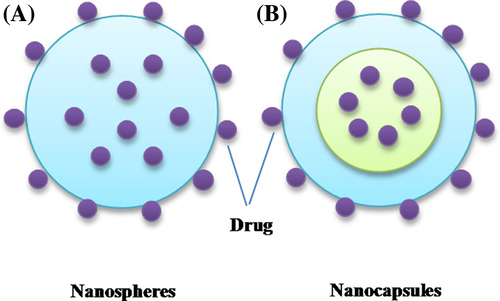
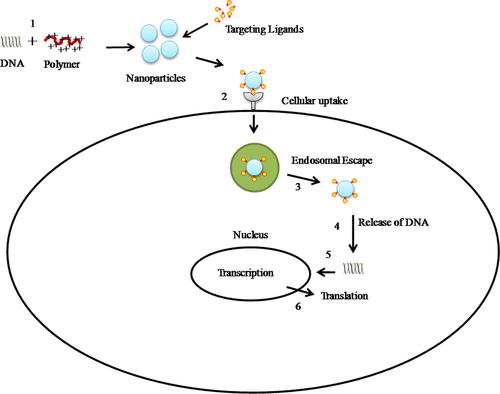
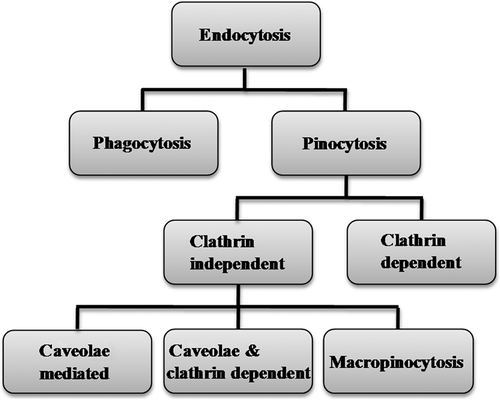
Figures & data
Table I. Effect of nanoparticles size on internalization.
Table II. Effect of nanoparticles size on transfection efficiency.
Table III. Effect of nanoparticles size on cytotoxicity.
Table IV. Effect of nanoparticles size on pharmacokinetics and biodistribution.