Abstract
Background
Glucagon-like peptide-1 receptor agonists (GLP-1RAs) act by increasing insulin secretion, decreasing glucagon secretion, slowing gastric emptying, and increasing satiety.
Objective
Published evidence directly comparing GLP-1RAs with other approved treatments for type 2 diabetes (T2D) was systematically reviewed.
Methods
A literature search was performed using MEDLINE and Embase databases to identify papers comparing GLP-1RAs with other classes of glucose-lowering therapy in patients with T2D.
Results
Of the 1303 papers identified, 57 met the prespecified criteria for a high-quality clinical trial or retrospective study. The efficacy and tolerability of approved GLP-1RAs (exenatide twice daily or once weekly, dulaglutide, liraglutide, lixisenatide, and albiglutide) were compared with insulin products (23 prospective studies + seven retrospective studies), dipeptidyl peptidase-4 inhibitors (11 prospective studies + three retrospective studies), sulfonylureas (nine prospective studies + one retrospective study), thiazolidinediones (five prospective studies), and metformin (two prospective studies). GLP-1RAs are effective as a second-line therapy in improving glycemic parameters in patients with T2D. Reductions in glycated hemoglobin from baseline with GLP-1RAs tended to be greater or similar compared with insulin therapy. GLP-1RAs were consistently more effective in reducing body weight than most oral glucose-lowering drugs and insulin and were associated with lower hypoglycemia risk versus insulin or sulfonylureas. GLP-1RAs improved cardiovascular risk factors, and preliminary data suggest they improve cardiovascular outcomes in patients with T2D compared with oral glucose-lowering drugs. However, results from ongoing studies are awaited to confirm these early findings.
Conclusion
This systematic review found that GLP-1RAs are an effective class of glucose-lowering drugs for T2D.
Introduction
Diabetes mellitus is a chronic disease affecting a substantial proportion of the population.Citation1 In adults (age 20–79 years), the 2015 prevalence of diabetes worldwide was estimated at 8.8%, with type 2 diabetes (T2D) comprising 91% of cases.Citation2 By 2030, diabetes is expected to be the seventh leading cause of death.Citation3
Several classes of glucose-lowering agents are currently available for the treatment of T2D, each with different mechanisms of action and therapeutic effects. Glucagon-like peptide-1 receptor agonists (GLP-1RAs) are a class of glucose-lowering drugs that act on the glucagon-like peptide-1 (GLP-1) receptor on pancreatic beta cells and increase insulin secretion, decrease glucagon secretion, slow gastric emptying, and increase satiety; clinical trials have shown that GLP-1RAs decrease body weight, postprandial glucose excursions, and some cardiovascular risk factors, without increasing the risk of hypoglycemia.Citation4 Clinical trials have also studied the effects of GLP-1RA therapy on cardiovascular outcomes in patients with T2D.Citation5,Citation6
The purpose of this systematic review was to compare the efficacy of GLP-1RAs with other glucose-lowering therapies, using comparative data from clinical trials or observational cohort studies.
Literature search strategy and filtering
Embase and MEDLINE databases were searched on April 8, 2016, using the following search string: (glucagon like peptide 1 receptor agonist or “Glucagon-Like Peptide 1”) OR ([“Glucagon-like peptide-1” or “Glucagon-like peptide 1” or GLP1R or GLP-1 or GLP-1-R] AND [agonist or suppress* or block* or inhibit* or anti* or mimetic]) OR (lixisenatide or exenatide or liraglutide or albiglutide or dulaglutide) AND ([diabetes or diabetic or DM] and [“Type 2” or “Type-2” or “Type2” or “Type II” or “Type-II” or “Noninsulin-dependent” or “Noninsulin dependent” or “Non insulin-dependent” or “Non insulin dependent”]).
The search string was limited to keywords in the abstract or title. Articles indexed as animal studies, case reports, books, and conference/symposium presentations were excluded. The limits applied were English language and articles published between January 2005 and April 2016. Duplicates were removed, leaving 1303 articles. The articles identified were filtered manually and only those describing a head-to-head comparison between a GLP-1RA and another class of glucose-lowering therapy in ≥100 patients, regardless of study design, follow-up duration, or medication doses, were included in the final total.
A total of 57 articles were included in the analysis (); of these, 37 were randomized controlled trials (RCTs), seven were open-label extensions of RCTs, 11 were retrospective analyses, and two were prospective observational studies.
Efficacy of GLP-1RAs versus other glucose-lowering therapies
GLP-1RAs versus dipeptidyl peptidase-4 inhibitors (DPP-4is)
Eleven prospective studiesCitation7–Citation17 and three retrospective studies compared GLP-1RAs with DPP-4is.Citation18–Citation20
Prospective studies
Exenatide once weekly (QW) was associated with significantly greater (P < 0.001) reductions in glycated hemoglobin (HbA1c) and fasting glucose (FG) compared with sitagliptin after 26 weeks in the Diabetes Therapy Utilization: Researching Changes in A1C, Weight and Other Factors Through Intervention with Exenatide Once Weekly (DURATION)-2 and DURATION-4 studies ().Citation8,Citation9 Although both exenatide QW and sitagliptin recipients lost weight, patients receiving exenatide QW had significantly greater weight loss from baseline.Citation8,Citation9 One study comparing exenatide twice daily (BID) with sitagliptin found that exenatide BID recipients had a reduction in FG similar to sitagliptin recipients, but a significantly greater reduction in weight.Citation7
Table 1 Study details and efficacy results of comparative trials of GLP-1RAs and oral glucose-lowering therapies
In the Assessment of Weekly Administration of LY2189265 in Diabetes (AWARD)-5 study, after 52 weeks of treatment, reductions from baseline in HbA1c, FG, and weight were significantly greater with dulaglutide than with sitagliptin ().Citation10 These benefits were sustained over 104 weeks of treatment.Citation11
Generally, liraglutide-treated patients had greater or similar reductions from baseline in HbA1c, FG, and weight compared with sitagliptin- or vildagliptin-treated patients ().Citation12–Citation14,Citation16 In an open-label extension study, patients switching from sitagliptin to liraglutide had further reductions in these parameters.Citation15
When administered for 24 weeks, lixisenatide produced reductions from baseline in HbA1c and FG that were similar to sitagliptin ();Citation17 however, weight loss was significantly greater among lixisenatide versus sitagliptin recipients.
