Abstract
Objective. Atherosclerotic cardiovascular diseases caused by traditional and non-traditional risk factors are the most common cause of morbidity and mortality in hemodialysis patients. Recently, much interest has been focused on non-traditional factors, such as oxidative stress, inflammation, and endothelial dysfunction. Hemodialysis patients are not only exposed to oxidative stress but also to inflammation. Although anticoagulants are the most frequently used drugs in hemodialysis patients, their effect upon oxidative stress and inflammation in dialysis patients are still unknown. Methods. Thirty-three hemodialysis patients were randomized into three groups. Group 1 received standard heparin while group 2 received low molecular weight heparin during the dialysis therapy. Group 3 (control group) did not receive any anticoagulant agent. Investigators were blinded to the therapy. Serum concentrations of oxidative stress and inflammation markers, including C-reactive protein, tumor necrosis factor alpha, superoxide dismutase, and malondialdehyde, were measured before and after dialysis session. Results. The oxidative stress and inflammation markers were significantly increased in groups 1 and 3 (p < 0.05 for each) compared to their baseline values. In contrast, baseline and end-treatment values of the oxidative stress and inflammation markers were comparable in the group 2 (p > 0.05). Conclusion. These findings indicate that the type of anticoagulants may take a role in the acute effect of hemodialysis upon oxidative stress and inflammation markers. A comparison of the groups revealed that low molecular weight heparin decreased the oxidative stress and inflammation, whereas standard heparin increased the oxidative stress and inflammation. Low molecular weight heparin appears to have an additive benefit for hemodialysis patients.
INTRODUCTION
Cardiovascular diseases are the major cause of morbidity and mortality among maintenance hemodialysis patients. Although the pathophysiology of cardiac dysfunction in patients undergoing hemodialysis (HD) is multifactorial, atherosclerosis plays a central role in this dysfunction.Citation[1],Citation[2] This is probably due to high prevalence of both traditional risk factors (such as hypertension, dyslipidemia) and non-traditional risk factors (such as oxidative stress, inflammation). In addition, it has been proposed that both dialysis-related and unrelated factors increase the concentrations of pro-inflammatory cytokines that are causing inflammatory state in advanced renal failure.Citation[3],Citation[4] HD patients are exposed to oxidative stress via several mechanisms.Citation[5],Citation[6] A relationship between oxidative stress and inflammation has also been proposed.Citation[7],Citation[8] In this respect, enhanced oxidative stress and inflammation in HD patients may be considered a risk factor for accelerated atherosclerosis.
Several treatment strategies improving oxidative stress and inflammation in HD patients have been reported. Anticoagulants, the most commonly used drugs among HD patients, have also been claimed to have some effects on oxidative stress and inflammation. It has been proposed that heparin exerts its anti-inflammatory properties through cell adhesion processes.Citation[9] Additionally, it was observed that standard heparin reduces oxidative stress in HD patients.Citation[10] Their anti-inflammatory and anti-oxidant effect on HD patients, however, is still controversial.
Briefly, oxidative stress and inflammation, which play an important role in cardiovascular morbidity and mortality in HD patients by causing endothelial dysfunction and atherosclerosis, are incidents that lie one inside the other. Consequently, evaluating the effect of low molecular weight heparin (LMWH) versus standard heparin (SH) on serum levels of inflammatory and oxidative stress markers in maintenance HD patients may reveal a superiority of one of these agents over another to improve oxidative stress and inflammation.
MATERIALS AND METHODS
Study Protocol
Thirty-three chronic hemodialysis patients were enrolled in this comparative study after having given informed consent. Patients with chronic hepatitis, cancer, hematological and inflammatory disorders, and severe hyperparathyroidism, as well as patients who have received erythropoietin, active D vitamins, statins, immunosuppressive therapy, angiotensin-converting enzyme inhibitors, and exchange transfusion for last three months were excluded. Intravenous iron therapy was stopped four weeks before the blood samples collected. Patients were randomly divided into three groups, each with 11 patients. SH and LMWH was administered to groups 1 and 2, respectively. Dialysis without anticoagulant therapy was applied to group 3 by the periodic rinsing of the blood circuit with saline. Patients received LMWH for anticoagulation prior to the study. Each patient in group 1, group 2, and group 3 underwent dialysis for only one session in the presence of SH, LMWH, and without heparin, respectively. Heparin (sodium heparin 5000 IU/mL) was administered as a bolus dose (50 IU/kg body weight) intravenously into the predialyser arterial line of the extracorporeal blood circuit, followed by a maintenance dose of 1000 IU heparin per hour. Infusion was discontinued 1 h prior to the cessation of HD. LMWH (dalteparin) was administered 3–4 min before dialysis as a bolus dose, 1mg (100 IU anti factor Xa activity) per kg body weight, injected into the arterial line predialyser. Anticoagulation was monitored by visual inspection of the arterial bubble trap every 30–60 min. The main characteristics of the HD patients are presented in . The HD patients received thrice-weekly treatment with polysulphone hollow-fiber dialyzer. The duration of dialysis was approximately four hours. Blood flow rates were 250–300 mL/min in all groups. Vascular accesses were via a native arterio-venous fistula in all patients. In addition, biocompatible membranes were used, though ultrapure dialysate was not used in the study groups.
