Abstract
Leishmaniasis is a vector-borne zoonotic disease caused by protozoan parasites of the genus Leishmania, which are responsible for numerous clinical manifestations, such as cutaneous, visceral, and mucocutaneous leishmaniasis, depending on the site of infection for particular species. These complexities threaten 350 million people in 98 countries worldwide. Amastigotes living within macrophage phagolysosomes are the principal target of antileishmanial treatment, but these are not an easy target as drugs must overcome major structural barriers. Furthermore, limitations on current therapy are related to efficacy, toxicity, and cost, as well as the length of treatment, which can increase parasitic resistance. Nanotechnology has emerged as an attractive alternative as conventional drugs delivered by nanosized carriers have improved bioavailability and reduced toxicity, together with other characteristics that help to relieve the burden of this disease. The significance of using colloidal carriers loaded with active agents derives from the physiological uptake route of intravenous administered nanosystems (the phagocyte system). Nanosystems are thus able to promote a high drug concentration in intracellular mononuclear phagocyte system (MPS)-infected cells. Moreover, the versatility of nanometric drug delivery systems for the deliberate transport of a range of molecules plays a pivotal role in the design of therapeutic strategies against leishmaniasis. This review discusses studies on nanocarriers that have greatly contributed to improving the efficacy of antileishmaniasis drugs, presenting a critical review and some suggestions for improving drug delivery.
Introduction
Leishmaniasis is a parasitic disease caused by Leishmania spp., transmitted to humans and animals by species of Phlebotomine and Lutzomyia sandflies. The Leishmania parasite has two distinct morphological forms in its life cycle: promastigote and amastigote. The sandfly-transmitted flagellated metacyclic promastigotes rapidly enter into cells of the mononuclear phagocyte system (MPS) as part of the normal phagocytic response. The phagosome formed around the parasites undergoes a process of maturation and remodeling of the membrane and forms a new organelle, the parasitophorous vacuole (PV). Within the PV, the promastigotes differentiate into amastigotes, which multiply until eruption of the MPS cell (over a period of 4–6 days); the infection then spreads further. Having different tropism characteristics, parasites may infect either superficial cells or visceral cells. The parasite’s life cycle is completed when an uninfected sandfly takes a blood meal from the infected host. Because the infection involves several overlapping species and sandfly vectors, the taxonomy, ecology, epidemiology, and pathogenicity of the disease are complex.Citation1,Citation2
Leishmaniasis has numerous clinical manifestations, depending on which it is classified into three types: 1) cutaneous leishmaniasis (CL), the most common; 2) mucocutaneous leishmaniasis (ML), which may disseminate to the mucosa; and 3) visceral leishmaniasis (VL), also known as kala-azar, the most serious form of the disease, which can be fatal if not properly treated and could be disseminated to several organs.Citation3
Leishmania infantum is responsible for VL infections in Latin America and North Africa, while Leishmania donovani is responsible for VL infections in areas of the Indian subcontinent and of East Africa. Although >90% of VL infections are concentrated in India, Brazil, Bangladesh, Nepal, and Sudan,Citation4 a study shows a rapid increase in VL infections worldwide and climate change is expected to cause VL to severely impact Europe in the near future.Citation5
Treatment failure and relapse rates are particularly high in cases of impaired cellular immunity, especially in human immunodeficiency virus (HIV) coinfection.Citation6 HIV and Leishmania infection mutually reinforce one another, and HIV patients are more likely to develop VL (due to reactivation of a dormant infection or clinical manifestation after primary infection).
Current chemotherapeutic treatments are successful, to some extent, and the major targets pursued by associations such as the Drugs for Neglected Diseases initiative (DNDi) and the World Health Organization (WHO) in conjunction with foundations and pharmaceutical companies, may chiefly be summarized as to develop an oral, safe, effective, low-cost, and short-course treatment for VL and to develop novel treatment regimens for patients coinfected with HIV and VL. Furthermore, as for all neglected tropical diseases, additional control mechanisms and tools are necessary, such as drugs, vaccines, reliable diagnostics, vector control agents, and control strategies, to eradicate infection.Citation7
Considering the small number of chemotherapeutic agents or innovative antileishmanial medicines that are available, in parallel with the search for more efficient and less toxic antileishmanial drugs, including the development of a successful vaccine, the push to design stable nanotechnology-based drug delivery systems (DDSs) is likely to be the main strategy in fighting the disease. The use of colloidal carriers loaded with active agents is a clearcut approach, thanks to the physiological uptake route of intravenous (iv) administered nanosystems (the MPS cells).Citation8 Nanosystems can thus lead to high drug concentrations in the intracellular MPS-infected cells and in the PV; colloidal carriers also protect the drug against in vivo degradation.
This review seeks to give an overview of the current therapeutic protocols and new approaches involved in the search for safer and more active drugs, as well as the strategies employed for choosing appropriate delivery systems to produce the next-generation agents for treating VL. Although the role of nanotechnology in DDSs for leishmaniasis has been reviewed elsewhere,Citation9,Citation10 this review also highlights the limits and issues involved in applying modern techniques and illustrates the most promising results. In particular, the different strategies, such as their potential, cost, feasibility, and limits, were critically evaluated and commented. As in vivo studies are imperative for estimating the clinical feasibility of a DDS, particular emphasis was given to approaches reporting preclinical/clinical data and/or to those showing research progress during recent years.
Although alternative immunotherapeutic strategies are potentially advantageous, these strategies and nanodevices that can be used as promising vaccine carriers were not discussed here.Citation9,Citation11
Current chemotherapy
The first therapeutic options were introduced in the early 1900s for mucocutaneous form, and other treatments have been added over the last decade; however, they are not devoid of limitations relating to efficacy, toxicity, cost, length of treatment, increasing parasitic resistance, and difficulty of administration, making treatment a complex issue. The principal drugs currently available are antimonials, amphotericin B (AmpB), paromomycin (PM), and miltefosine (MF). , taken from the 2010 WHO report, is presented to give a clear view of treatment regimens and their costs (report of a meeting of the WHO Expert Committee on the Control of Leishmaniases, Geneva, March 22–26, 2010). Several recent studies clearly describe the various combination approaches that have been used, in the search for the optimal and most cost-effective strategy, not only to treat patients but also to control infections in countries including Morocco, India, Sudan, and Nepal.Citation12–Citation16
Table 1 Price per visceral leishmaniasis treatment
Pentavalent antimonials have been used for the past several decades to treat VL. At present, two types of organoantimony (V) complexes are commercially available: Glucantime® (meglumine antimoniate [MA]) and Pentostam® (sodium stibogluconate [SSG]). These are the standard first-line medicines in most parts of the world (>90% overall cure rate). Initial treatment of VL should be based on a daily injection of 20 mg/kg body weight of Sb(V), and injections are usually given for 28–30 days. The cost for the course of treatment is <60 US dollars ().
The mechanism of action of these compounds, in one model, is based on the reductive bioconversion of Sb(V) to Sb(III), by the parasite or by the infected host cells, to create an active agent Sb(III)–trypanothione conjugate.Citation17 In a second model, Sb(V) is the active species, which directly exerts activity against Leishmania. Sb(V) is able to act selectively against the topoisomerase of the promastigote rather than against that of the monocyte.Citation18 Sb(V) is also reported to bind ribonucleosides in an environment similar to that of lysosomes.Citation19
Unfortunately for first-line antimonials, the development of resistance is a primary barrier to successful treatment. The resistance mechanism has been found to be multifactorial and is principally due to reduced Sb(III) uptake or/and increased cell efflux/sequestration through an abnormally high level of trypanothione and increased expression of the metalloid-thiol pump.Citation20 Resistance to therapeutics has also been imputed to different efflux pumps or ATP-binding cassette transporters, such as P-glycoprotein and multidrug resistance protein.Citation21
AmpB is a natural antibiotic with a macrolide polyene structure and significant antifungal and antiparasitic activities. Currently, AmpB is widely used to treat systemic Candida albicans and Aspergillus fumigatus infections.Citation22 The mechanism of action of AmpB is based on its binding to the ergosterol in the fungus or parasite rather than to human cholesterol. In general, AmpB binds to ergosterol through a hydrophobic interaction, in a series of events that eventually lead to pore formation in the parasite membrane.Citation23 In clinical trials in India, AmpB formulated with deoxycholate (Fungizone®) was given daily or on alternate days by iv slow infusion at a dose of 0.75–1.0 mg/kg/day for 15–20 doses and was 99% effective.Citation24
To reduce the severe side effects, which include nephrotoxicity and hematotoxicity, different formulations have been developed and are currently available on the market under the trade names Abelcet®, Amphocil®, Amphotec® (lipid complexes), AmBisome® (liposomes), and Ampholip®.
Used in India and Europe, all of these formulations were found to be effective; the liposomal formulation, in which the drug is intercalated in a lipid membrane consisting of hydrogenated soy phosphatidylcholine (PC), cholesterol, and distearoylphosphatidylglycerol, is the most widely used against VL. Given at a dose of 3–4 mg/kg/day, for a total dose of 15–24 mg/kg, administered to various regimens, AmpB liposomal formulation had 90%–98% efficacy in southern Europe.Citation25 Shorter treatments are also effective; in India, a single-course therapy of 10 mg/kg was found to give a 98% cure rate.Citation26 The principal benefit of AmBisome is its reduced toxicity to mammalian cells, allowing higher doses to be administered, rather than any increase in its antileishmanial activity per se. The major drawback of AmBisome is its high cost (126–250 US dollars) vs Fungizone (30 US dollars for a 30-day course of treatment), putting it out of patients’ reach in some countries; it is unclear whether this limitation is applicable to other geographical areas. Nevertheless, WHO and Gilead Sciences have signed agreements to donate hundreds of vials of liposomal AmpB, extending their previous agreement until 2021.Citation195 The new 5-year collaboration, whose estimated value is 20 million US dollars, includes funding that will allow populations affected by VL to benefit from enhanced access to diagnosis and treatment.