Hypoglycemia rates in patients receiving GLP-1RAs or DPP-4is were low, with only one instance of major/severe hypoglycemia reported across all studies (in a patient receiving liraglutide 1.2 mg).Citation13,Citation14 In studies of exenatide QW, minor hypoglycemia rates ranged from 1% to 3.6% with exenatide QW, with the highest hypoglycemia rates for concomitant sulfonylurea use, and from 0% to 3.0% with DPP-4is.Citation7–Citation9 Patients receiving dulaglutide 1.5 mg generally had numerically higher hypoglycemia rates than patients receiving dulaglutide 0.75 mg or sitagliptin (10.2% vs 5.3% and 4.8%; 12.8% vs 8.6% and 8.6%, respectively).Citation10,Citation11 Rates of hypoglycemia were similar for all liraglutide doses investigated versus DPP-4i comparators.Citation12–Citation14,Citation16 Furthermore, the incidence of gastrointestinal adverse events (AEs; nausea, diarrhea, and vomiting) was higherCitation7–Citation11,Citation13–Citation15,Citation17 or similarCitation16 in patients receiving GLP-1RAs versus DPP-4is; gastrointestinal AEs led to treatment withdrawal in a higher percentage of patients receiving GLP-1RA therapy in these studies.Citation8,Citation10,Citation11 Some studies reported that gastrointestinal AEs peaked on treatment initiation in the GLP-1RA group and then stabilized over the study period.Citation10,Citation11,Citation13
Retrospective studies
Retrospective studies had similar results to the prospective studies (), with GLP-1RAs demonstrating greater or similar reductions in HbA1c, FG, and weight versus DPP-4is.Citation18–Citation20 Generally, hypoglycemia rates were not reported, although in one study the rate was similar between GLP-1RAs and DPP-4is.Citation19 Nausea/vomiting was more frequent with exenatide compared with sitagliptin (incidence rate, 0.39 vs 0.032) in one study.Citation19
Table 2 Study details and efficacy results of retrospective studies of GLP-1RAs and oral glucose-lowering therapies or insulin
GLP-1RAs versus metformin
Two prospective studies comparing GLP-1RAs and metfor-min were identified.Citation9,Citation21
Prospective studies
Both studies (RCTs) reported reductions from baseline in HbA1c, FG, and weight ().Citation9,Citation21 In DURATION-4, exenatide QW and metformin reduced HbA1c from baseline by ~1.5%, with no significant difference between groups at 26 weeks. In AWARD-3, dulaglutide 0.75 mg and 1.5 mg QW reduced HbA1c significantly more than metformin (P < 0.05) after 26 weeks. After 52 weeks, significant differences were observed in the reduction from baseline in HbA1c between dulaglutide 1.5 mg and metformin, but not between dulaglutide 0.75 mg and metformin.
Two RCTs investigated the effects of exenatide QW and dulaglutide on FG and weight, with no significant differences between these drugs in the reductions of these endpoints at 26 weeks.Citation9,Citation21 Treatment with dulaglutide at both dosages resulted in similar reductions in FG versus metformin at 26 weeks; at 52 weeks, recipients of dulaglutide 1.5 mg had a significantly greater reduction in FG than metformin recipients (P < 0.05; ). The magnitude of weight loss achieved in AWARD-3 was similar between the dulaglutide 1.5 mg and metformin groups at 26 and 52 weeks; dulaglutide 0.75 mg recipients lost significantly less weight than patients receiving metformin at both time points.
Minor hypoglycemia was reported in 2.0% of exenatide QW recipients versus 0.0% of metformin recipients, while dulaglutide and metformin recipients had similar total hypoglycemia rates (12.3%, 11.1%, and 12.7% for dulaglutide 1.5 mg, dulaglutide 0.75 mg, and metformin, respectively).Citation9,Citation21 The incidence of gastrointestinal AEs was similar between treatment groups in AWARD-3,Citation21 whereas in DURATION-4, nausea was reported in 11.3% and vomiting in 4.8% of exenatide-treated patients compared with 3.3% who experienced vomiting on metformin.Citation9
GLP-1RAs versus sulfonylureas
Nine prospective studies investigated the comparative efficacy of GLP-1RAs and sulfonylureas.Citation22–Citation30 One retrospective study was identified: a comparison of liraglutide and glimepiride in outpatients in Italy.Citation31
Prospective studies
Generally, exenatide BID recipients had greater or similar reductions from baseline in HbA1c, FG, and weight compared with glyburide or glimepiride recipients ().Citation22–Citation24 Furthermore, glycemic control was maintained for a longer duration with exenatide BID compared with glimepiride in the European Exenatide Study (EUREXA).Citation24 Compared with glimepiride, liraglutide resulted in greater or similar reductions in HbA1c and FG, and consistently greater reductions in weight.Citation25,Citation26,Citation28,Citation32 Similar results were seen when liraglutide was compared with glyburide, with greater or similar reductions from baseline in HbA1c and FG; however, weight loss with liraglutide was not consistently greater versus glyburide.Citation27,Citation30
Patients receiving GLP-1RAs generally had significantly (P < 0.05) lower rates of hypoglycemia than sulfonylurea-treated patients;Citation22–Citation30 in cases where the P value was not stated, the rates were numerically different in favor of the GLP-1RA. The incidence of gastrointestinal AEs was generally higher with GLP-1RAs compared with sulfonylureas;Citation25–Citation28,Citation30 however, one study reported similar incidence of gastrointestinal AEs with exenatide and glibenclamide.Citation22 The incidence of treatment withdrawal due to gastrointestinal AEs was also higher with GLP-1RAs compared with sulfonylureas;Citation22–Citation24,Citation32 differences in rates of gastrointestinal AEs were seen within 4 weeks in three studiesCitation29,Citation30 and within 6 months in another study.Citation24
Retrospective studies
In the retrospective analysis, liraglutide produced significantly greater reductions in HbA1c and FG from baseline compared with glimepiride (; P < 0.001).Citation31 Liraglutide recipients also lost significantly more weight than glimepiride-treated patients (P < 0.001). As in the prospective studies, rates of hypoglycemia were lower with liraglutide than glimepiride.Citation31 The incidence of gastrointestinal AEs was higher in the liraglutide group compared with the glimepiride group (P < 0.001).Citation31
GLP-1RAs versus thiazolidinediones
Five prospective studies compared GLP-1RAs with thiazolidinediones.Citation8,Citation9,Citation33–Citation35
Prospective studies
Exenatide BID produced similar reductions in HbA1c to rosiglitazone and significantly greater reductions than pioglitazone (P < 0.01; ).Citation34,Citation35 Similar reductions in FG were seen between exenatide BID and rosiglitazone and pioglitazone. Patients receiving exenatide BID had significantly greater weight loss than those receiving thiazolidinediones (P < 0.0001); in both studies, exenatide BID recipients had weight loss of ~3 kg, while patients receiving rosiglitazone gained weight and patients receiving pioglitazone had no change in weight.
Studies of exenatide QW showed that reductions from baseline in HbA1c, FG, and weight were either greater or similar than with thiazolidinedione comparators ().Citation8,Citation9 In the DURATION-2 study, exenatide QW recipients had significantly greater reductions in HbA1c versus pioglitazone, while in DURATION-4, reductions in HbA1c with exenatide QW and pioglitazone were similar; reductions in FG with exenatide QW and pioglitazone were similar. Significant reductions in weight were seen with exenatide QW in DURATION-2 and -4, whereas patients receiving pioglitazone gained weight (as expected in the presence of a sulfonylurea) in both studies (P < 0.0001 between groups).