Table 1 Characteristics of treatment and control groups
Blood samples (10 ml) were drawn from the arteriovenous fistula just before and within 5 min after the cessation of hemodialysis session. Following centrifugation, serum was stored at −80°C.
Laboratory Analysis
High sensitivity C-reactive protein (hs-CRP), tumor necrosis factor alpha (TNF-α), superoxide dismutase (SOD), and malondialdehyde (MDA) were evaluated. Hs-CRP was determined with nephelometric test (Dade Behring, Marburg GmbH, Germany) by using BN 100 machine (Dade Behring, Liederbach, Germany). Normal range of hs-CRP is 0–2 mg/L. TNF-α levels were determined with ELISA method. (Medgenix, Biosource International, Camarillo, California, USA). Results were shown as pg/mL. Serum ferritin was measured by the Immulite method. Plasma MDA and SOD were determined with the SatohCitation[11] and Sun et al.Citation[12] spectrophotometric methods, respectively.
Statistical Analysis
Data are presented as mean ± SEM. Parameters converted to their logarithms because data were not normally distributed. Parameters between the treatment (groups 1 and 2) and control (group 3) were compared using Mann Whitney U test. To compare values obtained before and after the dialysis session, paired T test was used. Results with a p less than 0.05 were considered statistically significant.
RESULTS
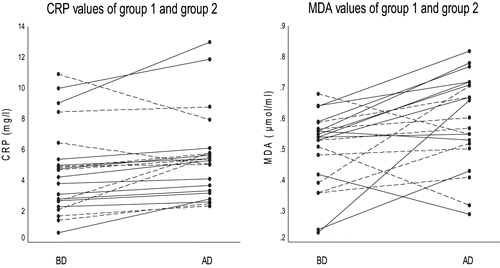
Baseline characteristics of the groups (age, gender, dry weight, blood pressure, etc.) were comparable. Mean hs-CRP value was significantly increased in group 1 from 4.5 ± 0.8 mg/L to 5.6 ± 1.1 mg/L after dialysis (p < 0.05). When LMWH was used as an anticoagulant therapy, no significant change was observed (from 4.8 ± 0.9 mg/L to 5.4 ± 0.6 mg/L, p > 0.05). However, serum concentrations of inflammatory and oxidative stress markers were significantly increased in group 3, which did not receive any anticoagulant therapy with dialysis (p < 0.05; see and ).
Table 2 Laboratory values of groups
DISCUSSION
HD patients were exposed to oxidative stress and inflammation via diverse mechanisms, including antioxidant deficiency, neutrophil activation during dialysis, and chronic inflammation. It is well known that both oxidative stress and inflammation are factors that contribute to the development of cardiovascular diseases due to atherosclerosis, and it has been pointed out that there is a link between oxidative stress and inflammation in HD patients.Citation[7],Citation[13]
It has been well established that LMWH has many superior advantages, such as lower hemorrhage risk, a positive effect on serum lipid profile, and easy usage, over SH. However, their higher cost limits their broad usage. Recently, Wang et al.Citation[9] observed that heparin exerts its anti-inflammatory properties by affecting cell adhesion processes. In addition, Sela et al.Citation[10] showed that heparin reduced oxidative stress among HD patients. Nonetheless, all of these data do not provide sufficient evidence to consider whether anticoagulants, which are the most commonly used drugs in dialysis, have any effect on serum concentrations of oxidative stress and inflammatory markers. If there is, it is also not clear whether different types of anticoagulants are superior to others with regard to these aforementioned effects.
The major finding of this study was that the administration of LMWH in chronic HD patients led to a significant reduction in oxidative stress and inflammation markers compared with the control group, whereas using SH in chronic HD patients showed a similar increase in oxidative stress and inflammation as in control group. This could be the first study comparing the acute effect of SH and LMWH on oxidative stress and inflammation in chronic HD patients.