In a study investigating the extent to which the side effects of AmpB were reduced by the use of a lipid emulsion, a preformed AmpB lipid emulsion (ABLE) was compared with Fungizone and AmBisome in terms of safety and efficacy.Citation27,Citation28 Indian clinical trials showed that ABLE was a well-tolerated, efficacious, and affordable lipid formulation. In a recent study on a population of 500 patients, the efficacy of single day infusions of ABLE at 15 mg/kg/day was satisfactory, with an initial cure rate of 95.9%, compared to 100% for AmBi-some. The proportion of patients with no clinical signs and symptoms of relapse of kala-azar during 6 months follow-up was 85.9% in the ABLE group compared to 98.4% in the AmBisome group.Citation28
Resistance to AmpB has been induced in vitro, and a study has shown it to be related to the increased cell membrane fluidity occurring when ergosterol is modified to its precursor cholesta-5,7,24-trien-3β-ol.Citation29 The modification reduced AmpB’s attachment to sterol-modified membranes. In clinical isolates of L. donovani, a multifactorial response was observed, in which altered membrane composition, ATP-binding cassette transporters, and an upregulated thiol metabolic pathway played roles in conferring AmpB resistance.Citation30
PM (aminosidine) is an aminoglycoside antibiotic, used to treat intestinal infections such as amebiasis and cryptosporidiosis, which was rediscovered as an antileishmanial agent. PM has been shown to be effective in Indian VL: a dose of 15 mg/kg for 21 days administered iv gave a cure rate of 93%–95%. The mechanism of action of PM in Leishmania is not precisely known, but protein synthesis machinery has been proposed as the target. Although there are no known clinical isolates of PM-resistant strains, a resistant L. donovani strain was readily generated experimentally: ribosomal proteins were found to be upregulated.Citation31 PM must be administered by the parenteral route; however, because it undergoes rapid clearance after iv administration, being excreted in the urine upon glomerular filtration, treatment requires multiple dosages. Adverse effects such as nephrotoxicity, ototoxicity, and hepatoxicity have been reported.Citation32
MF (hexadecylphosphocholine) was the first, and still the only, orally available drug for the treatment of VL.Citation33 MF belongs to a class of alkylphosphocholine drugs initially developed in the 1980s to treat solid tumors. Treatment with 150 mg/day per >50 kg body weight for 28 days in immunocompetents gave a cure rate of 94% in India and ~90% in Ethiopia.Citation34 Its antileishmanial activity involves action on several targets; however, no mechanism has been identified definitively: alkyl phospholipid metabolism, glycosylphosphatidylinositol anchor biosynthesis, signal transduction, mitochondrial dysfunction, and immunomodulatory effects might be involved.Citation35 Whatever its indication of use, pharmacovigilance for significant safety issues will remain a priority. Furthermore, MF should not be administered in pregnancy due to its teratogenicity.Citation33
In India, post-kala-azar dermal leishmaniasis (PKDL) is a sequel to VL occurring within a year or up to 32 years after VL has been cured; a study suggests that oral MF for 2–3 months could be considered as a treatment of choice for Indian PKDL.Citation36 MF has a median half-life of ~152 h, which might encourage the development of clinical resistance. Furthermore, being an oral agent, its improper use in endemic countries would increase the risk of resistance and would spread resistant parasites where the prevalence of infection is high.Citation37
The risk of emergence of drug resistance in the field is exacerbated by the relatively easy production of in vitro resistant Leishmania clones, combined with the occurrence of relapses in immunocompetent patients, the presence of HIV/VL coinfections, and high levels of anthroponotic transmission in both Africa and India.Citation38 Combination treatment has the potential advantages not only of shortening the treatment duration (thus increasing compliance) and reducing the overall drug dose (thereby reducing toxic effects and cost) but also of reducing the probability that drug-resistant parasites are selected, and thus prolonging the effective life of available medicines. Several trials of combinations have shown favorable results.Citation39,Citation40 The combination of PM and antimonials produced a higher cure rate in VL patients in a trial in Bihar, than did antimonials alone, in which the lack of response was common.Citation41
Many other compounds are considered to be the second-line drugs for leishmaniasis, including pentamidine and the antifungal azole fluconazole, as well as fexinidazole and oleyl phosphocholine (OlPC), with approval status at different stages. The diguanidine-based compound pentamidine was initially developed as a synthetic analog of insulin. Pentamidine has shown high activity against the acute phase of human African trypanosomiasis;Citation42 it has attracted renewed interest for its use as secondary prophylaxis in HIV-coinfected VL patients.Citation43 The drug is given intramuscularly or, preferably, by iv infusion. However, pentamidine is now no longer recommended as a treatment option for VL. High doses are needed to achieve a cure, and irreversible diabetes has been reported in 4%–12% of cases.Citation44
The primary action of pentamidine against Leishmania is not clearly understood, but the drug’s structural characteristics and experimental results suggest that the active transport system and the mitochondria are the final sites of inhibition. A series of experiments showed that it acts as an inhibitor of polyamine uptake and the arginine transporter,Citation45 while other findings indicate the mitochondrial replication system as possible target (through inhibition of topoisomerase II activity).Citation46 Resistance to pentamidine in Leishmania has been associated with changes in the concentrations of polyamines and arginine within the cell.Citation47
In collaboration with Sanofi, DNDi recently developed fexinidazole, a nitroimidazole derivative DNA synthesis inhibitor, for the oral treatment of human African trypanosomiasis. Fexinidazole has shown potent activity against L. donovani in vitro and in vivo in a VL mouse model.Citation48 A Phase II proof of concept study evaluating the efficacy of fexinidazole for the treatment of primary VL was initiated in November 2013 in Sudan (FEXI VL 001; NCT01980199); this open-label trial was designed to enroll 66 patients. DNDi terminated this trial in September 2015 due to lack of efficacy. However, the combination of fexinidazole and MF is still in trial.Citation196 Other members of the nitroimidazole class of molecules have emerged, and novel products are currently in advanced preclinical evaluations.Citation49,Citation50
Nanostructured anti-Leishmania delivery systems
The use of DDSs stands as a complementary strategy for developing new treatments and combination therapies for VL.Citation9 An appropriate DDS can be used in antileishmaniasis therapy to produce the necessary high drug concentration in the intracellular phagolysosome or PV, where the Leishmania is hosted. The major challenge in using a small drug is the difficulty of enabling it to penetrate inside the macrophages: multiple membranes must be traversed by the therapeutic agent to reach parasitic target. The nonspecificity to macrophages of current treatments leads to their accumulation in normal, healthy tissues, with prevalent toxicity as side effects. Improving the safety/efficacy ratio by delivering a high drug concentration at a controlled rate, using an appropriate targeting strategy, is an attractive goal that has been vigorously pursued.
DDS may also overcome problems relating to the low water solubility of drugs and could protect the active molecule from degradation in biological fluids. Furthermore, alternative routes to parenteral drug delivery (eg, nasal, pulmonary, and topical) that can improve patient compliance are achievable with appropriate DDS. In particular, developing oral formulations of current chemotherapy options is the most attractive prospect, although the lack of treatment supervision may lead to patients failing to complete the full course of treatment. This is a critical issue, as it may facilitate the development of drug resistance in Leishmania. Finally, novel materials that are able to become “smart”, capable of responding to the physiological environment, can also considerably improve drug availability.Citation51
The combination of antileishmanial drugs with nanocarriers is emerging as a promising approach, because the traditional nanocarriers (liposomes and polymer nanoparticles) are readily internalized by macrophages in the liver and spleen, releasing the drug inside the cell and thus leading to a high local concentration, and ultimately killing the protozoa. In this connection, the main strategy in treating Leishmania is to target the drugs directly to macrophages, using appropriate nanosized delivery systems.Citation52 A targeted approach entailing modification of the nanocarrier surface will also increase parasite selectivity.
Another advantage is the possibility of packing multiple payloads in a single carrier, providing a combination therapy that may have a synergist effect (overcoming resistance). However, the sequencing and scale-up of approaches of this type are still very challenging.
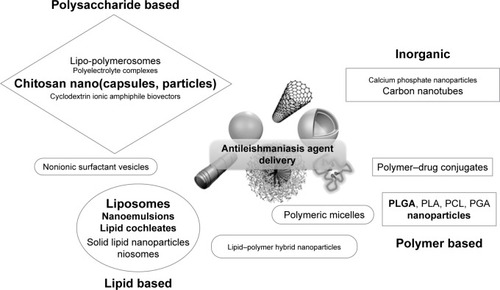
The following sections review the recently reported research concerning anti-VL nanotechnology, with special emphasis on the use of liposomes, polymers, polysaccharides, metal nanoparticles, and carbon-based materials loaded with chemotherapeutics currently used in VL treatment (a schematic presentation of different delivery systems is given in ). Since AmpB is currently the most potent antileishmanial agent, the majority of efforts in nanotechnology have been focused on this drug. For this reason, we pay attention and detail on innovative AmpB delivery systems. summarizes the principal characteristics and the results obtained.
Table 2 Principal characteristics of amphotericin delivery systems with the most advanced in vivo results available
Figure 1 Schematic representation of different classes of antileishmanial nanometric delivery systems.
Note: The character size (also bold type) evidences the number of publications related to each system.
Abbreviations: PCL, poly(caprolactone); PGA, poly(glycolic acid); PLA, poly(lactic acid); PLGA, poly(lactide-co-glycolide).
In particular, one of the most challenging achievements is the oral administration of AmpB. This is limited because AmpB is absorbed by passive diffusion through the intestinal membrane. However, the process of passive diffusion is dependent on drug’s molecular weight and is efficient for drugs up to molecular weight 500 Da; efficiency then decreases at higher molecular weights. AmpB has a molecular weight of 924.08 Da, and thus, passive diffusion may be expected to be very low.Citation53 Moreover, AmpB is relatively unstable at the acidic pH of the gastrointestinal tract, and upon oral administration might be degraded prior to absorption. In addition, it undergoes extensive P-gp efflux from the enterocytes, which further limits its oral bioavailability. Taken together, these factors lead to very low oral bioavailability of AmpB (~0.3% of the administered dose).
Several nanosystems have also been developed to improve the bioavailability of AmpB (for an exhaustive review refer Torrado et alCitation54), and some of them, related to VL treatment, are reported in the following sections. These delivery systems may have several advantages, as they might 1) enhance AmpB dissolution and also protect it from the acidic gastric environment, 2) increase intestinal permeability using either delivery systems of an appropriate size to enhance lymphatic uptake or carriers able to cross the gastrointestinal barrier, and 3) prolong gastrointestinal transit time through the use of bioadhesive systems. The biodistribution of AmpB is dependent on the type of formulation, and there is a weak correlation between drug plasma and organ levels.Citation55 For this reason, AmpB plasma levels must be considered in combination with target tissue pharmacokinetics. An oral formulation would be of benefit to VL patients, but several issues remain to be resolved.
For completeness, “Novel products with anti-leishmanial activity combined into nanocarriers” section comprises DDS loaded with different products with antileishmaniasis activity.
Currently, all these experimental compounds, as well as the majority of nanostructured systems, have been tested in vitro and in animal (mice or hamsters) models. Thus, a clear comparison with currently available medicines has to be carefully pondered.
Liposomes
Liposomes are small artificial vesicles of spherical shape that can be created from cholesterol and phospholipids. Due to their biocompatibility, stability, ease of scaling up, and ability to carry various molecules as cargo, liposomes are currently used as DDS in a range of different treatments. Most liposomal formulations available on the market target cancers. Information about this application can be find elsewhere.Citation56
The reticuloendothelial system (RES) is the main site of accumulation of liposomes following their systemic administration, and in the RES, liposomes are mainly cleared by resident macrophages. Primary organs associated with the RES include the liver, spleen, kidney, lungs, bone marrow, and lymph nodes.
The best clinical results in VL treatment, as reported earlier, have been achieved with the liposomal formulation AmBisome, which allows to load an high concentration of AmpB (drug loading [DL] =12.5%) (drug weight/[drug + liposomal components] weight) and represents the gold standard for novel delivery systems. Over recent years, lipid complex formulations of AmpB have been proposed,Citation57 showing interesting in vitro activity but less efficient than AmBisome in in vivo evaluations. More recently, liposomal formulations in which cholesterol was replaced by ergosterol, which constitutes 50% molarity of total lipids (Kalsome™ 10; Lifecare Innovations Ltd., Gurugram, India), have been evaluated.Citation58,Citation59 This change followed the in vitro observation that exogenous cholesterol, if added to the culture, enhanced the growth of Leishmania promastigotes; thus, the absence of cholesterol might make the drug more suitable for clearing parasites. Large multilamellar vesicles of ~1,000 nm in diameter were obtained and were kept separately by sonication. In vivo results confirmed Kalsome™ 10 efficacy in a murine VL model without major toxic effects. More recently, a possible cell death mechanism in Kalsome™ 10-treated L. donovani was proposed.Citation60 Interestingly, the component of the liposomal formulation induced apoptotic-like cell death in L. donovani parasites, demonstrating its antileishmanial function.
A number of older anti-Leishmania antimonials, formulated in liposomes, have been investigated, starting in 1977.Citation61 Liposomes with different surface charge (anionic and neutral) were subsequently tested,Citation62 but only recently, a robust effective treatment against infection with SSG-resistant Leishmania parasites in mice has been demonstrated, using cationic PC–stearylamine liposomes. When the charged phospholipids were encapsulated in liposomes, their binding to macrophages was shown to be greatly enhanced.Citation63
To improve liposome stability and enhance blood circulation time, sterically stabilized liposomes have been introduced: the addition of polyethylene glycol (PEG) to the liposome surface slows removal from the blood, improving half-life (eg, Doxil®).Citation64 PEGylated and uncoated liposomes bearing MA in VL-infected dogs were compared;Citation65 the former were more effective, although a mixed formulation of PEGylated and conventional liposomes showed greater activity, than either single formulation, for parasite elimination in the spleen and bone marrow.
