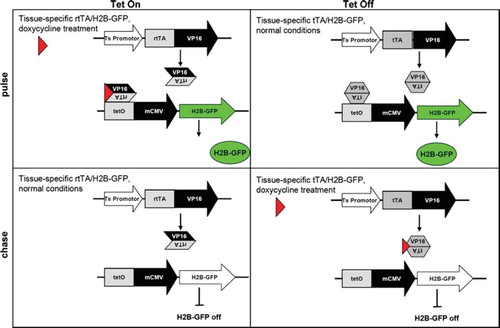
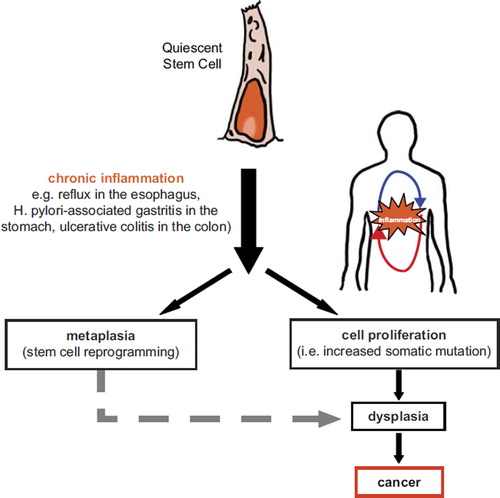
Figures & data
Table 1. Overview of quiescent stem cells characterized to date.
Table 2. Overview of quiescent cancer stem cells characterized to date.