ABSTRACT
Effects of garlic (Allium sativum. L.) extract on the temporal patterns of circulatory lipid peroxidation products and antioxidants during N.-nitrosodiethylamine (NDEA)-induced hepatic tumorigenesis were investigated in Wistar rats. Experimental animals were divided into control, NDEA-treated, NDEA + garlic-treated, and garlic-treated. The characteristics of circadian rhythms (acrophase, amplitude, and mesor) of lipid peroxidation products (thiobarbituric acid reactive substances) and antioxidants (reduced glutathione, glutathione peroxidase, superoxide dismutase, and catalase) were analyzed. Alterations in the temporal characteristics of all variables were observed in NDEA-treated rats when compared to other groups. The alterations in the characteristics of these variables in NDEA-treated and in other groups deserve further investigation for the prognosis and therapeutic efficacy of chronotherapy of cancer.
Introduction
Circadian rhythms are temporal variations over a 24-h period in various biological processes that are regulated by an endogenous clock (suprachiasmatic nucleus) in mammals. Rhythm alterations of white blood cell (WBC) count and alkaline phosphatase were reported in tumor-bearing mice (Zvulunov et al., 2002). Cortisol and adrenocorticotrophic hormone were found to display marked 24-h rhythms in serum and saliva of humans, and these rhythms are also of prognostic value in metastatic breast cancer in human beings (Mormont et al., Citation2002). DNA synthesis (in bone marrow cells) in mice bearing squamous cell carcinoma cells was found to vary in a temporal manner over a 24-h period (differently) when compared to normal mice (Sun et al., Citation2002). Some tumor-preventing biological agents (interferons) possess the ability to modulate the biological clock at the genetic level (Koyanagi & Ohdo, Citation2002).
Hepatocellular carcinoma ranks fifth in frequency among all malignancies in the world (Graveel et al., Citation2001). N.-Nitrosodiethylamine (NDEA), a potent hepatocarcinogen, is a by-product of nitrosation of primary amines in the acidic conditions of stomach. It is further converted into an active ethyl radical metabolite that reacts with DNA, leading to mutations and oncogenesis (Anis et al., Citation2001).
Experimental, clinical, and epidemiological studies have provided evidence that supports the role of reactive oxygen species in the etiology of cancer (Ray et al., Citation2000). Further, circadian variations of plasma superoxide dismutase (SOD) and malondialdehyde in healthy subjects were studied (Luo et al., Citation1997). The temporal patterns of reduced glutathione and SOD have also been studied (Hodoglugil et al., Citation1995).
Garlic (Allium sativum. L.) is known to have medicinal properties including antimicrobial, antihypertensive, hypolipidemic, antidiabetic, hepatoprotective, immunomodulatory, antifungal, insecticidal, and antitumor activities (Lamm & Riggs, Citation2000; Shobana & Naidu, Citation2000). Epidemiological, animal, and in vitro. studies showed inhibition of several types of tumors and a decrease of tumor growth and proliferation during garlic treatment (Gao et al., Citation1999; Lamm & Riggs, Citation2000).
The concepts of chronobiology have been studied in oncology in an attempt to improve the therapeutic index of anticancer drugs (Bjarnasorn & Jordan, Citation2000). Circadian rhythms of a number of biological variables influence anticancer efficacy of cancer treatments (Levi, Citation2001). Furthermore, a lack of synchronization, or an alteration of circadian clock function, make rhythm peaks and troughs unpredictable and may require specific measures for chronotherapy to improve therapeutic index (Fu & Lee, Citation2003). Hence, assessment of the relevance of a normal circadian system for a favorable outcome of chronotherapy is desirable (Levi, Citation2001). Failure to recognize the biochemical temporal organization may introduce unnecessary heterogeneity and hinder the full understanding of tumorigenesis/biological process. It is in this context, in the current study, that the alterations in the circadian rhythm characteristics of lipid peroxidation products (thiobarbituric acid reactive substances; TBARS) and antioxidants, reduced glutathione (GSH), superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase (GPx), in control, tumor-bearing, and garlic-treated rats were analyzed.