Other drugs commonly used as anticancer agents have been encapsulated in sterically stabilized liposomes: camptothecinCitation66 and, more recently, doxorubicin have been investigated to integrate MA treatment, so as to decrease the resistance of Leishmania against currently available drugs.Citation67
From the toxicology standpoint, liposome-mediated delivery leads to greatly increased and prolonged concentrations of antileishmanial drugs in the liver and spleen. This is an advantage in a disease such as VL, where macrophages are the target of the therapy, but the macrophages might already be impaired by the infection, and high tissue accumulation may lead to severe side effects.Citation68
In a study aiming to improve the oral bioavailability of AmpB, the antileishmanial activity of two nanocarriers was compared: soya lecithin (Lec)-based biodegradable nanocarriers and liposomes. The surface of the Lec–AmpB nanoparticles was modified with PEG and Tween 20 to further improve their stability and biological activity. PEG nanoparticles showed more promising antileishmanial activity, possibly because of their better interaction with biological systems and membranes.Citation69
To further increase the specificity of action to the diseased area, liposomes decorated with specific ligands have also been developed. This strategy offers a vast potential for sitespecific delivery of drugs to designated cell types or organs, which selectively express or overexpress specific ligands (eg, receptors and cell adhesion molecules) at the site of the disease.Citation70 However, their real therapeutic advantage over nontargeted liposomes is still debatable, with conflicting results being reported; no targeted liposomal product has yet reached the market.
The targeting approach was applied to liposome- encapsulated AmpB, by decorating the particle surface with the naturally occurring macrophage activator tetrapeptide, tuftsin (Thr–Lys–Pro–Arg). Increased in vivo antileishmanial activity vs standard liposomes was observed and appeared to be due both to improved drug tolerance of the liposomal formulation (like that of AmBisome but with a DL of 2%) and to the increased specific uptake of tuftsin by the macrophages.Citation71
C-type lectin receptors (CLRs) are highly conserved pattern recognition receptors with carbohydrate recognition domains that bind sugars (eg, mannose, fucose, and N-acetylglucosamine), present on the surface of many pathogens.Citation72 In particular, the mannose receptor (also called CD206) was the first reported member of a family of four endocytic CLRs receptors; it plays a role in the clearance of endogenous glycoproteins and in pathogen recognition/antigen presentation. The targeting of pattern recognition receptors on macrophages offers the advantage of triggering specific signaling pathways, thus inducing a tailored and robust immune response. Antileishmanial drugs have been encapsulated in mannosylated or fucosylated liposomes to target the corresponding receptors expressed by macrophages; they demonstrated successful treatment of experimental leishmaniasis in a hamster model.Citation73 Furthermore, a biodistribution study clearly showed that uptake of mannosylated liposomes in the liver and spleen was higher than either free AmpB solution or cationic liposomal formulation, indicating that active targeting to the RES occurred.Citation73
Nanoemulsions (NEs) and niosomes
NEs are mixtures of two normally immiscible liquids that are stabilized by using a surfactant (emulsifier). The addition of surfactant is critical for the creation of small droplets (~100 nm) as it decreases the interfacial tension. The emulsifier also plays a role in stabilizing NE through repulsive electrostatic interactions and steric hindrance.Citation74 Lipid AmpB formulations potentially suited to oral administration have been developed based on mono- and diglycerides with or without a lipophilic derivative of vitamin E, d-α-tocopheryl polyethylene glycol 1000 succinate (TPGS), as an alternative to the less temperature-stable monoglyceride. The formulation, named ICo-010, retained AmpB in simulated gastric and intestinal fluids and exhibited a significant antileishmanial activity in a VL-infected murine model.Citation75,Citation76 ICo-010, currently being developed by iCo therapeutics, was granted orphan drug status by the FDA, but recent data from the company website do not show any further improvement from preclinical evaluations.Citation197
A NE loaded with doxorubicin (NE–DOX) was further grafted with phosphatidylserine (PS) to enhance the cellular uptake.Citation77 There was significant improvement in activity with PS–NE–DOX on the spleen parasitic burden in Leishmania-infected hamsters, in comparison to uncoated NE or free drug.
Niosomes are vesicles consisting of nonionic surfactants with cholesterol. These are biodegradable, somewhat less toxic and more stable than liposomes, and relatively low in cost, making them an alternative to liposomes. These particular characteristics make niosomes promising candidates for commercial manufacturing.Citation78 In an early study on delivery systems for VL, niosomal SSG was found to be more effective than liposomal vesicular formulations or than free drug, against experimental murine VL.Citation79 More recently, the effect of itraconazole niosomes on the in vitro susceptibility of L. tropica was demonstrated.Citation80 Entrapment efficacy of AmpB into niosomes has been also reported.Citation81
Lipid cochleates
An interesting approach to prepare lipid systems for oral delivery is that of nanocochleates, cigar-shaped nanostructures composed of negatively charged lipid bilayers (usually PS) bridged by a divalent cation, normally calcium.Citation82 Calcium induces dehydration of the interbilayer domains, and consequently, the amount of water in this region is low, allowing better encapsulation efficiency of hydrophobic drugs, which have a high affinity for the hydrophobic interior of the lipid bilayers. AmpB is an ideal model for hydrophobic drugs. Nanocochleates containing AmpB or AmpB deoxycholate were in vitro tested against L. chagasi, and results showed similar activity.Citation83,Citation84 Lipid cochleates containing both AmpB and MF were also produced, and the incorporation stability was estimated.Citation85 Some clinical trials for VL were initiated on the basis of the results obtained with an oral nanocochleate formulation of AmpB (Bioral® Amphotericin B), which reached Phase I development in the USA for the treatment of mycoses, but nowadays, the research appears to be discontinued. However, Matinas BioPharma is developing MAT 2203, comprising nanocochleates of AmpB for oral administration in treating asperigillosis.Citation198
Solid lipid nanoparticles (SLNs)
SLNs are nanospheres made from solid lipids, with size in the range of 50–1,000 nm. They consist of a solid lipid matrix, ie, glycerides, fatty acids, and waxes, stabilized by physiologically compatible emulsifiers, such as phospholipids, bile salts, Tween, polyoxyethylene ethers, and polyvinyl alcohol. The lipids used in their production are solid at room temperature, and most of them have approved status, eg, Generally Recognized as Safe (GRAS), due to their low toxicity.Citation86
Chitosan-coated SLNs loaded with AmpB have been developed for immunoadjuvant chemotherapy of Leishmania infections. The in vitro antileishmanial activity revealed that chitosan-coated AmpB–SLNs were markedly more potent than commercial formulations (AmBisome and Fungizone). Furthermore, cytotoxic evaluation in macrophages and acute toxicity study in mice evidenced a better safety profile of the formulation compared to marketed products.Citation87
More recently, PM was incorporated into SLN made of stearic acid and Tween 80, and both characterization and in vivo data were reported.Citation88,Citation89 As with liposomes, it has been attempted to increase the uptake by macrophages by targeting to mannose receptors. Mannose-coated lipid nanoparticles of AmpB showed higher liver uptake and promising antileishmanial activity.Citation90 Interestingly, SLN can also be used to improve oral administration protocols (AmbiOnp formulation),Citation91 although this has not yet been applied in VL therapy. The loading of AmpB in the nontoxic superaggregated form results in enhancing the oral bioavailability and increasing their safety profile.Citation91
To reduce the side effects that limited the use of MF, delivery in lipid nanoparticles has been proposed.Citation92 MF formulations were prepared using glycerin, cottonseed oil, oleic acid, cholesterol, and the emulsifiers, such as egg PC and Tween 80. A reduction in epithelial irritation in the gastrointestinal tract was observed in an in vivo study performed in Balb/C mice. Compared to the free drug, the formulation reduced in vitro hemolytic action and cytotoxic activity against macrophages and resulted in comparable level of in vitro cytotoxicity against promastigote and amastigote forms of L. chagasi.Citation92
Polymer nanoparticles
Polymer nanoparticles may be of value in treating infectious diseases such as leishmaniasis, since their small size enables them to pass through biological barriers upon parenteral administration, also enhancing cellular uptake and enabling therapeutic agents to be delivered to infected tissues.Citation10 Polymer nanocarriers can be modulated to give them advanced physicochemical properties, such as increased bioavailability, and precisely defined biodegradability, affording release of the encapsulated drug. Polymer system may consist of either a polymeric matrix (nano- or microspheres) or a reservoir system (nano- or microcapsules). Several different synthetic polymers can be used to prepare nanoparticles, including poly(lactic acid) (PLA), poly(glycolic acid) (PGA), poly(lactide-co-glycolide) (PLGA), poly(caprolactone) (PCL), and poly(cyanoacrylate) (PCA).Citation93 However, natural protein polymers, such as albumin and gelatin, and polysaccharides, such as chitosan, alginate, and starch, have also been described as candidates for the delivery of particle-based antileishmanial systems.Citation94 Other polymers, such as PEG and N-(2-hydroxypropyl)methacrylamide (HPMA), have been used predominantly to produce polymer–drug conjugates in the form of macromolecular prodrugs. In this approach, the release mechanism entails splitting the linker between the polymer and the bioactive agent.
Concerning synthetic polymers, the first studies involved the use of PCA and PCL, loading different drugs with interesting results.Citation95 However, in recent years, PLGA has been one of the most attractive polymeric candidates, used to fabricate devices for drug delivery and tissue engineering applications.Citation96 Currently, 15 FDA-approved PLGA-based drug products are available on the US market.Citation199
Different research groups have investigated AmpB-loaded PLGA nanoparticles. In particular, the efficacy of PLGA nanoparticle-encapsulated AmpB was compared to free drug or marketed formulations against intracellular L. infantum amastigotes as well as promastigotes. One nanoparticle formulation was more effective than AmBi-some and, at the same time, less cytotoxic and hemotoxic.Citation97 Although very promising, especially as an antifungal agent, no further in vivo clinical applications have since been reported.
AmpB has been entrapped in PLGA nanoparticles to improve its oral bioavailability and to reduce its nephrotoxicity.Citation53 The addition of the stabilizer TPGS permitted the formation of stable nanoparticles of <200 nm in diameter. It is worth remembering that TPGS, reported as a P-gp efflux inhibitor, is currently approved by the FDA for use as an excipient in various nanoparticle formulations and also as a solubilizing and emulsifying agent for poorly soluble compounds to improve their absorption and bioavailability.Citation98 The AmpB–PLGA formulation containing TPGS reduced drug nephrotoxicity on administration by either the oral route or the iv route compared with Fungizone. The relative oral bioavailability of AmpB was found to be about eight times higher when it was formulated in polymer nanoparticles than when it was administered as Fungizone.Citation99 Further study on in vivo activity has only been reported for pulmonary aspergillosis.Citation100
MF loaded in PLGA–PEG nanoparticles also showed a fourfold increase in in vitro activity and in in vivo antileishmanial efficacy and bioavailability vs free drug.Citation101
Lipid–polymer hybrid nanoparticles (LPNPs) loading AmpB with the anionic core composed of PLGA polymer and TPGS surfactant and the shell made of cationic stearylamine lipid have also been prepared.Citation102 Stearylamine was selected for its cationic nature, biocompatibility, benign antiprotozoal activity, and immunopotentiation strength. LPNPs containing an interesting amount of AmpB (DL 9.8%, w/w) were reported. Receptor-mediated LPNP identification ensures considerable uptake into the macrophage cells and RES organs (spleen, liver, and so on), while uptake by the kidneys is very low. Furthermore, the positive charge of LPNPs allows them to bind to negatively charged sialic acid molecules on the macrophage surface, resulting in adsorption-mediated endocytosis. In vivo experiments demonstrated significantly stronger parasite growth inhibition (89.4%) in patients treated with AmpB–LPNPs than in those treated with carrier without stearylamine (63.6% inhibition), whereas 69.3% parasite inhibition was observed with the AmBisome formulation.Citation102
With regard to the active targeting approach, mannose-bearing PLGA nano/microparticles have been designed to further improve macrophage targeting of antileishmanial agents. Mannose-grafted PLGA nanoparticles, with and without a PEG spacer, are reported to target AmpB;Citation103 the presence of PEG spacer resulted in a more efficient inhibition of parasites in comparison to both the one without spacer and the free drug. Furthermore, both drug–polymer interaction and drug miscibility in the polymer affect the drug content of polymer nanoparticles. The PLGA hydrophobic core facilitates the loading of amphiphilic AmpB. The polymer slow precipitation, due to the slow rate of solvent removal, allowed more time for drug molecules to partition into the aqueous phase, leading to low % encapsulation efficiency. However, in the engineered version of polymer nanoparticles, in which mannose was directly linked to PLGA (M–PNPs) or to the edge of PEG (M–PEG–PNPs), an increased entrapment efficiency was observed, probably due to the higher drug–polymer miscibility, permitting the enhanced drug incorporation.Citation103 The significant rise in drug content in M–PNPs and M–PEG–PNPs might be due to the stronger drug–polymer interaction (between the hydroxyl group of mannose and PEG and the amine group of AmpB) and to the high miscibility in the case of amphiphilic AmpB. Another study suggested that the acidic end-group of PLGA is responsible for drug incorporation and that increased chain length may increase the drug–polymer interactions, leading to improved drug incorporation efficiency.Citation104
More recently, carbohydrate (mannose, mannosamine, and mannan)-functionalized PLGA nanospheres loaded with AmpB have been investigated in the treatment of murine VL.Citation105 The uptake of mannan-coated nanoparticles (MNs) by macrophages was elevated. Furthermore, a single injection and a 1-week course of treatment with MN–PLGA AmpB nanoparticles were sufficient to achieve a significant decrease in the visceral organs parasite load, although AmpB loading was only 0.58% (w/w). The successful results were probably due to increased production of cytokines relevant in VL infection, such as INF-gamma, and nitric oxide (NO), which plays a key role in the organism’s defense against parasite infection.Citation105
Polymeric nanosystems are also feasible as carriers for multiple drugs. In a recent study, PLGA nanoparticles loaded with AmpB and doxorubicin and further coated with chitosan as a macrophage targeting agent were developed. Chitosan-coated nanoparticles provide a signal for specialized phagocytes, because chitosan recognition involves multiple macrophage surface receptors inducing the drug internalization into cells. Furthermore, this system demonstrated slow drug release to macrophages, with minimal hemotoxicity and tissue toxicity.Citation106 Currently, no in vivo data on VL infected rodent models are available.