The Liraglutide Effect and Action in Diabetes (LEAD)-1 SU study showed that liraglutide (0.6, 1.2, and 1.8 mg/day) was generally more effective as a second-line therapy in improving glycemic parameters and weight than rosiglitazone ().Citation33 Liraglutide 1.2 mg and 1.8 mg was associated with significantly greater reductions in HbA1c (P < 0.0001) and FG (P < 0.001) than rosiglitazone. Recipients of lira-glutide 0.6 mg or 1.2 mg and rosiglitazone gained weight, whereas recipients of liraglutide 1.8 mg lost weight (); however, the weight gain with the lower dosages of liraglutide was significantly less than that with rosiglitazone, so the differences from baseline in weight were significant between rosiglitazone and all doses investigated (P < 0.0001).
Overall, the rate of hypoglycemia seen with GLP-1RAs was slightly greater than or similar to that seen with thiazolidinediones.Citation8,Citation9,Citation33–Citation35 The incidence of gastrointestinal AEs was higher with GLP-1RAs compared with thiazolidinediones.Citation8,Citation9,Citation33–Citation35 Gastrointestinal AEs led to treatment withdrawal in a higher number of patients in the exenatide group compared with thiazolidinediones in one study.Citation35
GLP-1RAs versus insulin products
Twenty-three prospective studiesCitation35–Citation57 and seven retrospective studiesCitation18,Citation58–Citation63 compared GLP-1RAs with insulin products.
Prospective studies
Four trials compared GLP-1RAs and insulin aspart ().Citation38,Citation47,Citation54,Citation55 Exenatide studies showed that exenatide BID produced a similar or lesser reduction in HbA1c and FG than insulin aspart.Citation38,Citation47,Citation55 In contrast, exenatide BID recipients lost weight, while those using insulin aspart gained weight. In two of the three exenatide studies, the between-group difference in weight was significant (P < 0.001).
Table 3 Study details and efficacy results of prospective (randomized controlled and noninterventional) trials of GLP-1RAs versus insulin products
In the BEGIN™: VICTOZA® ADD-ON study, liraglutide significantly reduced HbA1c and weight versus insulin aspart, while reductions in FG were similar between treatments ().Citation54 When liraglutide 1.8 mg was compared with insulin degludec, the reductions in HbA1c and FG were similar between groups; patients receiving liraglutide lost weight while patients receiving insulin gained weight.Citation50
After 26 weeks of treatment, exenatide QW recipients had a significantly greater reduction from baseline in HbA1c than insulin detemir recipients and similar reductions in FG. Similar to liraglutide, those receiving exenatide lost weight, while insulin detemir recipients gained weight (between-group difference, P < 0.0001; ).Citation41
Twelve studies compared GLP-1RAs with insulin glargine ().Citation36,Citation37,Citation39,Citation40,Citation42,Citation44–Citation46,Citation48,Citation51,Citation52,Citation57 In the exenatide (BID and QW) studies, reductions from baseline in HbA1c were greater or similar to those with insulin glargine, whereas reductions in FG were similar or smaller (). Furthermore, a significant difference was generally observed between the weight loss with exenatide and the weight gain with insulin glargine ().
In the AWARD-4 study, compared with insulin glargine, treatment with dulaglutide for 26 weeks resulted in significantly greater reductions in HbA1c in patients also receiving insulin lispro and significantly smaller reductions in FG.Citation39 Patients receiving dulaglutide 1.5 mg lost weight versus baseline, while recipients of dulaglutide 0.75 mg and insulin glargine gained weight; however, the differences between the dulaglutide and insulin glargine groups were significant, irrespective of dulaglutide dose ().
The comparisons between liraglutide and insulin glargine in the Efficacy Assessment of Insulin Glargine Versus Liraglutide After Oral Agents Failure (EAGLE) and LEAD-5 studies were inconsistent with regard to HbA1c and FG; in the EAGLE study, liraglutide resulted in a lesser reduction in HbA1c and FG than insulin glargine, whereas in the LEAD-5 study, the reduction in HbA1c was greater with liraglutide versus insulin glargine and the reduction in FG was similar between groups.Citation40,Citation57 The change in weight was consistent between the two studies, with liraglutide recipients losing weight and insulin glargine recipients gaining weight ().
Two studies compared exenatide BID with insulin lispro ().Citation35,Citation43 In both studies, exenatide BID and insulin lispro reduced HbA1c from baseline to a similar degree. Reductions from baseline in FG were greater or similar with exenatide BID versus insulin lispro; exenatide BID treatment consistently resulted in weight loss, while insulin lispro recipients consistently gained weight.
Generally, the rate of hypoglycemia was lower with GLP-1RAs versus insulin products,Citation35,Citation38–Citation40,Citation43,Citation46,Citation47 or similar between groups.Citation41,Citation55,Citation57 Overall, the incidence of gastrointestinal AEs among GLP-1RA-treated patients was higher than in insulin-treated patients;Citation37–Citation42,Citation44–Citation47,Citation50–Citation52,Citation54,Citation55,Citation57 gastrointestinal AEs were generally observed on initiation of treatment with GLP-1RAsCitation39,Citation44,Citation45,Citation47,Citation50,Citation57 and sometimes led to treatment withdrawal.Citation37,Citation39,Citation42,Citation47,Citation52
Retrospective studies
Seven retrospective studies compared GLP-1RAs and insulin products (),Citation18,Citation58–Citation63 with similar results to the prospective studies; the reduction in HbA1c was greater with GLP-1RAs than insulin in most studies. Where reported, reductions from baseline in FG were not statistically different between groups. In studies in which the change in weight was reported, GLP-1RA treatment resulted in weight loss, while insulin treatment resulted in weight gain.
Where reported, the incidence of hypoglycemia was either similar between GLP-1RA and insulin treatment groups or lower in those receiving GLP-1RAs.Citation58,Citation60–Citation63 Treatment-related nausea was reported in 10 patients in the exenatide group in one study and did not lead to treatment discontinuation.Citation63
Effect of GLP-1RAs versus other glucose-lowering therapies on cardiovascular risk factors
The comparative studies identified by the literature search reported only limited information on cardiovascular risk factors and did not assess cardiovascular outcomes.
GLP-1RAs versus DPP-4is
Of the 10 prospective trials of GLP-1RAs and DPP-4is, all reported effects on blood pressure (BP) and/or lipid cardiovascular risk factors (Table S1).Citation7–Citation16 Two retrospective studies comparing GLP-1RAs with DPP-4is also reported changes in cardiovascular risk factors (Table S2).Citation18,Citation20 Changes in both diastolic BP and systolic BP (SBP) were variable for GLP-1RAs and DPP-4is, and changes in lipids were generally not significantly different between treatment groups. However, in AWARD-5, dulaglutide 1.5 mg resulted in a significantly greater reduction in low-density lipoprotein cholesterol compared with sitagliptinCitation10 (Table S1).