Materials and Methods
Animals
Adult male Wistar rats (160–180 g) obtained from Central Animal House, Faculty of Medicine, Annamalai University, were used in the study. Commercial pellet diet (Hindustan Lever Ltd., Bangalore, India) and water were made available to animals ad libitum.. Animals were maintained in a controlled environment under standard conditions of humidity and temperature with alternating 12:12 light:dark cycles (Sundaresan & Subramanian, Citation2002; Citation2003). N.-Nitrosodiethylamine was purchased from Sigma Chemical Co. (St. Louis, MO, USA). All other chemicals and biochemicals used in the study were of analytical grade.
Preparation of garlic extract
Fresh garlic was purchased from a local market in Annamalainagar, Tamil Nadu, India. The plant was identified and authenticated by plant taxonomists of the botany department of our university. An aqueous extract of fresh garlic was prepared by homogenizing the required amount of freshly peeled garlic cloves in an appropriate volume of double-distilled water to give a concentration of 25 mg ml−1 (Sundaresan & Subramanian, Citation2002). The homogenate was centrifuged at 3000 rpm for 10 min. Particulate matter was removed, and the supernatant was used for the experiment within 30 min of preparation.
Treatment schedule
The animals were randomized and grouped into experimental and control rats (n = 6 in each group). Group I rats served as controls. Group II (NDEA + CCl4) rats received a single intraperitoneal injection of NDEA (200 mg/kg body weight) followed by weekly injections of CCl4 (3 ml/kg body weight) for 6 weeks (Sundaresan & Subramanian Citation2002; Citation2003). Group III rats received NDEA + CCl4 as in group II. In addition, they were orally treated with 1 ml of garlic extract in double-distilled water (Sundaresan & Subramanian, Citation2002). Group IV rats received garlic extract alone, as in group III.
Biochemical rhythms
Biochemical determinations listed in were performed after 20 weeks of experimental study. Blood samples were collected every 4 h from each group of rats (hours 0000, 0400, 0800, 1200, 1600, 2000, and 2400) throughout the 24-h period continuously. A minimal amount of blood was collected from orbital sinus with great care using heparinized tubes (Subramanian & Balamurugan, Citation1998; Subramanian et al., Citation2000). The values (mean±SD) obtained from each group were plotted versus the time of blood collection.
Table 1.. Twenty-four hour biochemical determinations.
The characteristics of circadian rhythms (acrophase, amplitude, and mesor) were analyzed by cosinor analysis (using the cosinorwin computer software program). Acrophase (time at which the level of variable is highest over a 24-h period) is expressed in hours. Amplitude (half the difference between maximum and minimum levels of the variable) and mesor (mean value of the variable for equidistant data covering a 24-h period) were expressed in the same units of documented variables.
Results
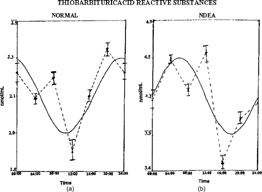
The biochemical variables chosen for the study in all the groups showed marked fluctuations over a 24-h period (). The characteristics of rhythms, r and p values indicating detectable rhythmicity or non-significant temporal variations over a 24-h period of all the groups are listed in Tables . The temporal patterns of TBARS revealed a detectable rhythmicity in groups I, III, and IV (). Detectable rhythmicity was found to be disturbed (p < 0.5) in group II rats. Acrophase was delayed in NDEA + CCl4 treated rats and advanced in groups III and IV when compared to controls (Fig.). Amplitude values in groups II and III showed increases and a decrease (in group IV) compared to controls.