AmpB was recently targeted by PLGA nanoparticles using a coating with glycoprotein lactoferrin (Lcf).Citation107 Lcf is an 80 kDa iron-binding glycoprotein with a single polypeptide chain of ~700 amino acids that provides several potential N-glycosylation sites. These sites of Lcf are recognized by the group of CLRs, ie, mannose receptors, which present as biomarkers on the monocyte/macrophage and dendritic cell surfaces. In vivo tests demonstrated significant in vivo antileishmanial activity of Lcf–PLGA–AmpB, compared with nontargeted and commercial formulations, as well as lower nephrotoxicity, attributed to the slower drug release. This might permit the administration of much higher drug doses, leading to increased efficacy in VL treating. It is worth noting that peptide fragments derived from Lcf, such as lactoferricin, exerted interesting activity against L. donovani promastigotes.Citation108 AmpB encapsulated into PEG–PLGA nanoparticles, further decorated with anti-CD14 antibodies, was recently evaluated for macrophage localization improvement.Citation109 CD14 is mainly present on macrophages surface and, to a lesser extent, on neutrophils and dendritic cells. Interestingly, evaluations on promastigote and amastigote revealed that the inhibition efficiency of nanoparticles was higher than that of free drug.
Polysaccharide polymers
Polysaccharides are ideal candidates for drug delivery and biomedical applications, as they are easily obtained from natural sources. Examples of polysaccharides of plant origin include starch, cellulose, hemicellulose, hyaluronic acids, alginate, and guar gums, whereas polysaccharides originating from animals include chitin and chondroitin sulfate.Citation110 Natural polysaccharides have been evaluated for their ability to act as antileishmanial agents, in some cases based on their use in traditional medicineCitation111 and in others from exploring novel sources such as marine macroalgae, which contain sulfated polysaccharides with antileishmanial activity.Citation112 Polysaccharides with different structures act by different mechanisms in macrophages:Citation113 sulfated heterorhamnan, iota-/nu-carrageenans, and arabinogalactan polysaccharides demonstrated significant macrophage activation and a subsequent inhibition of intracellular amastigotes of L. amazonensis. The antileishmanial effects of the polysaccharides occurred by different mechanisms: for sulfated polysaccharides, this may be an expressive increase in NO production by macrophages, while for arabinogalactan, it may be attributed to stimulation of O2•− and TNF production.Citation113
Polysaccharide polymers have also been used to deliver vanadium complexes, which are reported to be highly active metal-based drugs against a variety of trypanosomatids, including Leishmania species.Citation114 The immunomodulatory effects of the polysaccharides arabinogalactan, galactomannan, and xyloglucan, as well as their oxovanadium (IV/V) complexes, were evaluated on peritoneal macrophages exhibiting significant leishmanicidal activity (~60%–70%). The mechanism of action appears to be due to increasing levels of IL-1b and IL-6 released by macrophages.Citation115 Vanadium complexes of stilbene demonstrated leishmanicidal activity in promastigote forms of L. amazonensis, inducing reactive oxygen species (ROS) production and NO-dependent microbicidal action.Citation116
Biopolymers such as chitosan and sodium alginate are potent macrophages activators, inducing the release of a range of cytokines and cytotoxic agents.Citation117–Citation119 Development of a biocompatible polymers formulation, which enhances immunological responses, might thus provide a new approach to improve leishmaniasis treatment. Naturally occurring poly-saccharides, such as chitosan and alginate, have been utilized to deliver antileishmanial agents in nanoparticles. Nanometric AmpB-encapsulated chitosan nanocapsules (CNC–AmpB) have been formulated using a polymeric deposition technique mediated by NE template fabrication.Citation120 The lipid core of the nanocapsule consisted of surfactant, glycerol, soybean oil, and soya lecithin. Enhanced efficacy of CNC–AmpB is likely due to active macrophage targeting by N-acetyl-glucosamine unit of chitosan content of CNC, recognition being mediated by major histocompatibility complex class I and II, Fc receptors, and mannose receptors. The same team improved the targeting capability of chitosan, using mannosylated chitosan (Mnos-CS) copolymer, subsequently formulated in a nanocapsule system (MnosCNc) for the delivery of AmpB.Citation121 Using nanocapsules, DL of 9%–10% (w/w) was obtained. In vivo results showed that more drug was delivered by mannosylated nanocapsules than by CNC or plain AmpB in both liver and spleen.
Polyelectrolyte complexes (PECs) are formed by interactions between two macromolecules bearing oppositely charged groups. Encapsulation of a drug during formation of PECs has shown great promise for preparing drug delivery carriers. For this purpose, the native positively charged polymer chitosan, and chondroitin sulfate, of negative charge was used to load AmpB,Citation122 resulting in a good DL (11%, w/w). In evaluating the antileishmanial activity, it was observed that chitosan itself presented activity and a synergistic interaction between chitosan and chondroitin sulfate also occurred. The activity of AmpB nanoparticles was similar to those with pure AmpB; however, the cytotoxicity of the nanoparticle formulation was approximately one-tenth that of pure AmpB. Neither pure chitosan nor chondroitin sulfate, or the nanoparticles, presented any significant macrophages toxicity.Citation122
Chitosan was also chemically modified into glycol chitosan stearate, an amphiphilic copolymer that forms carriers with a hydrophilic shell and a hydrophobic core comprising stearic acid.Citation123 By increasing the rigidity of the bilayer of so-called lipo-polymerosomes (L-Psomes) using cholesterol, a very large amount of AmpB was loaded (25%), exploiting the hydrophobic interaction with the stearic acid chain. The AmpB–L-Psomes proved to be a biologically safe and stable delivery system, with improved efficacy compared to commercial formulations; they can thus be proposed as an alternative low-cost product. A further study exploited a surface interaction between chitosan and sodium alginate (natural polymer with anionic charge) to stabilize the nanoparticles, maintaining the high AmpB loading (22%).Citation124 The biopolymers sodium alginate and chitosan are potent macrophage activators, inducing the release of different cytokines and cytotoxic agents.Citation118 In vivo study demonstrated that L-Psomes facilitated the AmpB uptake in RES-rich organs and showed that AmpB remains in the tissues for prolonged periods, followed by slower distribution of the drug into the blood stream. Increased tolerance to AmpB was also observed.Citation118
Alginate has also been used as active coating of a liposomal AmpB preparationCitation117 and, more recently, of doxorubicin loaded in an NE (soybean oil with Tween as surfactant and protamine sulfate).Citation125 This treatment strategy is based on the immune response modulation, combined with chemotherapy, to improve efficacy during infection.
Ionic amphiphile biovectors, first reported by Loiseau et al,Citation126 comprised anionic lipids (dipalmitoyl phosphatidyl glycerol) included in a cationic cross-linked maltodextrin matrix, used as reservoir for AmpB. The nanoparticles of 100 nm were loaded with 20% (w/w) AmpB and demonstrated a high stability. The in vitro and in vivo activities remained in the same dose and concentration range as Fungizone and AmBisome, suggesting that their interaction with host cells, their uptake, and intracellular distribution were efficient, although the formulation is slightly less active and safe than AmBisome.Citation126 Although in the early development stage, ionic amphiphile biovectors have not been further improved or described. However, hydroxypropyl-γ-cyclodextrin was used to solubilize AmpB solution (AmpB–dextrin solution) by inclusion; it was then formulated into nonionic surfactant vesicles (NIVs) (consisting of a 3:3:1 molar ratio of mono-n-hexadecylether tetraethylene glycol to cholesterol to dicetylphosphate). The antileishmanial activity of NIV and chitosan-coated NIV was compared with that of commercial AmpB formulations.Citation127 It has been shown that the pulmonary route is not only suitable for lung conditions but can also be used to target the liver: treatment with an NIV formulation of AmpB significantly reduced L. donovani liver parasite burdens in treated mice and was also effective at reducing Aspergillus levels in the lungs.Citation100 Furthermore, a reduced toxicity in L. major-infected mice after several (15) doses indicated that both formulations, as well as the pulmonary route, minimized systemic exposure to AmpB.
Polymeric micelles
Polymeric micelles are nanoscopic core/shell structures made up of amphiphilic block copolymers. Both the intrinsic and the modifiable properties of polymeric micelles make them particularly well suited for drug delivery purposes.Citation128 Among the polymers used for micelle preparation, poloxamers (α-hydro-ω-hydroxypoly[oxyethylene]a poly[oxypropylene]b poly[oxyethylene]a block copolymers) present interesting behavior and have been applied in a wide range of biomedical and pharmaceutical industries. Particularly, poloxamer in aqueous media exhibits micellar structures, which can convert into gel-like structures depending on their length, concentration, and temperature. In the search for new delivery systems to treat tegumentary leishmaniasis, AmpB-containing a polymeric micelle system (namely AmpB/M) was developed using poloxamer P407 (Pluronic® F127) and its in vitro and in vivo potency against L. amazonensis was evaluated.Citation129 When administered by subcutaneous injection in mice, the AmpB/M formulation turns into a semirigid gel on contact with local tissue, creating a reservoir system and keeping the drug in the extracellular space; this allows it to act against parasites at the local infection site. Over time, as the gel matrix is diluted by body fluids and phagocytosis, the drug is gradually released into systemic circulation, enabling its systemic action to be controlled.
A poloxamer P407-based paramomycin-containing micelle nanogel system has been evaluated. The system was tested for in vivo tolerance, ex vivo cytotoxicity on cells, and antileishmanial activity against promastigotes. The results showed the product to possess negligible toxicity and effective antileishmanial activity against L. major and L. infantum promastigotes. It was concluded that the formulation provides controlled, effective, and safe delivery of paramomycin in mice.Citation130
An interesting approach to obtain stable micelles with high DL is based on the self-assembling ability of prodrugs with amphiphilic properties. Regarding pentavalent antimonials, the use of nonionic surfactants as complex agents has been investigated.Citation131 The formation of an amphiphilic antimony (V) complex using N-alkyl-N-methylglucamide (with carbon chain lengths of 8 and 10) produced stable nanodispersions (~115 nm). The most significant advantages of the system were its great solubility in water, its superior bioavailability, and sustained drug release properties. These results were much better than those obtained with a cyclodextrin-based oral formulation of MA proposed by the same group.Citation132 Micelles of the amphiphilic antimony (V) complex may also offer a possibility for solubilizing lipophilic drugs and achieving oral coadministration of anti-VL drugs.