GLP-1RAs versus metformin
Two studies reported data on cardiovascular risk factors after treatment with GLP-1RAs compared with metformin, neither of which showed any clinically significant between-group differences in cardiovascular risk factors (Table S1).Citation9,Citation21
GLP-1RAs versus sulfonylureas
Seven of the nine studies comparing GLP-1RAs with sulfonylureas reported data on cardiovascular risk factors (Table S1),Citation24–Citation28,Citation30,Citation32 along with one retrospective study (Table S2).Citation31 In general, results indicated that the GLP-1RAs were associated with greater reductions in SBP than the sulfo-nylureas, with numerical reductions in various lipids observed in several studies comparing GLP-1RAs with sulfonylureas.
GLP-1RAs versus thiazolidinediones
All five prospective studies comparing GLP-1RAs with thiazolidinediones reported reductions from baseline in BP or fasting lipids (Table S1).Citation8,Citation9,Citation33–Citation35 Exenatide significantly reduced measures of cholesterol versus thiazolidinediones in three of these studies.Citation8,Citation34,Citation35
GLP-1RAs versus insulin
Two of four trials comparing GLP-1RAs and insulin aspart reported changes in cardiovascular risk factors (Table S3).Citation54,Citation55 One trial reported a significantly smaller increase in high-density lipoprotein cholesterol with exenatide BID versus insulin aspart.Citation55
Eight of ten prospective studies and two extension studies comparing GLP-1RAs with insulin glargine reported changes in cardiovascular risk factors (Table S3).Citation36,Citation39,Citation40,Citation42,Citation44–Citation46,Citation48,Citation52,Citation57 Overall, numerical reductions in SBP were observed in most of these studies, with greater reductions generally seen with GLP-1RAs than for insulin glargine; significant differences between treatments for fasting lipids also generally favored GLP-1RAs.
Exenatide BID was compared with insulin lispro in two studies (Table S3).Citation35,Citation43 No significant differences between treatment groups were reported for changes in BP or lipids.
Of the seven retrospective studies comparing GLP-1RAs and insulin products, four reported data on cardiovascular risk factors (Table S2).Citation18,Citation61–Citation63 Numerical reductions in BP and lipid measures were observed across exenatide and insulin therapies.
Discussion
GLP-1RAs represent an effective therapeutic option for patients with T2D. Currently, multiple studies provide data on the efficacy of approved GLP-1RAs in T2D (). Although their glycemic efficacy is well established versus multiple classes of agents, including insulin, albeit with a notable lack of comparative studies versus sodium-glucose cotransporter 2 inhibitors, GLP-1RAs have additional benefits of modest weight loss and a favorable tolerability profile ().Citation64 These beneficial effects of GLP-1RAs should be considered in context with the increased frequency of nausea with GLP-1RAs, which may limit adherence, and the need for self-injection on a once- or twice-daily or weekly basis.
Figure 2 Efficacy of glucose-lowering treatments in type 2 diabetes. Changes from baseline in (A) glycated hemoglobin (HbA1c), (B) fasting glucose (FG), and (C) body weight (BW) in prospective studies comparing glucagon-like peptide-1 receptor agonists (GLP-1RAs) with other oral glucose-lowering therapies and changes from baseline in (D) HbA1c, (E) FG, and (F) BW in prospective studies comparing GLP-1RAs with insulin products.
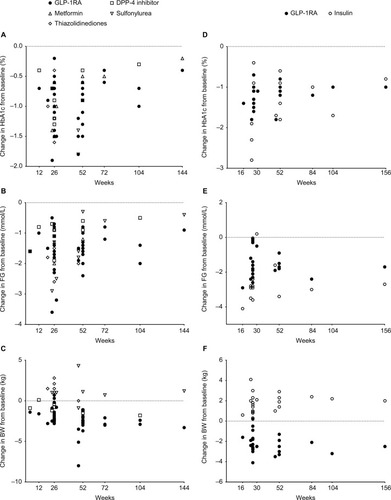
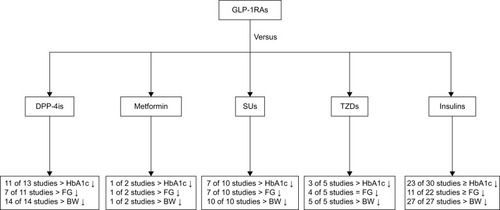
Figure 3 Comparison of efficacy of GLP-1RAs with other glucose-lowering treatments in type 2 diabetes. General trends in glycemic parameters and body weight (BW) in comparative trials of glucagon-like peptide-1 receptor agonists (GLP-1RAs) and other glucose-lowering therapies. The total number of studies includes studies that reported these parameters.
The efficacy of GLP-1RAs is generally greater than DPP-4is, owing to the supraphysiological concentrations of GLP-1 after administration of the former.Citation65 This effect on incretin also accounts for the greater weight loss experienced by patients who receive GLP-1RAs versus the neutral weight effects produced by DPP-4is.Citation65,Citation66 Glycemic control was also mostly similar to that with insulin, largely driven by improvements in postprandial glucose with the short-acting GLP-1RAs, with similar effects on FG as insulin. However, real-world evidence suggests similar glycemic reductions between GLP-1RAs and insulin and within the GLP-1RA class. A real-world study in patients with T2D reported that addition of exenatide BID to basal insulin was as effective as addition of mealtime insulin in reducing HbA1c levels in these patients, with significant reductions in weight (P < 0.01) and hypoglycemia (P < 0.03) compared with mealtime insulin.Citation67
Data comparing the effects of GLP-1RAs and other classes of glucose-lowering therapy on cardiovascular risk factors were limited but showed favorable effects on BP and lipid levels. Increases in heart rate have been observed with GLP-1RAs in clinical trials, although the underlying mechanisms and clinical relevance of these increases have yet to be established.Citation68 The size and duration of increases in mean 24-hour heart rate vary between GLP-1RAs, ranging from transient (1–12 hours) increases of 1–3 beats per minute (bpm) with the short-acting GLP-1RAs exenatide BID and lixisenatide to more prolonged increases during treatment with longer-acting GLP-1RAs (3–4 bpm with exenatide QW or dulaglutide and 6–10 bpm with liraglutide or albiglutide).Citation68 Data from cardiovascular outcome trials indicate that the observed increases in heart rate were not associated with an adverse effect on cardiovascular outcomes in patients with T2D.Citation5,Citation6,Citation68,Citation69
Limited clinical data are available regarding the effects of the various GLP-1RAs on cardiovascular outcomes, and none of these studies compared a GLP-1RA with another class of glucose-lowering therapy. The first published study with prospective outcomes data was the Evaluation of Lixisenatide in Acute Coronary Syndrome (ELIXA) trial,Citation70 which found no significant difference in rates of cardiovascular events with lixisenatide versus placebo.Citation6 A similar study investigating the effects of liraglutide on cardiovascular outcomes, the Liraglutide Effect and Action in Diabetes: Evaluation of Cardiovascular Outcome Results (LEADER) trial, showed improved effects of liraglutide on cardiovascular outcomes versus placebo.Citation5 Finally, the Trial to Evaluate Cardiovascular and Other Long-term Outcomes With Semaglutide in Subjects With Type 2 Diabetes (SUSTAIN-6), which evaluated the effect of semaglutide (added on to standard care) on cardiovascular outcomes, demonstrated the noninferiority of semaglutide to placebo, with a significant reduction in cardiovascular death, nonfatal stroke, or nonfatal myocardial infarction in the semaglutide group.Citation69
Results of the EXenatide Study of Cardiovascular Event Lowering (EXSCEL) trial, evaluating the effect of exenatide QW on major adverse cardiovascular events (MACE) when given in addition to usual care;Citation71 the Researching Cardiovascular Events With a Weekly Incretin in Diabetes (REWIND) study, evaluating the effects of dulaglutide on MACE;Citation72 and the HARMONY Outcomes study, evaluating the effects of albiglutide on MACE,Citation73 are all awaited and are expected to help contribute to the existing evidence regarding the impact of the GLP-1RAs on cardiovascular outcomes.