Figure 1 Temporal patterns of TBARS in Wistar rats: (A) normal, (B) NDEA-treated, (C) NDEA+garlic-treated, (D) garlic treated. Dotted lines represent the raw data (with mean±SD) and smooth lines represent the best fitting cosinor curve (obtained using cosinorwin computer software program).
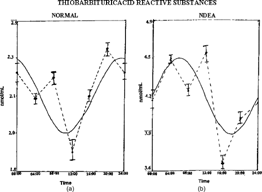
Figure 2 Twenty-four hour patterns of reduced glutathione in Wistar rats: (A) normal, (B) NDEA-treated, (C) NDEA+garlic-treated, (D) garlic-treated. Other details as in Figure 1.
Figure 3 Temporal oscillations of superoxide dismutase in Wistar rats: (A) normal, (B) NDEA-treated, (C) NDEA+garlic-treated, (D) garlic-treated. Other details as in Figure 1.
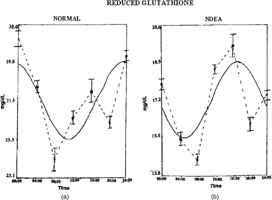
Figure 4 Rhythms of glutathione peroxidase in Wistar rats: (A) normal, (B) NDEA-treated, (C) NDEA+garlic-treated, (D) garlic-treated. Othr details as in Figure 1.
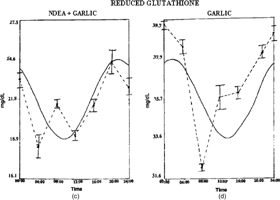
Figure 5 Temporal patterns of catalase in Wistar rats: (A) normal, (B) NDEA-treated, (C) NDEA+garlic-treated, (D) garlic-treated. Other details as in Figure 1.
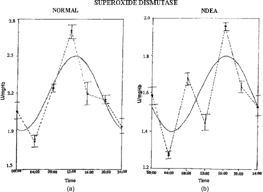
Table 2.. Changes in the temporal characteristics of thiobarbituric acid reactive substances (TBARS).
Table 3.. Changes in the temporal characteristics of reduced glutathione (GSH).
Table 4.. Changes in the temporal characteristics of superoxide dismutase (SOD).
Table 5.. Changes in the temporal characteristics of glutathione peroxidase (GPx).
Table 6.. Changes in the temporal characteristics of catalase (CAT).
The temporal pattern of GSH showed a detectable rhythm in control and groups III and IV (). Detectable rhythmicity was disturbed in NDEA + CCl4 treated rats. Acrophase was advanced in group II and delayed in groups III and IV when compared to controls (). Mesor values were decreased in group II and increased in groups III and IV (). The detectable circadian rhythms of SOD, GPx, and CAT were significant in groups I, III, and IV (Tables ). Acrophase was delayed in group II and advanced in groups III and IV () when compared to controls. Mesor values were decreased in group II and increased in groups III and IV (Tables ).
Discussion
The biochemical variables chosen in the study in the rat model system exhibit marked fluctuations over a 24-h period. Results of the study indicated that control, NDEA, and garlic treated rats differ in the temporal characteristics. Experimental models that mimic human tumors are needed to understand malignant processes and to further guide subsequent patient investigations (Granda & Levi, Citation2002).
Alterations in period, amplitude, mesor, and acrophase were detected in DNA synthesis of spleen, liver, and bone marrow of tumor-bearing mice (Sun et al., Citation2002). Our results also revealed that the rhythms in tumor-bearing animals are not synchronized with and did not exhibit a phasing of that of normal rats. This lack of synchronization reflected an alteration of circadian clock function in tumor-bearing animals and may require specific measures for chronotherapy to improve therapeutic index.
