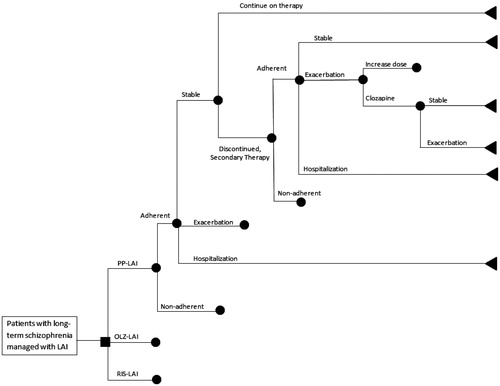
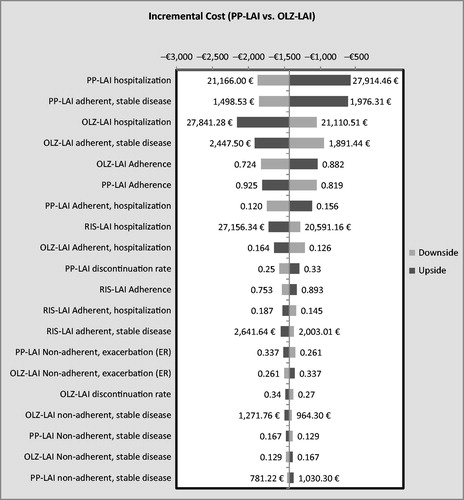
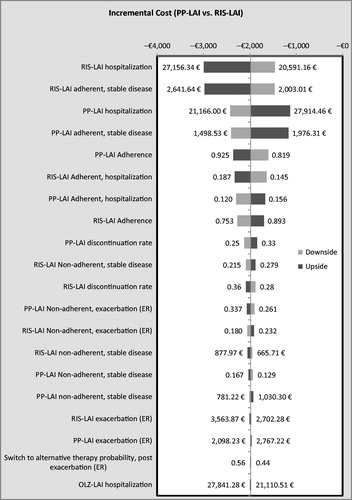
Figures & data
Table 1. Doses used to treat patients (source of data).
Table 2. Probability rates used in the model and sources of information.
Table 3. Costs (in 2011 euros) used in the model.
Table 4. Results of the economic analysis.