With the limited availability of prospective data, we can glean some information from meta-analyses and pooled analyses that have been conducted in an attempt to elucidate the effects of GLP-1RAs on cardiovascular outcomes. For example, a prespecified meta-analysis, which evaluated cardiovascular risk in the dulaglutide clinical development program,Citation74 indicated that there were no significant differences between dulaglutide and placebo groups with regard to the risk of MACE. Similarly, a meta-analysis of cardiovascular events occurring during treatment with albiglutide, placebo, or active comparators in the HARMONY clinical trial programCitation75 determined there was no significant difference between albiglutide and comparator for the risk of MACE or hospital admission for unstable angina.
The effect of exenatide BID on cardiovascular outcomes has also been assessed in a pooled meta-analysis of cardiovascular safety data from 12 long-term, randomized, placebo- or insulin comparator-controlled trials.Citation76 The pooled relative risk for primary MACE for users of exenatide BID versus the comparator favored exenatide, suggesting no increased cardiovascular risk associated with exenatide use versus insulin or placebo. Two additional meta-analyses did not find any increase in the incidence of MACE in users of GLP-1RAs compared with comparators.Citation77,Citation78 Both studies showed that the GLP-1RAs were associated with a significantly lower risk of MACE compared with placebo, but not with active comparators (with the exception of pioglitazone).Citation77,Citation78 Consistent with these two studies, a sequential analysis of long-term trials comparing the effect of GLP-1RAs with other glucose-lowering drugs or placeboCitation79 as well as a large comparative safety study of GLP-1RAs versus other glucose-lowering agentsCitation80 both demonstrated no significant difference in the risk of cardiovascular events between drug classes.
Real-world data can also provide some insight into the impact of the GLP-1RAs on cardiovascular risk in patients with T2D. Retrospective studies have suggested that exena-tide BID, with or without concomitant insulin, significantly reduced the risk of cardiovascular events compared with insulinCitation81 and that exenatide BID is associated with a lower risk of cardiovascular events and hospitalizations versus other glucose-lowering therapies.Citation82
The present review included prospective and retrospective studies that compared the efficacy and safety of approved GLP-1RAs with other currently available glucose-lowering therapies in the treatment of patients with T2D, with data showing clinical benefit of GLP-1RAs over other glucose-lowering therapies. However, it is critical to consider the patient selection criteria, background medications, properties of the individual GLP-1RAs, drug exposure across dose intervals, and outcome definitions of each study. Furthermore, the patient characteristics in these studies need to be taken into account to assess the generalizability of the results to the T2D population at large; overly strict criteria limit the applicability of the data to everyday clinical practice.
Conclusion
This systematic analysis shows that GLP-1RAs were generally as effective as, or more effective than, oral glucose-lowering therapies in improving glycemic parameters such as HbA1c and FG in patients with T2D. The reduction in HbA1c with GLP-1RAs tended to be similar or smaller compared with the reductions achieved with insulin therapy, with less hypoglycemia. GLP-1RAs were consistently more effective at reducing weight than oral glucose-lowering therapies and insulin. Additionally, the GLP-1RAs appeared to have favorable effects on cardiovascular risk factors such as BP and lipid levels. In summary, GLP-1RAs are an effective, safe, and well-tolerated treatment option for T2D, with minimal risk of hypoglycemia and providing modest weight loss.
Author contributions
All authors have contributed sufficiently to the project to be included as authors. The authors contributed equally to the conception and design of the systematic review and literature search, interpretation of the results, and drafting of the manuscript.
Acknowledgments
Sheridan Henness, PhD, of inScience Communications, Springer Healthcare, provided medical writing support funded by AstraZeneca. The development of this manuscript was supported by AstraZeneca.
Disclosure
PA Levin has received grants from AstraZeneca, Boehringer Ingelheim, Eli Lilly, Merck, Novo Nordisk, and Sanofi. He is a consultant for AstraZeneca, Novo Nordisk, and Sanofi, and a speaker for AstraZeneca, Boehringer Ingelheim, Eli Lily, GlaxoSmithKline, Janssen, Novo Nordisk, and Sanofi. H Nguyen was an employee of AstraZeneca at the time this review was conceived and drafted. ET Wittbrodt is an employee of AstraZeneca. SC Kim has received research grants to the Brigham and Women’s Hospital from Astra-Zeneca, Bristol-Myers Squibb, Genentech, Lilly, and Pfizer. The authors report no other conflicts of interest in this work.