Lipid peroxidation and associated membrane damages are key features of NDEA-induced toxicity (Anis et al., Citation2001). Increase in lipid peroxidation indicated as TBARS in NDEA-treated rats might result in elevated mesor values. The temporal pattern of TBARS depends on the (i) nature of 24-h rhythms of total lipids, (ii) lipid peroxidation levels, and (iii) superoxide dismutase and catalase activities in blood (Subramanian et al., Citation2000). The altered acrophase observations in groups II to IV indicate that exogenous perturbations could influence the temporal organization of TBARS levels by influencing one or more factors mentioned above. Decrease in mesor value found in group III indicates that garlic extract could decrease the deleterious effects of NDEA by reducing the formation of TBARS.
The elevation of mesor values of antioxidants in garlic administered to NDEA treated rats showed that constituents of garlic could elevate the levels of nonenzymatic and enzymatic antioxidants (Hong et al., Citation1991). Glutathione (GSH) rhythm was reported in humans showing acrophase at 0830 and lowest concentration at 2030 (Smaaland et al., Citation2002). The peak of GSH content preceded the peak in DNA synthesis by about 4 h (Smaaland et al., Citation2002). These findings are consistent with other reports demonstrating a relationship between GSH and DNA synthesis when cells are activated into cell cycle progression or are in exponential growth (Lee et al., Citation1982). The earlier results support the theory that elevation of intracellular GSH content is a signal by which cells are activated into cell cycle progression (Smaaland et al., Citation2002). In the current study, the altered rhythmicity of GSH in tumor-bearing rats could reflect the cell cycle modulations as well.
Circadian rhythms of SOD and CAT were reported in liver and blood (Barros & Beetara, Citation2001). The decreased activities of SOD and CAT in NDEA-treated rats as reflected in decreased mesor values could be due to overuse of these enzymatic antioxidants to scavenge the products of lipid peroxidation. Our study also revealed that non-significant detectable rhythm characteristics in tumor-bearing rats could influence the temporal patterns of lipid peroxidation and other antioxidant levels in rats. In murine models, the extent of the rhythm disturbances appears to be related to tumor type, growth rate, and differentiation grade (Granda & Levi, Citation2002). Increased mesor values of SOD, CAT, and GPx observed in group III indicates that garlic extract may have roles in controlling the formation of free radicals by elevating the levels of antioxidants. Alterations in the circadian variation in the mitotic index of NDEA-induced hepatoma in rats were reported (Barbasen & Betz, Citation1980).
Cancer processes have been reported to alter circadian functions in experimental tumor models and patients, as several cancers produce growth factors and hormones that can modify central circadian system and/or peripheral circadian rhythms (Levi, Citation2001; Septan et al., Citation2001). Furthermore, the circadian time of administration of more than 30 anticancer agents were found to influence the extent of toxicity in mice and rats (Granda et al., Citation2001). Better use of anticancer agents may result from integrating a chronopharmacologic strategy at an early stage with drug development. Rhythms in target tissues, including cell division cycle and reduced glutathione, may rather account for the temporal changes that characterize the cytotoxicities of chronotherapeutic drugs as indicated previously (Boughattas et al., Citation1994).
Several circadian rhythms relevant to the treatment of cancer have been identified in rodents and humans (Levi, Citation2001). Adapting chemotherapy delivery to circadian rhythms has indeed achieved meaningful clinical improvements in chemotherapy tolerability and efficacy. The detectable circadian rhythms of lipid peroxidation products and antioxidants and their alterations during NDEA/garlic treatments, in the current study, deserve further investigation for the diagnosis, prognosis, and for the therapeutic efficacy of chronotherapy of cancer.
Acknowledgment
Financial assisantance from University Grants Commission, New Delhi (F.3–68/2002 (SR-II)), is gratefully acknowledged.
References
- Anis KV, Rajeshkumar NV, Kuttan R (2001): Inhibition of chemical carcinogenesis by biberine in rats and mice. J Pharm Pharmacol 53: 763–768. [PUBMED]
- Barbasen H, Betz EH (1980): Liver-cell after discontinuation of DENA feeding in hepatocarcinogenesis. Eur J Cancer 17: 149–156.
