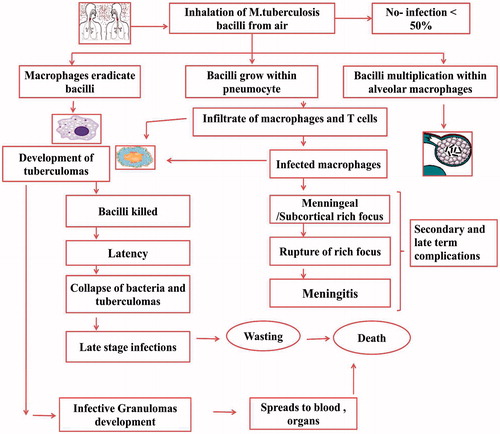
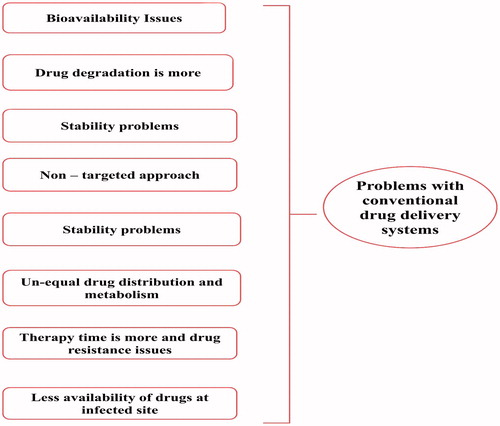
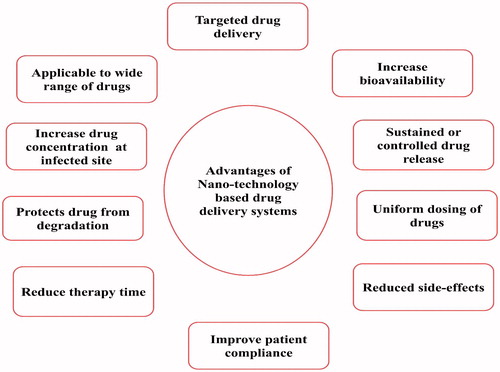
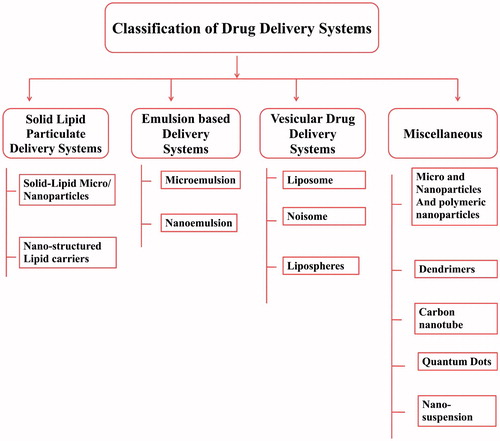
Figures & data
Table 1. Classification of anti-TB drugs and their mechanism of action.
Table 2. Examples of solid-lipid micro and nanoparticles in tuberculosis treatment.
Table 3. Examples of anti-tubercular drug loaded emulsion systems.
Table 4. Examples of liposomal-based preparations for tuberculosis.
Table 5. Examples of niosomes preparations in tuberculosis.
Table 6. Dendrimer-based anti-tuberculosis preparations.
Table 7. Examples of microparticles/nanoparticles/polymeric nanoparticle-based anti-TB preparations.
Table 8. First line drugs and their resistance.
Table 9. Newer drug candidates in tuberculosis treatment (Mohan et al., Citation2013).
Table 10. Second line anti-tubercular drugs used in the treatment of MDR-TB and XDR-TB (Prasad, Citation2007).
Table 11. Examples of immunostimulant (Patil et al., Citation2012).
Table 12. Examples of immunosupressants (Patil et al., Citation2012).
Table 13. Vaccines used in immunotherapy in tuberculosis.
Table 14. Antibodies in tuberculosis immunotherapy.
Table 15. Studies showing role of immunomodulators as adjuvant to chemotherapy in the treatment of tuberculosis.