Inorganic compounds as delivery systems
The majority of nanoparticles of pharmaceutical interest are made of organic polymers (biodegradable or not), but inorganic systems, as well as organic–inorganic hybrid materials, are now also receiving considerable attention in the pharmaceutical field. Mesoporous silica nanoparticles, hydroxyapatite nanostructures, metallic nanoparticles such as Fe3O4, gold, and silver nanoparticles, and carbon-based nanostructures (nanotubes, graphene, nano-onions, and horns) exhibit several interesting features and enable different functionalities (eg, diagnostic and therapeutic) to be combined in a single device.Citation133,Citation134
Small clearly defined aggregates of noble metals can be produced with very precise and controlled methods, modulating the reaction conditions used in their preparation. Metals in nanoparticulate form might be of interest in treating leishmaniasis, owing to their ability to produce ROS. In particular, silver nanoparticlesCitation135 were found to inhibit the survival of amastigotes in host cells and, in the presence of UV light, the effect was more significant. L. tropica promastigotes lost their shape, and their internal organelles were no longer distinguishable.Citation135
Metal oxide nanoparticles, especially titanium dioxide (TiO2), silver oxide (Ag2O), zinc oxide (ZnO), magnesium oxide (MgO), and, more recently, selenium oxide (SeO2) nanoparticles, have been extensively explored, demonstrating a significant antibacterial activity.Citation136 Among metals, the highest antileishmanial activity was observed for nanoparticles composed of Ag, followed by Au, Ti, Zn, and Mg. Both UV and infrared irradiation increased the antileishmanial properties of all these nanoparticles. However, they were found to possess macrophages cytotoxicity. Selenium nanoparticles demonstrated less cytotoxic effect on uninfected macrophages but have stronger antiamastigote and antipromastigote activities than SeO2.Citation137
Another study focused on the development of six biocompatible nanoparticles of ZnO doped with different concentrations of Cu and their in vitro efficacy against the Leishmania parasite.Citation138 Especially those from ZnCuO to ZnCuO5 have been proposed against leishmaniasis, because of their biocompatibility and enhanced ROS yield: after irradiation, they generated measurable quantities of singlet oxygen. Similarly, with Ag as dopant of the semiconductor, nanoparticles of zinc oxide demonstrated the production of ROS when activated by daylight and were able to kill L. tropica.Citation139
More recently, two approaches were combined, using AmpB adsorbed on preformed spherical biogenic silver nanoparticles surface. AmB–Ag nanoparticles were more potent in inhibiting L. tropica in in vitro tests. Furthermore, the antileishmanial activity of the prepared nanoparticles was enhanced upon irradiation with visible light, which may be attributed to the formation of excess ROS by the released silver ions. The study limitation is the lack of the information about the amount of AmpB loaded.Citation140
Among the inorganic nanomaterials, carbon compounds are attracting interest due to their excellent mechanical, thermal, and optical properties. Drugs may be covalently linked to carbon nanotubes (CNTs) or strongly absorbed by π–π stacking on their surface (eg, doxorubicin). However in its early ages in anti-Leishmania applications, a way of using multiwalled CNT to deliver covalently bound AmpB has been devised.Citation141 Unusually, this product was obtained without using any condensing agent and was thought to be covalently linked via an ether bond instead of an amide bond. The intraperitoneal administration route led to a higher percentage suppression of parasites in the spleen with CNT–AmpB (89.8%) than with AmpB (68.9%). Without any improvement in characterization, the same formulation was orally administered, resulting in antileishmanial activity in hamster spleen tissues.Citation142
Due to their dimensions, similar to those of the inorganic components of calcified tissues, nanosized calcium phosphate materials are expected to have better bioactivity and biocompatibility than conventional materials. Furthermore, calcium phosphate is a cheap drug cargo that is chemically stable, composed of inorganic minerals native to the body, which can be metabolized as a nontoxic degradation product.Citation143,Citation144
The preparation of calcium phosphate nanoparticles loaded with AmpB has been reported.Citation145 The method involved mixing two microemulsions containing, separately, calcium chloride and disodium phosphate, and sodium silicate in the presence of a surfactant. Nanoparticles <200 nm in size, with an interesting AmpB loading of 11%, were obtained. In vivo efficacy of the formulation was assessed in a L. donovani-infected golden hamster model. The in vivo activity of AmpB-loaded nanoparticles was stronger than that of marketed formulations or AmpB suspension, which is the collective outcome of higher internalization by macrophages via clathrin receptor-mediated phagocytosis and enhancement of Th1-biased immune response by nanoparticles, which ultimately targeted macrophages and retarded nonspecific distribution of AmpB. Furthermore, the calcium phosphate nanoformulation was able to release the encapsulated drug in a slow and sustained manner, with preferential accumulation in macrophage-rich organs such as liver and spleen. A toxicity assessment study was also reported.Citation145
Similar approach was also used to investigate the potential oral administration of pentavalent antimonials. Nanoparticle phosphate-based composites containing Sb(V) have been explored in terms of their in vitro activity.Citation146 This system allowed the Sb(V) delivery into the cells in which the parasite is internalized, and the elevation of calcium concentrations inside the vacuoles might increase parasite apoptosis.
Covalently linked drug to polymers
In the nanosystems described earlier, the drug was always adsorbed, loaded, or entrapped in its native form. The covalent conjugation of drugs to an appropriate carrier is another strategy used to deliver drugs to particular target sites. The development of specific carrier–drug conjugates was first proposed for the delivery of small hydrophobic drug molecules to their sites of actionCitation147 and has recently reached significant results in anticancer therapy with antibody–drug conjugates. These delivery systems basically comprise a water-soluble polymer, bearing a number of drug molecules covalently linked. The main advantages of this approach are 1) increased water solubility of little soluble or insoluble drugs, therefore enhancing drug bioavailability; 2) protection of drugs from deactivation and relative activity preservation during circulation; and 3) most importantly, specifically drug targeting to the site of action. Unlike other approaches, in this case, the active agent must be considered as a prodrug that generates the activity only after release. For this reason, the role of linker between polymer and drug is of paramount importance, because it defines the mechanism for drug release (pH, enzyme derived, chemical reaction, and so on). Although many are undergoing the final phase of clinical trials and several polymer–protein conjugates have been approved by the FDA for clinical use, to date no polymer–synthetic drug conjugate has been approved.Citation148
Polymer–drug conjugates have been used for targeting macrophages and have already shown potential in antileishmanial chemotherapy. HPMA is undoubtedly the best-known synthetic polymer exploited in macromolecular conjugates. It is water soluble and nonimmunogenic and has undergone clinical trials in the cancer field.Citation149 In a detailed article, the synthesis of HPMA conjugate of AmpB obtained through a degradable GlyPheLeuGly linker is described. The linker was chosen as it is known to be cleaved by cathepsin B, which has been found in the PV.Citation150 Its in vitro activity was comparable to that of free AmpB and Fungizone, and although less active than AmBisome in in vivo experiments (though of a similar order), no signs of toxicity were recorded, even at the maximum used dose of 3 mg/kg AmpB equivalent used. It should be noted that this dose is above the reported 50% lethal dose (LD50) of 2.5 mg/kg for Fungizone. No significant difference in antileishmanial activity was found using a similar but targeted conjugate with mannose.Citation150 Further studies on polymer–drug conjugates in this field have not been reported, and clinical evaluation is thus lacking.
Another interesting natural polymer that can act as delivery platform and targeting scaffold simultaneously is hyaluronic acid.Citation151 Recently, a versatile synthetic method to link pentamidine to hyaluronic acid, affording DL of up to 33% (w/w), was presented. Assayed against the intracellular amastigote form of the parasite, the bioconjugate was more efficient than free drug in the internalization of infected macrophages but, at the same time, less cytotoxic.Citation152
Novel products with antileishmanial activity combined into nanocarriers
The need for alternative treatments, in particular to reduce the duration of clinical treatment, led to a program to screen natural products for potential use in leishmaniasis therapy, as recent reviews report,Citation153–Citation156 and also to the screening of currently available medicines with potential use in VL. In parallel, quantitative structure–activity relationship (QSAR) study has been used to identify new organic compounds with stronger activity.Citation157 Another approach examined the role played by known active molecules in medicinal plants traditionally used against leishmaniasis, as well as in documented plants that have yet to be explored.Citation158–Citation161
Several active agents from different chemical classes are also reported to have in vitro antileishmanial activity. The more promising candidates appear to be the nitroimidazoles, quinoline scaffold-based derivatives such as indolyl quinoline analogs, naphthoquinones, the cytotoxic drug doxorubicin, and also antimicrobial peptides.Citation108,Citation162 A recent review clearly summarizes these results.Citation163 Disulfiram, a drug extensively used to treat alcoholism, has recently been reported to be active at nanomolar concentrations against the intracellular amastigotes of L. donovani.Citation164 Although clinical trials have not yet been carried out to assess the efficacy of disulfiram as monotherapy or in combination to treat different types of leishmaniasis, the implementation of a clinical trial is feasible, since disulfiram is a known and approved drug and, furthermore, is administered orally.
OlPC is a structural analog of MF. The in vitro efficacy of OlPC against the intracellular amastigotes of different Leishmania spp. was similar to the profile of MF, with IC50 values in the low micromolar range, except for L. major and L. braziliensis.Citation165 When tested on a VL hamster model with L. infantum, OlPC was found to be more potent than MF after multiple or single oral dosing. Furthermore, short oral treatment with OlPC improves clinical signs of canine L. infantum leishmaniasis.Citation166 The pharmacokinetic profiles of OlPC, administered either in water or as a liposomal preparation, have also been reported recently.Citation167
Natural agent, namely andrographolide, is a diterpenoid lactone extracted from the leaves of Andrographis paniculata, with strong antiparasitic and antileishmanial activity.Citation168 Further progress was achieved upon studying andrographolide’s behavior regarding resistance, with PLGA nanoparticles loaded with andrographolide and stabilized with vitamin E TPGS.Citation169 Nanoparticles were delivered into macrophage cells infested with sensitive and drug-resistant amastigotes of L. donovani parasites. Antileishmanial activity was found to be significant for the nanoparticles with TPGS at about one-tenth of the free drug dosage and one-third of the nanoparticles without TPGS dosage. When andrographolide was incorporated in TPGS nanoparticles, the selectivity of andrographolide increased eightfold in resistant cells.Citation169
Recently, the same approach was used to deliver oleanolic acid (OA), a naturally occurring pentacyclic triterpene with antileishmanial activity.Citation170 In vivo evaluation indicated that OA suppressed the amastigote burden in the spleen after the fourth dose (67.69%±4.12%), whereas the percentage of suppression was increased to 98.82%±1.92% in the case of OA PLGA nanoformulation.
A further example concerns bisnaphthalimidopropyl (BNIP) derivatives, which exert significant antiproliferative activity on L. infantum.Citation171 Encapsulation of BNIP aminooctane derivative into PLGA nanoparticles resulted in a reduction of aspecific toxicity and in the 80% increase of in vivo efficiency on L. infantum compared to free drug.Citation172 This nanoformulation remained active after oral administration in infected BALB/c mice.
Artemisinin, a potent drug used against Plasmodium falciparum malaria, potentially possesses antileishmanial activity, although bioavailability and stability are low.Citation173 Encapsulating artemisinin into PLGA nanoparticles reduced the free drug macrophage toxicity, increasing leishmanicidal activity.Citation174 Furthermore, NO production may intercede in the antileishmanial effect of the nanoformulations. Unfortunately, the curative effect was not as high as the standard liposomal AmpB formulation.Citation175
Curcumin (diferuloylmethane), the main yellow bioactive component of turmeric, has been shown to have a wide spectrum of biological action, including leishmanicidal activity in vitro.Citation176 Different approaches have been proposed to deliver curcumin. A particular type of prodrug-based approach, named “squalenoylation” has been proposed.Citation177 In this approach, the derivatization of small molecules by conjugation with a squalene moiety enables conjugates to be produced that spontaneously self-assemble into 60–300 nm nanoparticles. Although to date it has mainly been applied in the anticancer field,Citation178 a squalenoyl prodrug of curcumin was recently investigated in promastigote and amastigote forms of L. donovani and showed enhanced activity vs the parent drug.Citation179 Recently, PLGA nanoparticles loaded with curcumin (nanocurcumin) were proposed to support MF in exerting leishmanicidal effect at a subcurative dose through immunomodulation. Nanocurcumin monotherapy or combination therapy with MF did not exhibit any hepato- or nephrotoxicity effect.Citation180
Compound 8-hydroxyquinoline (8-HQN) and its derivates have been experimentally evaluated in different studies as chemotherapeutic agents with interesting anti-leishmaniasis activity.Citation181,Citation182 Recently, polymeric micelle system was exploited to deliver 8-HQN.Citation183 After daily subcutaneous injections for 15 days in L. infantum-infected mice, the treated group showed a more marked reduction in the parasite load than the control group. The formulation was highly effective in treating the disease, more so than free 8-HQN or AmpB; the highest levels of nitrite were found in the 8-HQN/M-treated mice, together with a significant Th1 immune response.