- Barros MP, Beetara EJ (2001): Daily variations of antioxidant enzymes and luciferase activities in luminescent chick beetle Pyrearinus termitilluminans.: Co-operation against oxygen toxicity. Insect Biochem Mol Biol 31: 393–400. [PUBMED], [INFOTRIEVE], [CSA], [CROSSREF]
- Bjarnasorn GA, Jordan R (2000): Circadian variation of cell proliferation and cell cycle protein expression in man: clinical implications. Prog Cell Cycle Res 4: 193–206. [CSA]
- Boughattas NA, Hecqnet C, Fournier B, Bruguerolle H, Trabelsi K, Bouzohita B, Omrange F, Levi F (1994): Comparative pharmocokinetics of oxaliplatin and carboplatin in mice with reference to circadian dosing time. Biopharm Drug Disposit 15: 1–13. [CSA]
- Ellman GL (1959): Tissue sulphydryl groups. Arch Biochem Biophys 82: 70–77. [PUBMED], [INFOTRIEVE], [CROSSREF]
- Fu L, Lee CC (2003): The circadian clock: Pacemaker and tumor suppressor. Nat Rev Cancer 3: 350–361. [PUBMED], [INFOTRIEVE], [CSA], [CROSSREF]
- Gao CM, Takezaki T, Ding JH, Li MS, Tajima K (1999): Protective effect of Allium. vegetables against both esophageal and stomach cancer: A simultaneous case reference study of a high epidemic area in Jiangsu. province, China. Jpn J Cancer Res 90: 614–621. [PUBMED], [INFOTRIEVE], [CSA]
- Granda TG, Levi F (2002): Tumor-based rhythms of anticancer efficacy in experimental models. Chronobiol Int 19: 21–41. [PUBMED], [INFOTRIEVE], [CSA], [CROSSREF]
- Granda TG, Filipski E, Attino D (2001): Experimental chronotherapy of mouse mammary adenocarcinoma MA13/C with docetaxel and doxorubicin as single agents and in combination. Cancer Res 61: 1996–2001. [PUBMED], [INFOTRIEVE]
- Graveel CR, Jatkoe T, Madore SJ, Holt Al, Farnham DJ (2001): Expression profiling and identification of novel genesis of hepatocellular carcinoma. Oncogenes 20: 2704–2714.
- Hodoglugil U, Ongun O, Guney Z, Altan N, Zenil H (1995): Temporal variation in hepatic superoxide dismutase activity in mice. Chronobiol Int 12: 152–155. [PUBMED], [INFOTRIEVE], [CSA]
- Hong JY, Smith T, Lee MJ, Li WS, Ma BL, Ning SM, Brady JF, Thomas PE, Yang CS (1991): Metabolism of carcinogenic nitrosamines by rat nasal mucosa and the effect of diallyl sulphide. Cancer Res 51: 1509–1514. [PUBMED], [INFOTRIEVE]
- Koyanagi S, Ohdo S (2002): Alteration of intrinsic biological rhythms during interferon treatment and its possible mechanism. Mol Pharmacol 62: 1393–1399. [PUBMED], [INFOTRIEVE], [CSA], [CROSSREF]
- Kakkar P, Das B, Viswanathan PN (1984): A modified spectrophotometric assay of superoxide dismutase. Indian J Biochem Biophys 21: 130–132. [PUBMED], [INFOTRIEVE], [CSA]
- Lamm DL, Riggs DR (2000): The potential application of Allium sativum. (garlic) for the treatment of bladder cancer. Urol Clin North Am 27: 157–162. [PUBMED], [INFOTRIEVE], [CSA]
- Lee FY, Allalunis M, Turner MJ, Siemann DW (1982): Depletion of tumor versus normal tissue glutathione by butathione sulfoximide. Br J Cancer 56: 33–37.