References
- American Diabetes Association Standards of medical care in diabetes-2015 Diabetes Care 2015 38 Suppl 1 S1 S93
- International Diabetes Federation IDF Diabetes Atlas 7th Ed 2015 Available from: http://www.diabetesatlas.org/resources/2015-atlas.html Accessed January 31, 2017
- Mathers CD Loncar D Projections of global mortality and burden of disease from 2002 to 2030 PLoS Med 2006 3 11 e442 17132052
- Inzucchi SE Bergenstal RM Buse JB Management of hyperglycemia in type 2 diabetes, 2015: a patient-centered approach: update to a position statement of the American Diabetes Association and the European Association for the Study of Diabetes Diabetes Care 2015 38 1 140 149 25538310
- Marso SP Daniels GH Brown-Frandsen K LEADER Steering Committee; LEADER Trial Investigators Liraglutide and cardiovascular outcomes in type 2 diabetes N Engl J Med 2016 375 4 311 322 27295427
- Pfeffer MA Claggett B Diaz R ELIXA Investigators Lixisenatide in patients with type 2 diabetes and acute coronary syndrome N Engl J Med 2015 373 23 2247 2257 26630143
- Berg JK Shenouda SK Heilmann CR Gray AL Holcombe JH Effects of exenatide twice daily versus sitagliptin on 24-h glucose, glucoregulatory and hormonal measures: a randomized, double-blind, crossover study Diabetes Obes Metab 2011 13 11 982 989 21615670
- Bergenstal RM Wysham C Macconell L DURATION-2 Study Group Efficacy and safety of exenatide once weekly versus sitagliptin or pioglitazone as an adjunct to metformin for treatment of type 2 diabetes (DURATION-2): a randomised trial Lancet 2010 376 9739 431 439 20580422
- Russell-Jones D Cuddihy RM Hanefeld M DURATION-4 Study Group Efficacy and safety of exenatide once weekly versus metformin, pioglitazone, and sitagliptin used as monotherapy in drug-naive patients with type 2 diabetes (DURATION-4): a 26-week double-blind study Diabetes Care 2012 35 2 252 258 22210563
- Nauck M Weinstock RS Umpierrez GE Guerci B Skrivanek Z Milicevic Z Efficacy and safety of dulaglutide versus sitagliptin after 52 weeks in type 2 diabetes in a randomized controlled trial (AWARD-5) Diabetes Care 2014 37 8 2149 2158 24742660
- Weinstock RS Guerci B Umpierrez G Nauck MA Skrivanek Z Milicevic Z Safety and efficacy of once-weekly dulaglutide versus sitagliptin after 2 years in metformin-treated patients with type 2 diabetes (AWARD-5): a randomized, phase III study Diabetes Obes Metab 2015 17 9 849 858 25912221
- Charbonnel B Steinberg H Eymard E Efficacy and safety over 26 weeks of an oral treatment strategy including sitagliptin compared with an injectable treatment strategy with liraglutide in patients with type 2 diabetes mellitus inadequately controlled on metformin: a randomised clinical trial Diabetologia 2013 56 7 1503 1511 23604551
- Pratley R Nauck M Bailey T 1860-LIRA-DPP-4 Study Group One year of liraglutide treatment offers sustained and more effective glycaemic control and weight reduction compared with sitagliptin, both in combination with metformin, in patients with type 2 diabetes: a randomised, parallel-group, open-label trial Int J Clin Pract 2011 65 4 397 407 21355967
- Pratley RE Nauck M Bailey T 1860-LIRA-DPP-4 Study Group Liraglutide versus sitagliptin for patients with type 2 diabetes who did not have adequate glycaemic control with metformin: a 26-week, randomised, parallel-group, open-label trial Lancet 2010 375 9724 1447 1456 20417856
- Pratley RE Nauck MA Bailey T 1860-LIRA-DPP-4 Study Group Efficacy and safety of switching from the DPP-4 inhibitor sitagliptin to the human GLP-1 analog liraglutide after 52 weeks in metformin-treated patients with type 2 diabetes: a randomized, open-label trial Diabetes Care 2012 35 10 1986 1993 22851600
- Takeshita Y Takamura T Kita Y Establishment of Rationale for Antiaging Diabetic Medicine (ERA-DM) Study Chapter 2 Group Vildagliptin vs liraglutide as a second-line therapy switched from sitagliptin-based regimens in patients with type 2 diabetes: a randomized, parallel-group study J Diabetes Investig 2015 6 2 192 200
- Van Gaal L Souhami E Zhou T Aronson R Efficacy and safety of the glucagon-like peptide-1 receptor agonist lixisenatide versus the dipeptidyl peptidase-4 inhibitor sitagliptin in young (<50 years) obese patients with type 2 diabetes mellitus J Clin Transl Endocrinol 2014 1 2 31 37
- Horton ES Silberman C Davis KL Berria R Weight loss, glycemic control, and changes in cardiovascular biomarkers in patients with type 2 diabetes receiving incretin therapies or insulin in a large cohort database Diabetes Care 2010 33 8 1759 1765 20460445
- Montilla S Marchesini G Sammarco A AIFA Anti-diabetics Monitoring Group Drug utilization, safety, and effectiveness of exena-tide, sitagliptin, and vildagliptin for type 2 diabetes in the real world: data from the Italian AIFA Anti-diabetics Monitoring Registry Nutr Metab Cardiovasc Dis 2014 24 12 1346 1353 25300980
- Nyeland ME Ploug UJ Richards A Evaluation of the effectiveness of liraglutide and sitagliptin in type 2 diabetes: a retrospective study in UK primary care Int J Clin Pract 2015 69 3 281 291 25302822
- Umpierrez G Tofé Povedano S Pérez Manghi F Shurzinske L Pechtner V Efficacy and safety of dulaglutide monotherapy versus metformin in type 2 diabetes in a randomized controlled trial (AWARD-3) Diabetes Care 2014 37 8 2168 2176 24842985
- Derosa G Maffioli P Salvadeo SA Exenatide versus glibenclamide in patients with diabetes Diabetes Technol Ther 2010 12 3 233 240 20151774
- Derosa G Putignano P Bossi AC Exenatide or glimepiride added to metformin on metabolic control and on insulin resistance in type 2 diabetic patients Eur J Pharmacol 2011 666 1–3 251 256 21645507
- Gallwitz B Guzman J Dotta F Exenatide twice daily versus glimepiride for prevention of glycaemic deterioration in patients with type 2 diabetes with metformin failure (EUREXA): an open-label, randomised controlled trial Lancet 2012 379 9833 2270 2278 22683137
- Garber A Henry R Ratner R LEAD-3 (Mono) Study Group Liraglutide versus glimepiride monotherapy for type 2 diabetes (LEAD-3 Mono): a randomised, 52-week, phase III, double-blind, parallel-treatment trial Lancet 2009 373 9662 473 481 18819705
- Garber A Henry RR Ratner R Hale P Chang CT Bode B LEAD-3 (Mono) Study Group Liraglutide, a once-daily human glucagon-like peptide 1 analogue, provides sustained improvements in glycae-mic control and weight for 2 years as monotherapy compared with glimepiride in patients with type 2 diabetes Diabetes Obes Metab 2011 13 4 348 356 21205128
- Kaku K Rasmussen MF Nishida T Seino Y Fifty-two-week, randomized, multicenter trial to compare the safety and efficacy of the novel glucagon-like peptide-1 analog liraglutide vs glibenclamide in patients with type 2 diabetes J Diabetes Investig 2011 2 6 441 447