In a different approach NPC1161, a model 8-aminoquinoline drug with antileishmanial activity was conjugated with HPMA and targeted with N-acetylmannosamine.Citation184
Inorganic carrier such as CNTs was recently used to deliver betulin, a pentacyclictriterpenoid with antileishmanial properties.Citation185 Counting the amastigote cells inside the infected macrophage cells using a microscope, a dose-dependent response was observed with betulin and with CNT-linked betulin, whereas CNT alone did not show any antileishmanial effect. In addition, CNT was seen to downregulate the activity of P-gp efflux, which would help to increase the bioavailability of any drug incorporated into the carrier system and overcome resistance mechanisms. However, toxicity data are still limited, and further effort will be required to evaluate in depth not only the potency but also the in vivo fate of these formulations (eg, biodistribution and elimination from the body).Citation185 A complete toxicological evaluation must also be made before they can be administered safely.
Discussion
At present, only a small number of antileishmanial drugs are available for clinical use: three injectables and the sole orally administrable MF. There is thus a pressing need to develop other antileishmanial drugs as each of the available drugs suffers from various drawbacks (toxicity, repeated dose requirement, and so on).
To reduce dosage, enable improved release, and promote high drug concentrations in the intracellular (macrophage) vesicle where Leishmania is hosted, DDSs may play a crucial role. Colloidal carriers can also protect drugs against extracellular degradation, improving the pharmacokinetic profile of the active agent. The most significant result is the case of AmBisome, which has significantly improved VL treatment, as it provides almost complete eradication of parasite burden, with minor toxic effects. However, the cost of this lipid formulation is a severe deterrent, in the light of the need to provide a reasonably priced therapeutic option to third world countries. Resistance to AmpB has not yet been observed in clinical practice, although it has been reported in in vitro study.Citation29 Thus, the DDS approach had not been yet evaluated on resistant lines.
Since AmpB is currently the most potent antileishmanial agent, the majority of efforts in nanotechnology have been addressed to improving its specificity, reducing its adverse effects, and in some cases, using noninvasive route. For this reason, in this section, we critically evaluate and discuss the role and advantages that may be played by innovative AmpB delivery systems. The principal systems, methods and results, described in the text, are summarized in . Considering the structure, the majority of the systems comprise polymeric nanoparticles or nanocapsules of size (100–300 nm) greater than that of AmBisome. As far as, the stability of the carrier is concerned, the polymeric systems are much more stable than liposomes, and from the regulatory standpoint, the use of polymers that are already approved by FDA/EMA may reduce the need for toxicological studies, in comparison to other systems. PLA and PLGA are widely used in many FDA-approved drug products.Citation96 DL is an important parameter to predict the strength of an approach. If drug capacity is <4%–5% (w/w) of the carrier material, either the quantity of drug administered will not be sufficient to achieve a pharmacologically active concentration in the relevant body compartment capable of eradicating the parasite burden, or the amount of carrier material required will be too large, leading to toxicity.Citation186 The hydrophobic core of PLGA nanoparticle (NP) and LPNP facilitates the loading of AmpB, which can reach significant levels. Another issue in nanoparticle systems that may lead to lower activity, and potentially to more toxicity, is the rapid release or premature “burst release” of the encapsulated drug. This phenomenon is not frequent in the case of AmpB, because in polymeric NP, only a small fraction of the drug is present in the free, monomeric form, the majority remaining superaggregated and associated with the delivery system.Citation97
The majority of nanostructures achieved interesting ranges of DL (up to 44% in one case). Passive or “natural” targeting, since the MPS cells are considered to be the main target for therapeutic interventions in leishmaniasis, is a mechanism that may be used to enhance drug concentration, reducing aspecific toxicity (hematotoxicity and kidney toxicity). Nevertheless, the active targeting approach (using mannose, stearylamine, mannan, and antibody fragments) appears to slightly increase the uptake in liver and spleen lesions. In particular, the surface charge of LPNPs created by stearylamine enhanced uptake by macrophages, demonstrating interesting immunoadjuvant properties. In LPNP, the lipid component of the formulation also behaves as a targeting agent, while in the majority of formulations, surface decoration was preferred. In the latter case, clearly illustrated by the use of antibodies, difficulties may arise because there is as yet no clear information concerning the “optimum” range of ligand characteristics (ie, valency, avidity, conformation, and packing) on the surface of nanocarriers that results in reproducible target binding and internalization. Manufacturing these products may be challenging; this is clearly of great practical importance, as is shown by the lack of actively targeted liposomes on the market for cancer therapy.
Owing to their complexity, site-directed DDSs will always require full and in-depth characterization before being proposed for industrial development and manufacturing. Especially when novel components (lipids, lipid polysaccharides, and so on) with unknown toxicological properties are added to the formulation, or when too many complex steps are required, industrial interest decreases sharply. Regarding cost-effectiveness, PLGA, a pharmaceutically accepted polymer, including from the regulatory standpoint, is more advantageous than more “sophisticated” systems, although these may be of interest, as is the case of L-Psomes or targeted systems.
Nevertheless, it is important to notice that each study, able to combine novelty and promising in vivo results, should be compared with the best treatment available (AmBisome). This comparison was done only in 44% of the studies reported in .
Also molecular conjugates, typically prepared to increase drug pharmacokinetic profile, did not significantly improve the AmpB antileishmanial activity.Citation150 A remarkable review by DuncanCitation148 reflects very clearly on the current status of polymer therapeutics, the major issues concerned, and future opportunities.
Conclusion
Bearing in mind the results obtained in some clinical trials where a single-course iv therapy of 10 mg/kg AmBisome was found to give a 98% cure,Citation26 the space for robust and market-oriented innovation seems to be represented mainly by the delivery of AmpB by oral route, but this delivery must be directed in targeted tissues to elicit its pharmacological effect while reducing its nephrotoxicity and infusion-related side effects.
Even if it remained difficult to select and design an appropriate formulation to improve AmpB oral absorption, some novel DDSs have emerged as effective. In particular, lipid cochleates and an SLN formulation (AmbiOnp) appear very promising, not least because they are already involved in industrial development. Both approaches provide high DL-reduced AmpB toxicity and production protocols that avoid expensive compounds or processes. Further improvement could involve a polymer such as chitosan, which combines bioadhesion and absorption-enhancing properties, to prolong contact between the DDS and the mucosa, increasing the amount of AmpB that can be absorbed.
In recent years, material sciences and biomedical sciences have produced numerous new systems and materials with the potential to treat disease. However, two major limitations must be borne in mind. First, as the recent literature concerning DDSs shows, there is a flourishing strain of articles in which increased complexity appears to be seen as an advantage, without any real justification in terms of the balance between complexity and feasibility. Second, complex delivery systems obviously cost more to develop and manufacture than do conventional therapeutic agents. The resulting higher prices may only be acceptable if the performance of these systems is sufficiently increased, thereby reducing the treatment period and overall cost, leading to savings to the health care system. It is becoming ever more imperative to formulate standards for system characterization, and robust means of comparative testing, so as to facilitate close and critical analysis of proposed strategies. Further efforts and investments will still be necessary for this approach to achieve greater clinical applicability in humans.
Meanwhile, with regard to the identification of novel drugs, there can be no doubt that the availability of the complete genome sequence of Leishmania has given the scientific community the possibility for large-scale analysis, which may lead to a better understanding of the parasite’s biology and consequent identification of novel drug targets.Citation187 Furthermore, the use of powerful CRISPR–Cas9 techniques of genome editing is now making it possible to accelerate the characterization of Leishmania genes, so as to develop new drugs, diagnostics, and vaccines.Citation188 From another standpoint, the medicinal chemistry approach continues to improve “old drugs”, suggesting novel AmpB derivatives with high activity and increased safety.Citation189
However, since the goal is to eliminate parasitic infections, a “discovery science” for the development of drugs and diagnostics must be conducted on the global stage, involving, in the local context, scientists, clinicians, regulators, and health ministries in disease-endemic countries. Furthermore, “implementation science” must take place in the local context. To achieve the goal, it is of fundamental importance to involve multidisciplinary teams, including pharmaceutical and medical scientists, policymakers, social scientists, health administrators, and communication scientists, working together in new ways.Citation190
Disclosure
The authors report no conflicts of interest in this work.
References
- BañulsALHideMPrugnolleFLeishmania and the leishmaniases: a parasite genetic update and advances in taxonomy, epidemiology and pathogenicity in humansAdv Parasitol2007641–109455458
- SéguinODescoteauxALeishmania, the phagosome, and host responses: the journey of a parasiteCell Immunol20163091627531526
- MurrayHWBermanJDDaviesCRSaraviaNGAdvances in leishmaniasisLancet2005366949615611577
- AlvarJVélezIDBernCLeishmaniasis worldwide and global estimates of its incidencePLoS One201275e3567122693548
- LindgrenEAnderssonYSukJESudreBSemenzaJCPublic health: monitoring EU emerging infectious disease risk due to climate changeScience2012336608041841922539705
- Van GriensvenJCarrilloELópez-VélezRLynenLMorenoJLeishmaniasis in immunosuppressed individualsClin Microbiol Infect201420428629924450618
- HotezPJPecoulBRijalSEliminating the neglected tropical diseases: translational science and new technologiesPLoS Negl Trop Dis2016103e000389526934395
- GustafsonHHHolt-CasperDGraingerDWGhandehariHNanoparticle uptake: the phagocyte problemNano Today201510448751026640510
- GutiérrezVSeabraABRegueraRMKhandareJCalderónMNew approaches from nanomedicine for treating leishmaniasisChem Soc Rev201645115216826487097
- AsthanaSGuptaPKChaurasiaMDubeAChourasiaMKPolymeric colloidal particulate systems: intelligent tools for intracellular targeting of antileishmanial cargosExpert Opin Drug Deliv201310121633165124147603
- SrivastavaSShankarPMishraJSinghSPossibilities and challenges for developing a successful vaccine for leishmaniasisParasit Vectors20169127727175732