- Levi F (2001): Circadian chronotherapy. Lancet Oncol 2: 307–315. [PUBMED], [INFOTRIEVE], [CSA], [CROSSREF]
- Levi F, Zidane R, Brieaza SA (1999): Multicentre evalution of intensified ambulatory chronomodulated chemotherapy with oxaliplatin, fluorouracil and leucovarin as initial treatment of patients with metastatic colorectal cancer. Cancer 85: 2532–2540. [PUBMED], [INFOTRIEVE], [CROSSREF]
- Luo H, Guo H, Xiao J, Xue Z (1997): Circadian variations of plasma SOD and MDA in healthy subjects. Hua Xi Da Xue Xue Bao 28: 401–403. [CSA]
- Mormont MC, Langouet AM, Claustrat B, Bogdan A, Marion S, Waterhouse J, Touitou Y, Levi F (2002): Marker rhythms of circadian system function: A study of patients with metastasic colorectal cancer and good performance status. Chronobiol Int 19: 141–155. [PUBMED], [INFOTRIEVE], [CSA], [CROSSREF]
- Ray G, Batra S, Shukla NK, Deo S, Raina V, Ashok S, Husain SA (2000): Lipid peroxidation, free radicals production and antioxidant status in breast cancer. Breast Cancer Res Treat 59: 163–170. [PUBMED], [INFOTRIEVE], [CSA], [CROSSREF]
- Rotruck JT, Pope AH, Ganther HE, Suransan AB, Hofeman DG, Hoekstra WG (1973): Selenium: Biochemical roles as a component of glutathione peroxidase. Science 179: 588–590. [PUBMED], [INFOTRIEVE]
- Septan SE, Sapolsky RM, Kremer HC, Spiegel D (2001): Diurnal cortisol rhythm as a predictor of breast cancer survival. J Natl Cancer Inst 92: 994–1000.
- Shobana S, Naidu A (2000): Antioxidant activity of selected Indian spices. Prostaglandins Leukot Essen Fatty Acids 62: 107–110. [CSA], [CROSSREF]
- Smaaland R, Southern RB, Laerum OD, Abraham JF (2002): Rhythms in human bone marrow and blood cells. Chronobiol Int 19: 101–127. [PUBMED], [INFOTRIEVE], [CSA], [CROSSREF]
- Subramanian P, Balamurugan E (1998): Temporal oscillations of serum electrolytes in n-phthaloyl GABA treated rats. Pharmacol Biochem Behav 62: 511–514. [CSA], [CROSSREF]
- Subramanian P, Sivabalan S, Menon VP, Vasudevan K (2000): Influence of chronic zinc supplementation on biochemical variables and circadian rhythms in Wistar rats. Nutr Res 20: 413–425. [CROSSREF]
- Sundaresan S, Subramanian P (2002): Evaluation of chemopreventive potential of garlic extract in N.-nitrosodiethylamine-induced carcinogenesis. Pharm Biol 40: 548–551.
- Sundaresan S, Subramanian P (2003): S.-Allylcysteine inhibits circulatory lipid peroxidation and promotes antioxidants in N.-nitrosodiethylamine induced carcinogensis. Pol J Pharmacol 55: 37–42. [PUBMED], [INFOTRIEVE], [CSA]
- Sun J, Xian LJ, Cao QY, Ye YL, Zeng ZL, Liu XH, Li XM, Levi F (2002): Circadian rhythms of DNA synthesis and apotosis correlated genes expression in bone marrow cells of nude mice bearing human nasopharyngeal carcinoma. Ai Zheng 21: 833–837. [PUBMED], [INFOTRIEVE], [CSA]
- Yagi K (1978): Lipid peroxides and human disease. Chem Physiol Lipids 45: 3337–3351. [CSA]
- Zvulunov A, Peleg L, Carlebach R, Ashkenazi IE (2003): Chemotoxicity and survival of tumor-bearing mice under exposure to randomized photoperiodic regimen. Life Sci 72: 237–245. [CROSSREF]