Since Bergström et al.Citation[14] had found a strong relationship between elevated CRP and increased mortality in hemodialysis patients, CRP attracted attention as a predictive marker of cardiovascular mortality. CRP may also reflect the consequences of elevated levels of circulating pro-inflammatory cytokines, which are known as mediators of the acute phase response.Citation[15],Citation[16] Moreover, pro-inflammatory cytokines per se may contribute to the atherosclerosis.Citation[17–19] In these patients, CRP values were between 0.6 and 12 mg/L, indicating the presence of low grade inflammation.Citation[20] No significant increase was observed in serum levels of CRP and serum levels of TNF-α in the group 2. On the contrary, TNF-α and CRP were significantly increased in the group 1 and 3
Boaz et al. demonstrated that serum levels of MDA have also been associated with cardiovascular morbidity in the dialysis population.Citation[21] In the present study, while plasma MDA levels significantly increased in group 1 and group 3, SOD levels decreased significantly. However, in group 2, plasma MDA and SOD levels did not significantly change. The absence of significant changes in MDA and SOD in group 2 suggests that LMWHs may improve the imbalance between oxidant and antioxidant status and protect HD patients from the harmful effects of oxidative stress. However, SH did not show these effects. A recent study reported both SH and LMWH increased the oxidative stress;Citation[22] this result, however, is not consistent with the current study's findings. Nevertheless, in experimental research, it has been found that dalteparin has significantly greater anti-inflammatory effects than SH.Citation[23]
Recent investigations have demonstrated a link between oxidative stress and inflammation.Citation[24–26] In addition, emerging evidence suggests that an improvement in the oxidative stress status can have a direct beneficial effect upon inflammation in the dialysis population.Citation[7],Citation[8] The similar alteration of both serum levels of oxidative stress and inflammation markers in these HD patients may be explained with this information. Moreover, hypoalbuminemia and chronic volume overload are associated with oxidative stress and inflammation, as well;Citation[27],Citation[28] however, no findings about the inadequacy of hemodialysis were observed in any of the present study groups with respect to serum albumin levels, cardio-thoracic rate, blood pressure, and Kt/V.
Consequently, the results observed in one dialysis session suggest that LMWH improves the oxidative stress and inflammation in chronic HD patients. Long-term and cross-over studies are warranted to demonstrate the effects of SH and LMWH to illuminate the exact role on cardiovascular morbidity and mortality due to oxidative stress and inflammation.
REFERENCES
- Lindner A, Charra B, Sherrard DJ, Schribner BH. Accelerated atherosclerosis in prolonged maintenance hemodialysis. N Eng J Med 1974; 290: 697–701, [CSA]
- Foley R, Parfrey PS, Sarnak MJ. Clinical epidemiology of cardiovascular disease in chronic renal disease. Am J Kidney Dis 1998; 32(suppl.)S112–119, [INFOTRIEVE], [CSA]
- Locatelli F, Canaud B, Eckardt K, Stenvinkel P, Wanner C, Zoccali C. Oxidative stress in end-stage renal disease: an emerging threat to patient outcome. Nephrol Dial Transplant 2003; 18: 1272–1280, [INFOTRIEVE], [CROSSREF], [CSA]
- Stenvinkel P, Alverstand A. Inflammation in end stage renal disease: sources, consequences and therapy. Semin Dial 2002; 15(5)329–337, [INFOTRIEVE], [CROSSREF], [CSA]
- Himmelfarb J, Stenvinkel P. İkizler TA, Hakım RM. The elephant in uremia: Oxidant stress as a unifying concept of cardiovascular disease in uremia. Kidney Int 2002; 62: 1524–1538, [INFOTRIEVE], [CROSSREF], [CSA]
- Descamps-Latscha B, Drüeke T, Witko-Sarsat V. Dialysis induced oxidative stress: biological aspects, clinical consequences, and therapy. Semin Dial 2001; 14: 193–199, [INFOTRIEVE], [CROSSREF], [CSA]
- Himmelfarb J. Linking oxidative stress and inflammation in kidney disease: Which is the chicken and which is the egg?. Semin Dial 2004; 17(6)449–454, [INFOTRIEVE], [CROSSREF], [CSA]