- BiswasSSubramanianAElmojtabaIMChattopadhyayJSarkarRROptimal combinations of control strategies and cost-effective analysis for visceral leishmaniasis disease transmissionPLoS One2017122e017246528222162
- MeheusFBalasegaramMOlliaroPCost-effectiveness analysis of combination therapies for visceral leishmaniasis in the Indian subcontinentPLoS Negl Trop Dis201049e81820838649
- MeheusFAbuzaidAABaltussenRThe economic burden of visceral leishmaniasis in Sudan: an assessment of provider and household costsAm J Trop Med Hyg20138961146115324189368
- TachfoutiNNajdiAAlonsoSCost of pediatric visceral leishmaniasis care in MoroccoPLoS One2016116e015548227257808
- UranwSMeheusFBaltussenRRijalSBoelaertMThe household costs of visceral leishmaniasis care in south-eastern NepalPLoS Negl Trop Dis201372e206223469298
- Shaked-MishanPUlrichNEphrosMZilbersteinDNovel intracellular SbV reducing activity correlates with antimony susceptibility in Leishmania donovaniJ Biol Chem200127663971397611110784
- WalkerJSaraviaNGInhibition of Leishmania donovani promastigote DNA topoisomerase I and human monocyte DNA topoisomerases I and II by antimonial drugs and classical antitopoisomerase agentJ Parasitol20049051155116215562618
- DemicheliCFrézardFLecouveyMGarnier-SuillerotAAntimony(V) complex formation with adenine nucleosides in aqueous solutionBiochim Biophys Acta20021570319219812020809
- JeddiFPiarrouxRMaryCAntimony resistance in Leishmania, focusing on experimental researchRe Dai Yi Xue Za Zhi20112011695382
- LeandroCCampinoLLeishmaniasis: efflux pumps and chemoresistanceInt J Antimicrob Agents200322335235713678842
- WalshTJGoodmanJLPappasPSafety, tolerance, and pharmacokinetics of high-dose liposomal amphotericin B (AmBisome) in patients infected with Aspergillus species and other filamentous fungi: maximum tolerated dose studyAntimicrob Agents Chemother200145123487349611709329
- BrajtburgJPowderlyWGKobayashiGSMedoffGAmphotericin B: current understanding of mechanisms of actionAntimicrob Agents Chemother19903421831882183713
- OlliaroPLGuerinPJGerstlSHaaskjoldAARottingenJ-ASundarSTreatment options for visceral leishmaniasis: a systematic review of clinical studies done in India, 1980–2004Lancet Infect Dis200551276377416310148
- BalasegaramMRitmeijerKLimaMALiposomal amphotericin B as a treatment for human leishmaniasisExpert Opin Emerg Drugs201217449351023167833
- SundarSChakravartyJAgarwalDRaiMMurrayHWSingle-dose liposomal amphotericin B for visceral leishmaniasis in IndiaN Engl J Med2010362650451220147716
- SundarSChakravartyJAgarwalDShahAAgrawalNRaiMSafety of a pre-formulated amphotericin B lipid emulsion for the treatment of Indian Kala-azarTrop Med Int Health20081391208121218664241
- SundarSPandeyKThakurCPEfficacy and safety of amphotericin B emulsion versus liposomal formulation in Indian patients with visceral leishmaniasis: a randomized, open-label studyPLoS Negl Trop Dis201489e316925233346
- MbongoNLoiseauPMBillionMARobert-GeroMMechanism of amphotericin B resistance in Leishmania donovani promastigotesAntimicrob Agents Chemother19984223523579527785
- PurkaitBKumarANandiNMechanism of amphotericin B resistance in clinical isolates of Leishmania donovaniAntimicrob Agents Chemother20125621031104122123699
- ChawlaBJhingranAPanigrahiAStuartKDMadhubalaRParomomycin affects translation and vesicle-mediated trafficking as revealed by proteomics of paromomycin–susceptible–resistant Leishmania donovaniPLoS One2011610e2666022046323
- SundarSChakravartyJParomomycin in the treatment of leishmaniasisExpert Opin Investig Drugs2008175787794
- DorloTPCBalasegaramMBeijnenJHde VriesPJMiltefosine: a review of its pharmacology and therapeutic efficacy in the treatment of leishmaniasisJ Antimicrob Chemother201267112576259722833634
- World-Health-OrganizationControl of the Leishmaniasis: Report of a Meeting of the WHO Expert Committee (N° 949) on the Control of LeishmaniasesGenevaWorld Health Organization2010
- LuxHHeiseNKlennerTHartDOpperdoesFREther-lipid (alkyl-phospholipid) metabolism and the mechanism of action of ether-lipid analogues in LeishmaniaMol Biochem Parasitol2000111111411087912
- SundarSSinhaPJhaTKOral miltefosine for Indian post-kalaazar dermal leishmaniasis: a randomised trialTrop Med Int Health20131819610023136856
- SrivastavaSMishraJGuptaAKSinghAShankarPSinghSLaboratory confirmed miltefosine resistant cases of visceral leishmaniasis from IndiaParasit Vectors20171014928137296
- AlvarJGonzálezUPinartMInterventions for visceral leishmaniasisCochrane Database Syst Rev20106
- van GriensvenJBoelaertMCombination therapy for visceral leish-maniasisLancet2011377976444344421255829
- SundarSSinhaPKRaiMComparison of short-course multidrug treatment with standard therapy for visceral leishmaniasis in India: an open-label, non-inferiority, randomised controlled trialLancet2011377976447748621255828
- WasunnaMNjengaSBalasegaramMEfficacy and safety of ambisome in combination with sodium stibogluconate or miltefosine and miltefosine monotherapy for African visceral leishmaniasis: phase II randomized trialPLoS Negl Trop Dis2016109e000488027627654
- BarrettMPBoykinDWBrunRTidwellRRHuman African trypanosomiasis: pharmacological re-engagement with a neglected diseaseBr J Pharmacol200715281155117117618313
- DiroERitmeijerKBoelaertMUse of pentamidine as secondary prophylaxis to prevent visceral leishmaniasis relapse in HIV infected patients, the first twelve months of a prospective cohort studyPLoS Negl Trop Dis2015910e000408726431253
- GeorgiadouSPStefosASpanakosGCurrent clinical, laboratory, and treatment outcome characteristics of visceral leishmaniasis: results from a seven-year retrospective study in GreeceInt J Infect Dis201534465025743761
- BasselinMCoombsGHBarrettMPPutrescine and spermidine transport in LeishmaniaMol Biochem Parasitol20001091374610924755
- SinghGDeyCSInduction of apoptosis-like cell death by pentamidine and doxorubicin through differential inhibition of topoisomerase II in arsenite-resistant L. donovaniActa Trop2007103317218517655815
- BasselinMDeniseHCoombsGHBarrettMPResistance to pentamidine in Leishmania mexicana involves exclusion of the drug from the mitochondrionAntimicrob Agents Chemother200246123731373812435669
- WyllieSPattersonSStojanovskiLThe anti-trypanosome drug fexinidazole shows potential for treating visceral leishmaniasisSci Transl Med20124119119re1
- ThompsonAMO’ConnorPDBlaserARepositioning antitubercular 6-Nitro-2,3-dihydroimidazo[2,1-b][1,3]oxazoles for neglected tropical diseases: structure–activity studies on a preclinical candidate for visceral leishmaniasisJ Med Chem20165962530255026901446
- GuptaSYardleyVVishwakarmaPNitroimidazo-oxazole compound DNDI-VL-2098: an orally effective preclinical drug candidate for the treatment of visceral leishmaniasisJ Antimicrob Chemother201570251852725389223
- LuYAimettiAALangerRGuZBioresponsive materialsNat Rev Mater2016116075
- JainKJainNKNovel therapeutic strategies for treatment of visceral leishmaniasisDrug Discov Today20131823–241272128123973338
- ItaliaJLYahyaMMSinghDRavi KumarMNVBiodegradable nanoparticles improve oral bioavailability of amphotericin B and show reduced nephrotoxicity compared to intravenous fungizone®Pharm Res20092661324133119214716
- TorradoJJSerranoDRUchegbuIFThe oral delivery of amphotericin BTher Deliv20134191223401912
- TorradoJJEspadaRBallesterosMPTorrado-SantiagoSAmphotericin B formulations and drug targetingJ Pharm Sci20089772405242517893903
- AllenTMCullisPRLiposomal drug delivery systems: from concept to clinical applicationsAdv Drug Deliv Rev2013651364823036225
- LarabiMYardleyVLoiseauPMToxicity and antileishmanial activity of a new stable lipid suspension of amphotericin BAntimicrob Agents Chemother200347123774377914638481
- MishraJDeyASinghNSomvanshiRSinghSEvaluation of toxicity and therapeutic efficacy of a new liposomal formulation of amphotericin B in a mouse modelIndian J Med Res2013137476777623703346
- AsadMBhattacharyaPBanerjeeAAliNTherapeutic and immunomodulatory activities of short-course treatment of murine visceral leishmaniasis with KALSOME™10, a new liposomal amphotericin BBMC Infect Dis201515118825884796
- ShadabMJhaBAsadMDeepthiMKamranMAliNApoptosis-like cell death in Leishmania donovani treated with KalsomeTM10, a new liposomal amphotericin BPLoS One2017122e017130628170432
- NewRRCChanceMLThomasSCPetersWAntileishmanial activity of antimonials entrapped in liposomesNature197827256485556203863
- CarterKCDolanTFAlexanderJBaillieAJMcColganCVisceral leishmaniasis: drug carrier system characteristics and the ability to clear parasites from the liver, spleen and bone marrow in Leishmania donovani infected BALB/c miceJ Pharm Pharmacol198941287912568434
- RoychoudhuryJSinhaRAliNTherapy with sodium stibogluconate in stearylamine-bearing liposomes confers cure against SSG-resistant Leishmania donovani in BALB/c micePLoS One201163e1737621423750
- ImmordinoMLDosioFCattelLStealth liposomes: review of the basic science, rationale, and clinical applications, existing and potentialInt J Nanomedicine20061329731517717971
- AzevedoEGRibeiroRRda SilvaSMMixed formulation of conventional and pegylated liposomes as a novel drug delivery strategy for improved treatment of visceral leishmaniasisExpert Opin Drug Deliv201411101551156024962630
- ProulxM-EDésormeauxAMarquisJ-FOlivierMBergeronMGTreatment of visceral leishmaniasis with sterically stabilized liposomes containing camptothecinAntimicrob Agents Chemother20014592623262711502539
- KeighobadiMFakharMEmamiSHypothesis: the potential application of doxorubicin against cutaneous leishmaniasisTrop Parasitol201551697025709959
- AlvingCRDelivery of liposome-encapsulated drugs to macrophagesPharmacol Ther19832234074246361805
- JavedIHussainSZUllahISynthesis, characterization and evaluation of lecithin-based nanocarriers for the enhanced pharmacological and oral pharmacokinetic profile of amphotericin BJ Mater Chem B201534283598365
- SapraPAllenTMLigand-targeted liposomal anticancer drugsProg Lipid Res200342543946212814645
- AgrawalAKAgrawalAPalAGuruPYGuptaCMSuperior chemotherapeutic efficacy of Amphotericin B in tuftsin-bearing liposomes against Leishmania donovani infection in hamstersJ Drug Target2002101414511996085
- Vázquez-MendozaACarreroJCRodriguez-SosaMParasitic infections: a role for C-type lectins receptorsBiomed Res Int2013201345635223509724
- RathoreAJainAGulbakeAMannosylated liposomes bearing amphotericin B for effective management of visceral leishmaniasisJ Liposome Res201121433334021612342
- GuptaAEralHBHattonTADoylePSNanoemulsions: formation, properties and applicationsSoft Matter201612112826284126924445
- WasanEKGershkovichPZhaoJA novel tropically stable oral amphotericin B formulation (iCo-010) exhibits efficacy against visceral leishmaniasis in a murine modelPLoS Negl Trop Dis201041216
- HnikPinventoriCo Therapeutics Inc., CanLaherty, Carol D, assigneeStable formulations for the oral administration of amphotericin BUS patentWO2016112339A120162016
- KansalSTandonRDwivediPDevelopment of nanocapsules bearing doxorubicin for macrophage targeting through the phosphatidylserine ligand: a system for intervention in visceral leishmaniasisJ Antimicrob Chemother201267112650266022872448
- ShingithaKPA review: niosomes a novel tool for drug deliveryInt J Pharm Technol20156330173026
- BaillieAJCoombsGHDolanTFLaurieJNon-ionic surfactant vesicles, niosomes, as a delivery system for the anti-leishmanial drug, sodium stibogluconateJ Pharm Pharmacol19863875025052875149
- KhazaeliPSharifiITalebianEHeraviGMoazeniEMostafaviMAnti-leishmanial effect of itraconazole niosome on in vitro susceptibility of Leishmania tropicaEnviron Toxicol Pharmacol201438120521124956400
- SalernoCChiappettaDAMonteagudoEBregniCInfluence of surfactant structure in the encapsulation and stability of amphotericin B in niosomesLatin Am J Pharm201130917281736
- ZarifLDrug delivery by lipid cochleatesMethods Enzymol200539131432915721389