- Nauck M Frid A Hermansen K Long-term efficacy and safety comparison of liraglutide, glimepiride and placebo, all in combination with metformin in type 2 diabetes: 2-year results from the LEAD-2 study Diabetes Obes Metab 2013 15 3 204 212 22985213
- Nauck M Marre M Adding liraglutide to oral antidiabetic drug mono-therapy: efficacy and weight benefits Postgrad Med 2009 121 3 5 15
- Seino Y Rasmussen MF Nishida T Kaku K Efficacy and safety of the once-daily human GLP-1 analogue, liraglutide, vs glibenclamide monotherapy in Japanese patients with type 2 diabetes Curr Med Res Opin 2010 26 5 1013 1022 20199137
- Chiefari E Capula C Vero A Add-on treatment with liraglutide improves glycemic control in patients with type 2 diabetes on metformin therapy Diabetes Technol Ther 2015 17 7 468 474 25844858
- Nauck M Frid A Hermansen K LEAD-2 Study Group Efficacy and safety comparison of liraglutide, glimepiride, and placebo, all in combination with metformin, in type 2 diabetes: the LEAD (liraglu-tide effect and action in diabetes)-2 study Diabetes Care 2009 32 1 84 90 18931095
- Marre M Shaw J Brändle M LEAD-1 SU study group Liraglutide, a once-daily human GLP-1 analogue, added to a sulphonylurea over 26 weeks produces greater improvements in glycaemic and weight control compared with adding rosiglitazone or placebo in subjects with type 2 diabetes (LEAD-1 SU) Diabet Med 2009 26 3 268 278 19317822
- DeFronzo RA Triplitt C Qu Y Lewis MS Maggs D Glass LC Effects of exenatide plus rosiglitazone on beta-cell function and insulin sensitivity in subjects with type 2 diabetes on metformin Diabetes Care 2010 33 5 951 957 20107105
- Xu W Bi Y Sun Z Comparison of the effects on glycaemic control and β-cell function in newly diagnosed type 2 diabetes patients of treatment with exenatide, insulin or pioglitazone: a multicentre randomized parallel-group trial (the CONFIDENCE study) J Intern Med 2015 277 1 137 150 25039675
- Araki E Inagaki N Tanizawa Y Oura T Takeuchi M Imaoka T Efficacy and safety of once-weekly dulaglutide in combination with sulpho-nylurea and/or biguanide compared with once-daily insulin glargine in Japanese patients with type 2 diabetes: a randomized, open-label, phase III, non-inferiority study Diabetes Obes Metab 2015 17 10 994 1002 26179754
- Barnett AH Burger J Johns D Tolerability and efficacy of exena-tide and titrated insulin glargine in adult patients with type 2 diabetes previously uncontrolled with metformin or a sulfonylurea: a multinational, randomized, open-label, two-period, crossover noninferiority trial Clin Ther 2007 29 11 2333 2348 18158075
- Bergenstal R Lewin A Bailey T Chang D Gylvin T Roberts V Novo-Log Mix-vs.-Exenatide Study Group Efficacy and safety of biphasic insulin aspart 70/30 versus exenatide in subjects with type 2 diabetes failing to achieve glycemic control with metformin and a sulfonylurea Curr Med Res Opin 2009 25 1 65 75 19210140
- Blonde L Jendle J Gross J Once-weekly dulaglutide versus bedtime insulin glargine, both in combination with prandial insulin lispro, in patients with type 2 diabetes (AWARD-4): a randomised, open-label, phase 3, non-inferiority study Lancet 2015 385 9982 2057 2066 26009229
- D’Alessio D Häring HU Charbonnel B EAGLE Investigators Comparison of insulin glargine and liraglutide added to oral agents in patients with poorly controlled type 2 diabetes Diabetes Obes Metab 2015 17 2 170 178 25359159
- Davies M Heller S Sreenan S Once-weekly exenatide versus once- or twice-daily insulin detemir: randomized, open-label, clinical trial of efficacy and safety in patients with type 2 diabetes treated with metformin alone or in combination with sulfonylureas Diabetes Care 2013 36 5 1368 1376 23275363
- Davies MJ Donnelly R Barnett AH Jones S Nicolay C Kilcoyne A Exenatide compared with long-acting insulin to achieve glycaemic control with minimal weight gain in patients with type 2 diabetes: results of the Helping Evaluate Exenatide in patients with diabetes compared with Long-Acting insulin (HEELA) study Diabetes Obes Metab 2009 11 12 1153 1162 19930005
- Diamant M Nauck MA Shaginian R 4B Study Group Glucagon-like peptide 1 receptor agonist or bolus insulin with optimized basal insulin in type 2 diabetes Diabetes Care 2014 37 10 2763 2773 25011946
- Diamant M Van Gaal L Guerci B Exenatide once weekly versus insulin glargine for type 2 diabetes (DURATION-3): 3-year results of an open-label randomised trial Lancet Diabetes Endocrinol 2014 2 6 464 473 24731672
- Diamant M Van Gaal L Stranks S Safety and efficacy of once-weekly exenatide compared with insulin glargine titrated to target in patients with type 2 diabetes over 84 weeks Diabetes Care 2012 35 4 683 689 22357185
- Diamant M Van Gaal L Stranks S Once weekly exenatide compared with insulin glargine titrated to target in patients with type 2 diabetes (DURATION-3): an open-label randomised trial Lancet 2010 375 9733 2234 2243 20609969
- Gallwitz B Böhmer M Segiet T Exenatide twice daily versus premixed insulin aspart 70/30 in metformin-treated patients with type 2 diabetes: a randomized 26-week study on glycemic control and hypoglycemia Diabetes Care 2011 34 3 604 606 21285388
- Giorgino F Benroubi M Sun JH Zimmermann AG Pechtner V Efficacy and safety of once-weekly dulaglutide versus insulin glargine in patients with type 2 diabetes on metformin and glimepiride (AWARD-2) Diabetes Care 2015 38 12 2241 2249 26089386
- Gough SC Bode BW Woo VC One-year efficacy and safety of a fixed combination of insulin degludec and liraglutide in patients with type 2 diabetes: results of a 26-week extension to a 26-week main trial Diabetes Obes Metab 2015 17 10 965 973 25980900
- Gough SCL Bode B Woo V NN9068-3697 (DUAL-I) trial investigators Efficacy and safety of a fixed-ratio combination of insulin degludec and liraglutide (IDegLira) compared with its components given alone: results of a phase 3, open-label, randomised, 26-week, treat-to-target trial in insulin-naive patients with type 2 diabetes Lancet Diabetes Endocrinol 2014 2 11 885 893 25190523
- Heine RJ Van Gaal LF Johns D Mihm MJ Widel MH Brodows RG GWAA Study Group Exenatide versus insulin glargine in patients with suboptimally controlled type 2 diabetes: a randomized trial Ann Intern Med 2005 143 8 559 569 16230722
- Inagaki N Atsumi Y Oura T Saito H Imaoka T Efficacy and safety profile of exenatide once weekly compared with insulin once daily in Japanese patients with type 2 diabetes treated with oral anti-diabetes drug(s): results from a 26-week, randomized, open-label, parallel-group, multicenter, noninferiority study Clin Ther 2012 34 9 1892 1908 22884767