- Gordon CA, Himmelfarb J. Antioxidant therapy in uremia: evidence-based medicine?. Semin Dial 2004; 17: 327–332, [INFOTRIEVE], [CROSSREF], [CSA]
- Wang L, Brown RJ, Varki A, Esko DJ. Heparin's anti-inflammatory effects require glucoseamine 6-0-sulfation and are mediated by blockade of L- and P-selectins. J Clin Invest 2002; 110(1)127–136, [INFOTRIEVE], [CROSSREF], [CSA]
- Sela S, Shurtz-Swırskı R, Shapıro G, Nasser L, Hamzı M, Shasha S, Kristal B. Oxidative stress during hemodialysis: effect of heparin. Kidney Int 2001; 39(suppl. 78)159–163, [CROSSREF], [CSA]
- Satoh K. Serum lipid peroxide in cerebrovascular disorders determined by a new colorimetric method. Clin Chim Acta 1978; 90: 37–43, [INFOTRIEVE], [CROSSREF], [CSA]
- Sun Y, Oberley LW, Ying L. A simple method for clinical assay of superoxide dismutase. Clin Chem 1988; 34: 497–500, [INFOTRIEVE], [CSA]
- Stenvinkel P. Inflammation in end-stage renal failure: could it be treated?. Nephrol Dial Transplant 2002; 17(suppl. 8)33–38, [INFOTRIEVE], [CROSSREF], [CSA]
- Bergström J, Heimbürger O, Lindholm B, Qureshi AR. Elevated serum C-reactive protein is a strong predictor of increased mortality and low serum albumin in hemodialysis (HD) patients. J Am Soc Nephrol 1995; 6: 573, [CSA]
- Wanner C, Metzger T. C-reactive protein a marker for all-cause and cardiovascular mortality in hemodialysis patients. Nephrol Dial Transplant 2002; 17(suppl. 8)29–32, [INFOTRIEVE], [CROSSREF], [CSA]
- Arici M, Walls J. End-stage renal disease, atherosclerosis, and cardiovascular mortality: Is C-reactive protein the missing link?. Kidney Int 2001; 59(2)407–414, [INFOTRIEVE], [CROSSREF], [CSA]
- Tintut Y, Patel J, Parhami F, Demer LL. Tumor necrosis factor-α promotes in vitro calcification of vascular cells via the cAMP pathway. Circulation 2000; 102: 2636–2642, [INFOTRIEVE], [CSA]
- Yudkin JS, Kumari M, Humphries SE, Mohamed-Ali V. Inflammation, obesity, stress and coronary heart disease: Is interleukin-6 the link?. Atherosclerosis 2000; 148: 209–214, [INFOTRIEVE], [CROSSREF], [CSA]
- Hojo Y, Ikeda U, Takahashi M, Shimada K. Increased levels of monocyte-related cytokines in patients with unstable angina. Atherosclerosis 2002; 161: 403–408, [INFOTRIEVE], [CROSSREF], [CSA]
- Tsirpanlis G, Chatzipanagiotou S, Nicolaou C. Microinflammation versus inflammation in chronic renal failure patients. Kidney Int 2004; 66: 2093–2094, [INFOTRIEVE], [CROSSREF], [CSA]
- Boaz M, Mataz Z, Biro A, Katzir Z, Green M, Fainaru M, Smetana S. Serum malondialdehyde and prevalent cardiovascular disease in hemodialysis. Kidney Int 1999; 56: 1078–1083, [INFOTRIEVE], [CROSSREF], [CSA]
- Gritters M, Grooteman MP C, Schoorl M, et al. Citrate anticoagulation abolishes degranulation of polymorphonuclear cells and platelets and reduces oxidative stress during haemodialysis. Nephrol Dial Transplant 2006; 21: 153–159, [INFOTRIEVE], [CROSSREF], [CSA]
- Vignoli A, Marchetti M, Balducci D, Barbui T, Falanga A. Differential effect of the low-molecular-weight heparin, dalteparin, and unfractionated heparin on microvascular endothelial cell hemostatic properties. Haematologica 2006; 91(2)207–214, [INFOTRIEVE], [CSA]
- Nguyen-Khoa T, Massy ZA, De Bandt JP, Kebede M, Salama L, Lambrey G, Witko-Sarsat V, Drueke TB, Lacour B, Thevenin M. Oxidative stress and haemodialysis: role of inflammation and duration of dialysis treatment. Nephrol Dial Transplant 2001; 16: 335–340, [INFOTRIEVE], [CROSSREF], [CSA]
- Handelman GJ, Walter MF, Adhikarla R, Gross J, Dallal GE, Levin NW, Blumberg JB. Elevated plasma F2-isoprostanes in patients on long-term hemodialysis. Kidney Int 2001; 51: 1960–1966, [CROSSREF], [CSA]
- Ikizler TA, Morrow JD, Roberts LJ, Evanson JA, Becker B, Hakim RM, Shyr Y, Himmelfarb J. Plasma F2-isoprostane levels are elevated in chronic hemodialysis patients. Clin Nephrol 2002; 58: 190–197, [INFOTRIEVE], [CSA]
- Belch JJ, Chopra M, Hutchison S, Lorimer R, Sturrock RD, Forbes CD, Smith WE. Free radical pathology in chronic arterial disease. Free Radic Biol Med 1989; 6: 375–378, [INFOTRIEVE], [CROSSREF], [CSA]
- Prasad K, Gupta JB, Kalra J, Lee P, Mantha SV, Bharadwaj B. Oxidative stress as a mechanism of cardiac failure in chronic volume overload in canine model. J Mol Cell Cardiol 1996; 28: 375–385, [INFOTRIEVE], [CROSSREF], [CSA]