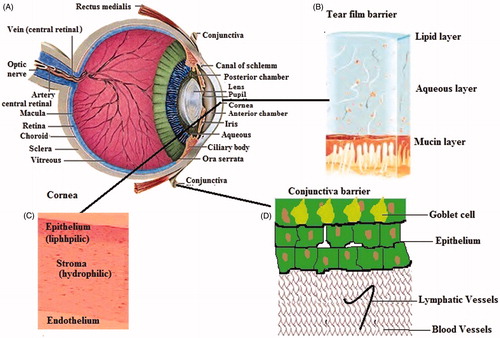
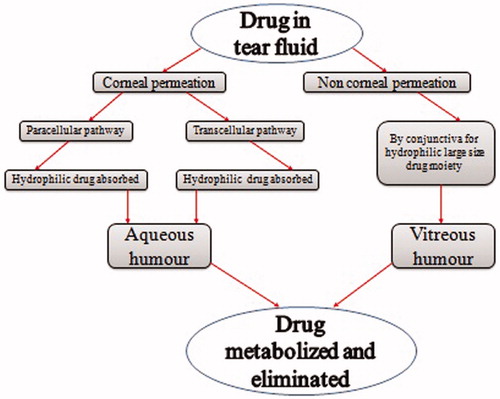
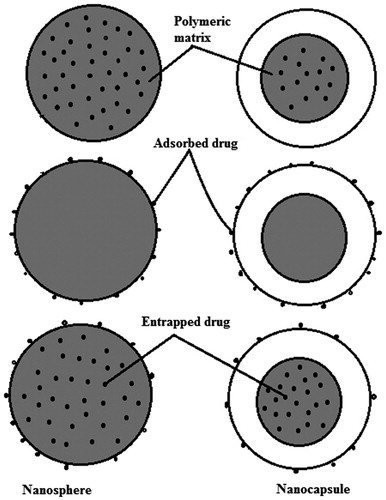
Figures & data
Table 1. Colloidal approaches of various drugs for ocular delivery.