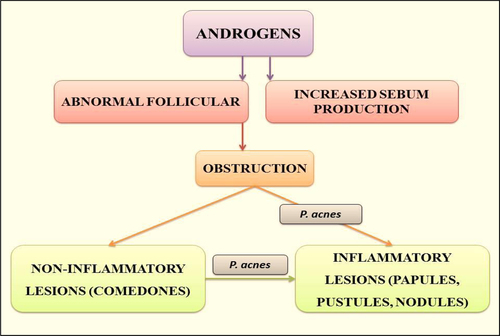
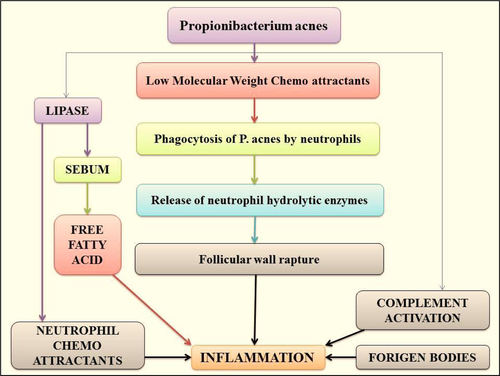
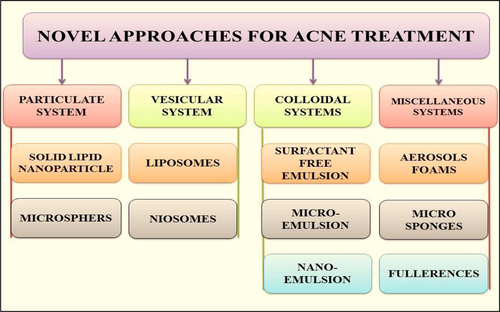
Figures & data
Table I. Characteristics of various grade of acne.
Table II. Topical drug for acne treatment.
Table III. Systemic drug for acne treatment.
Table IV. Applications of nano-carrier systems for treatment of acne.