- Mathieu C Ostenson CG Matthaei S Using exenatide twice daily or insulin in clinical practice: results from CHOICE Diabetes Ther 2013 4 2 285 308 24018835
- Mathieu C Rodbard HW Cariou B BEGIN: VICTOZA ADD-ON (NN1250-3948) study group A comparison of adding liraglutide versus a single daily dose of insulin aspart to insulin degludec in subjects with type 2 diabetes (BEGIN: VICTOZA ADD-ON) Diabetes Obes Metab 2014 16 7 636 644 24443830
- Nauck MA Duran S Kim D A comparison of twice-daily exenatide and biphasic insulin aspart in patients with type 2 diabetes who were suboptimally controlled with sulfonylurea and metformin: a non-inferiority study Diabetologia 2007 50 2 259 267 17160407
- Ostenson CG Matthaei S Reaney M Treatment outcomes after initiation of exenatide twice daily or insulin in clinical practice: 12-month results from CHOICE in six European countries Diabetes Metab Syndr Obes 2013 6 171 185 23667315
- Russell-Jones D Vaag A Schmitz O Liraglutide Effect and Action in Diabetes 5 (LEAD-5) met+SU Study Group Liraglutide vs insulin glargine and placebo in combination with metformin and sulfonylurea therapy in type 2 diabetes mellitus (LEAD-5 met+SU): a randomised controlled trial Diabetologia 2009 52 10 2046 2055 19688338
- Baser O Wei W Baser E Xie L Clinical and economic outcomes in patients with type 2 diabetes initiating insulin glargine disposable pen versus exenatide BID J Med Econ 2011 14 6 673 680 21892858
- Bounthavong M Tran JN Golshan S Retrospective cohort study evaluating exenatide twice daily and long-acting insulin analogs in a Veterans Health Administration population with type 2 diabetes Diabetes Metab 2014 40 4 284 291 25059703
- Dalal MR Xie L Baser O DiGenio A Adding rapid-acting insulin or GLP-1 receptor agonist to basal insulin: outcomes in a community setting Endocr Pract 2015 21 1 68 76 25148821
- Pawaskar M Li Q Hoogwerf BJ Metabolic outcomes of matched patient populations initiating exenatide BID vs. insulin glargine in an ambulatory care setting Diabetes Obes Metab 2012 14 7 626 633 22321776
- Pawaskar M Li Q Reynolds MW Metabolic outcomes of elderly patient populations initiating exenatide BID versus insulin glargine in an ambulatory care setting Curr Med Res Opin 2012 28 6 991 997 22519390
- Sudhakaran C Fathima M Anjana RM Unnikrishnan RI Mohan V Effectiveness of exenatide in Asian Indians in a clinical care setting Diabetes Technol Ther 2010 12 8 613 618 20615102
- Reid T Choosing GLP-1 receptor agonists or DPP-4 inhibitors: weighing the clinical trial evidence Clin Diabetes 2012 30 1 3 12
- Kim W Egan JM The role of incretins in glucose homeostasis and diabetes treatment Pharmacol Rev 2008 60 4 470 512 19074620
- Smilowitz NR Donnino R Schwartzbard A Glucagon-like peptide-1 receptor agonists for diabetes mellitus: a role in cardiovascular disease Circulation 2014 129 22 2305 2312 24891623
- Lang K Nguyen H Huang H Kaufman E Levin P Real-world treatment responses from electronic medical record (EMR) data among patients with type 2 diabetes (T2D) receiving basal insulin either with mealtime insulin or exenatide BID, by A1c attainment level and baseline A1c [abstract 1540-P] Diabetes 2016 65 Suppl 1 A360 A431
- Lorenz M Lawson F Owens D Differential effects of glucagon-like peptide-1 receptor agonists on heart rate Cardiovasc Diabetol 2017 16 1 6 28086882
- Marso SP Bain SC Consoli A SUSTAIN-6 Investigators Sema-glutide and cardiovascular outcomes in patients with type 2 diabetes N Engl J Med 2016 375 19 1834 1844 27633186
- Bentley-Lewis R Aguilar D Riddle MC ELIXA Investigators Rationale, design, and baseline characteristics in Evaluation of LIX-isenatide in Acute Coronary Syndrome, a long-term cardiovascular end point trial of lixisenatide versus placebo Am Heart J 2015 169 5 631 638 25965710
- Holman RR Bethel MA George J Rationale and design of the EXenatide Study of Cardiovascular Event Lowering (EXSCEL) trial Am Heart J 2016 174 103 110 26995376
- Eli Lilly and Company The effect of dulaglutide on major cardiovascular events in patients with type 2 diabetes: Researching Cardiovascular Events with a Weekly INcretin in Diabetes (REWIND) ClinicalTrials.gov record: NCT01394952. Available from: https://clinicaltrials.gov/ct2/show/NCT01394952 Accessed October 11, 2016
- GlaxoSmithKline A long term, randomised, double blind, placebo-controlled study to determine the effect of albiglutide, when added to standard blood glucose lowering therapies, on major cardiovascular events in patients with type 2 diabetes mellitus (HARMONY Outcomes) ClinicalTrials.gov identifier: NCT02465515. Available from: https://clinicaltrials.gov/ct2/show/NCT02465515 Accessed October 11, 2016
- Ferdinand KC Botros FT Atisso CM Sager PT Cardiovascular safety for once-weekly dulaglutide in type 2 diabetes: a pre-specified meta-analysis of prospectively adjudicated cardiovascular events Cardiovasc Diabetol 2016 15 38 26912057
- Fisher M Petrie MC Ambery PD Donaldson J Ye J McMurray JJ Cardiovascular safety of albiglutide in the Harmony programme: a meta-analysis Lancet Diabetes Endocrinol 2015 3 9 697 703 26276240
- Ratner R Han J Nicewarner D Yushmanova I Hoogwerf BJ Shen L Cardiovascular safety of exenatide BID: an integrated analysis from controlled clinical trials in participants with type 2 diabetes Cardiovasc Diabetol 2011 10 22 21410975
- Monami M Cremasco F Lamanna C Glucagon-like peptide-1 receptor agonists and cardiovascular events: a meta-analysis of randomized clinical trials Exp Diabetes Res 2011 2011 215764 21584276
- Monami M Dicembrini I Nardini C Fiordelli I Mannucci E Effects of glucagon-like peptide-1 receptor agonists on cardiovascular risk: a meta-analysis of randomized clinical trials Diabetes Obes Metab 2014 16 1 38 47 23829656
- Wu S Sun F Zhang Y The cardiovascular effects of glucagon-like peptide-1 receptor agonists: a trial sequential analysis of randomized controlled trials J Clin Pharm Ther 2014 39 1 7 13 24127768
- Patorno E Everett BM Goldfine AB Comparative cardiovascular safety of glucagon-like peptide-1 receptor agonists versus other anti-diabetic drugs in routine care: a cohort study Diabetes Obes Metab 2016 18 8 755 765 27003762
- Paul SK Klein K Maggs D Best JH The association of the treatment with glucagon-like peptide-1 receptor agonist exenatide or insulin with cardiovascular outcomes in patients with type 2 diabetes: a retrospective observational study Cardiovasc Diabetol 2015 14 10 25616979
- Best JH Hoogwerf BJ Herman WH Risk of cardiovascular disease events in patients with type 2 diabetes prescribed the glucagon-like peptide 1 (GLP-1) receptor agonist exenatide twice daily or other glucose-lowering therapies: a retrospective analysis of the LifeLink database Diabetes Care 2011 34 1 90 95 20929995