- SesanaAMMonti-RochaRVinhasSAMoraisCGDietzeRLemosEMIn vitro activity of amphotericin B cochleates against Leishmania chagasiMem Inst Oswaldo Cruz2011106225125321537689
- ManninoRJinventorBiodelivery Sciences International, Inc., USA, assigneePharmaceutical cochleate compositions comprising amphotericin B for treating leishmaniasisUS patentWO2010091090A120102010
- PhamTTBarrattGMichelJPLoiseauPMSaint-Pierre-ChazaletMInteractions of antileishmanial drugs with monolayers of lipids used in the development of amphotericin B-miltefosine-loaded nanocochleatesColloids Surf B Biointerfaces201310622423323434716
- BattagliaLGallarateMLipid nanoparticles: state of the art, new preparation methods and challenges in drug deliveryExpert Opin Drug Deliv20129549750822439808
- JainVGuptaAPawarVKChitosan-assisted immunotherapy for intervention of experimental leishmaniasis via amphotericin B-loaded solid lipid nanoparticlesAppl Biochem Biotechnol201417441309133025106894
- Heidari-KharajiMTaheriTDoroudDHabibzadehSRafatiSSolid lipid nanoparticle loaded with paromomycin: in vivo efficacy against Leishmania tropica infection in BALB/c mice modelAppl Microbiol Biotechnol2016100167051706026960322
- Heidari-KharajiMTaheriTDoroudDHabibzadehSBadirzadehARafatiSEnhanced paromomycin efficacy by solid lipid nanoparticle formulation against Leishmania in mice modelParasite Immunol2016381059960827213964
- VeerareddyPRVobalaboinaVAliNAntileishmanial activity, pharmacokinetics and tissue distribution studies of mannose-grafted amphotericin B lipid nanospheresJ Drug Target200917214014719089691
- ChaudhariMBDesaiPPPatelPAPatravaleVBSolid lipid nanoparticles of amphotericin B (AmbiOnp): in vitro and in vivo assessment towards safe and effective oral treatment moduleDrug Deliv Transl Res20166435436426712123
- da Gama BitencourtJJPazinWMItoASMiltefosine-loaded lipid nanoparticles: improving miltefosine stability and reducing its hemolytic potential toward erythtocytes and its cytotoxic effect on macrophagesBiophys Chem2016217203127497059
- ElsabahyMWooleyKLDesign of polymeric nanoparticles for biomedical delivery applicationsChem Soc Rev20124172545256122334259
- LiuZJiaoYWangYZhouCZhangZPolysaccharides-based nanoparticles as drug delivery systemsAdv Drug Deliv Rev200860151650166218848591
- CouvreurPPuisieuxFNano- and microparticles for the delivery of polypeptides and proteinsAdv Drug Deliv Rev1993102141162
- MakadiaHKSiegelSJPoly lactic-co-glycolic acid (PLGA) as biodegradable controlled drug delivery carrierPolymers (Basel)2011331377139722577513
- Van De VenHPaulussenCFeijensPBPLGA nanoparticles and nanosuspensions with amphotericin B: potent in vitro and in vivo alternatives to Fungizone and AmBisomeJ Control Release2012161379580322641062
- ZhangZTanSFengS-SVitamin E TPGS as a molecular biomaterial for drug deliveryBiomaterials201233194889490622498300
- ItaliaJLRavi KumarMNVCarterKCEvaluating the potential of polyester nanoparticles for per oral delivery of amphotericin B in treating visceral leishmaniasisJ Biomed Nanotechnol20128469570222852479
- AlsaadiMItaliaJLMullenABThe efficacy of aerosol treatment with non-ionic surfactant vesicles containing amphotericin B in rodent models of leishmaniasis and pulmonary aspergillosis infectionJ Control Release2012160368569122516093
- KumarRSahooGCPandeyKDevelopment of PLGA-PEG encapsulated miltefosine based drug delivery system against visceral leishmaniasisMater Sci Eng C Mater Biol Appl20165974875326652429
- AsthanaSJaiswalAKGuptaPKDubeAChourasiaMKTh-1 biased immunomodulation and synergistic antileishmanial activity of stable cationic lipid–polymer hybrid nanoparticle: biodistribution and toxicity assessment of encapsulated amphotericin BEur J Pharm Biopharm201589627325477079
- NaharMJainNKPreparation, characterization and evaluation of targeting potential of amphotericin b-loaded engineered PLGA nanoparticlesPharm Res200926122588259819842021
- BudhianASiegelSJWineyKIHaloperidol-loaded PLGA nanoparticles: systematic study of particle size and drug contentInt J Pharm2007336236737517207944
- BarrosDLimaSACCordeiro-Da-SilvaASurface functionalization of polymeric nanospheres modulates macrophage activation: relevance in leishmaniasis therapyNanomedicine201510338740325707974
- SinghPKSahPMeherJGMacrophage-targeted chitosan anchored PLGA nanoparticles bearing doxorubicin and amphotericin B against visceral leishmaniasisRSC Adv20166757170571718
- AsthanaSGuptaPKJaiswalAKDubeAChourasiaMKTargeted chemotherapy of visceral leishmaniasis by lactoferrin-appended amphotericin B-loaded nanoreservoir: in vitro and in vivo studiesNanomedicine20151071093110925929567
- SilvaTAbengózarMAFernandez-ReyesMEnhanced leishmanicidal activity of cryptopeptide chimeras from the active N1 domain of bovine lactoferrinAmino Acids20124362265227722543751
- KumarRSahooGCPandeyKDasVDasPStudy the effects of PLGA-PEG encapsulated Amphotericin B nanoparticle drug delivery system against Leishmania donovaniDrug Deliv201522338338824601828
- PushpamalarJVeeramachineniAKOwhCLohXJBiodegradable polysaccharides for controlled drug deliveryChemPlusChem2016816504514
- SharmaGKarSBasu BallWGhoshKDasPKThe curative effect of fucoidan on visceral leishmaniasis is mediated by activation of MAP kinases through specific protein kinase C isoformsCell Mol Immunol201411326327424561457
- Lehnhardt PiresCRodriguesSDBristotDEvaluation of macroalgae sulfated polysaccharides on the Leishmania (L.) amazonensis promastigoteMar Drugs201311393494323519148
- Kangussu-MarcolinoMMdo RosárioMMTNosedaMDAcid heteropolysaccharides with potent antileishmanial effectsInt J Biol Macromol20158116517026162246
- MatteCMarquisJFBlanchetteJPeroxovanadium-mediated protection against murine leishmaniasis: role of the modulation of nitric oxideEur J Immunol20003092555256411009089
- AmaralAEDPetkowiczCLOMercêALRLeishmanicidal activity of polysaccharides and their oxovanadium(IV/V) complexesEur J Med Chem20159073274125506811
- MachadoPAMotaVZCavalliACLHigh selective antileishmanial activity of vanadium complex with stilbene derivativeActa Trop201514812012725917716
- SingodiaDKharePDubeATalegaonkarSKharRKMishraPRDevelopment and performance evaluation of alginate-capped amphotericin B lipid nanoconstructs against visceral leishmaniasisJ Biomed Nanotechnol20117112312421485834
- YangDJonesKSEffect of alginate on innate immune activation of macrophagesJ Biomed Mater Res A200990241141818523947
- PorporattoCBiancoIDRieraCMCorreaSGChitosan induces different L-arginine metabolic pathways in resting and inflammatory macrophagesBiochem Biophys Res Commun2003304226627212711309
- AsthanaSJaiswalAKGuptaPKPawarVKDubeAChourasiaMKImmunoadjuvant chemotherapy of visceral leishmaniasis in hamsters using amphotericin B-encapsulated nanoemulsion template-based chitosan nanocapsulesAntimicrob Agents Chemother20135741714172223357762
- AsthanaSGuptaPKJaiswalAKDubeAChourasiaMKOverexpressed macrophage mannose receptor targeted nanocapsules-mediated cargo delivery approach for eradication of resident parasite: in vitro and in vivo studiesPharm Res20153282663267725715698
- RibeiroTGChávez-FumagallMAValadaresDGNovel targeting using nanoparticles: an approach to the development of an effective anti-leishmanial drug-delivery systemInt J Nanomedicine20149187789024627630
- GuptaPKJaiswalAKKumarVCovalent functionalized self-assembled lipo-polymerosome bearing amphotericin B for better management of leishmaniasis and its toxicity evaluationMol Pharm201411395196324495144
- GuptaPKJaiswalAKAsthanaSSelf assembled ionically sodium alginate cross-linked amphotericin B encapsulated glycol chitosan stearate nanoparticles: applicability in better chemotherapy and non-toxic delivery in visceral leishmaniasisPharm Res20153251727174025425053
- KansalSTandonRVermaACoating doxorubicin-loaded nanocapsules with alginate enhances therapeutic efficacy against Leishmania in hamsters by inducing Th1-type immune responsesBr J Pharmacol2014171174038405024837879
- LoiseauPMImbertieLBoriesCBetbederDDe MiguelIDesign and antileishmanial activity of amphotericin B-loaded stable Ionic Amphiphile Biovector formulationsAntimicrob Agents Chemother20024651597160111959611
- MullenABCarterKCBaillieAJComparison of the efficacies of various formulations of amphotericin B against murine visceral leishmaniasisAntimicrob Agents Chemother19974110208920929333030
- KataokaKHaradaANagasakiYBlock copolymer micelles for drug delivery: design, characterization and biological significanceAdv Drug Deliv Rev200147111313111251249
- MendonçaDVCLageLMRLageDPPoloxamer 407 (Pluronic® F127)-based polymeric micelles for amphotericin B: in vitro biological activity, toxicity and in vivo therapeutic efficacy against murine tegumentary leishmaniasisExp Parasitol2016169344227427166
- BruguésAPNaverosBCCalpena CampmanyACPastorPHSaladrigasRFLizandraCRDeveloping cutaneous applications of paromomycin entrapped in stimuli-sensitive block copolymer nanogel dispersionsNanomedicine201510222724025600968
- FernandesFRFerreiraWACamposMAAmphiphilic antimony(V) complexes for oral treatment of visceral leishmaniasisAntimicrob Agents Chemother20135794229423623796930
- RibeiroRRFerreiraWAMartinsPSProlonged absorption of antimony(V) by the oral route from non-inclusion meglumine antimoniate-β-cyclodextrin conjugatesBiopharm Drug Dispos2010312–310911920014042
- Ali-BoucettaHKostarelosKCarbon nanotubes in medicine & biology – therapy and diagnosticsAdv Drug Deliv Rev201365151897189824220279
- BaezaAColillaMVallet-RegíMAdvances in mesoporous silica nanoparticles for targeted stimuli-responsive drug deliveryExpert Opin Drug Deliv201512231933725421898
- AllahverdiyevAMAbamorESBagirovaMAntileishmanial effect of silver nanoparticles and their enhanced antiparasitic activity under ultraviolet lightInt J Nanomedicine201162705271422114501
- JebaliAKazemiBNano-based antileishmanial agents: a toxicological study on nanoparticles for future treatment of cutaneous leishmaniasisToxicol In Vitro20132761896190423806227
- SoflaeiSDalimiAAbdoliAAnti-leishmanial activities of selenium nanoparticles and selenium dioxide on Leishmania infantumComp Clin Pathol20142311520
- NadhmanANazirSKhanMIVisible-light-responsive ZnCuO nanoparticles: benign photodynamic killers of infectious protozoansInt J Nanomedicine2015106891690326604755
- NadhmanANazirSIhsanullah KhanMPEGylated silver doped zinc oxide nanoparticles as novel photosensitizers for photodynamic therapy against LeishmaniaFree Radic Biol Med20147723023825266330
- AhmadAWeiYSyedFIsatis tinctoria mediated synthesis of amphotericin B-bound silver nanoparticles with enhanced photoinduced antileishmanial activity: a novel green approachJ Photochem Photobiol B2016161172427203567
- PrajapatiVKAwasthiKGautamSTargeted killing of Leishmania donovani in vivo and in vitro with amphotericin β attached to functionalized carbon nanotubesJ Antimicrob Chemother201166487487921393222
- PrajapatiVKAwasthiKYadavTPRaiMSrivastavaONSundarSAn oral formulation of amphotericin B attached to functionalized carbon nanotubes is an effective treatment for experimental visceral leishmaniasisJ Infect Dis2012205233333622158723
- KesterMHeakalYFoxTCalcium phosphate nanocomposite particles for in vitro imaging and encapsulated chemotherapeutic drug delivery to cancer cellsNano Lett20088124116412119367878
- ChenFZhuYWuJHuangPCuiDNanostructured calcium phosphates: preparation and their application in biomedicineNano Biomed Eng2012414149
- ChaurasiaMSinghPKJaiswalAKBioinspired calcium phosphate nanoparticles featuring as efficient carrier and prompter for macrophage intervention in experimental leishmaniasisPharm Res201633112617262927401407
- AlvarengaBMMeloMNFrézardFNanoparticle phosphate-based composites as vehicles for antimony delivery to macrophages: possible use in leishmaniasisJ Mater Chem B201534892509259
- SinghJDesaiSYadavSNarasimhanBKaurHPolymer drug conjugates: recent advancements in various diseasesCurr Pharm Des201622192821284326898741
- DuncanRPolymer therapeutics: top 10 selling pharmaceuticals – what next?J Control Release201419037138024818766
- DuncanRVicentMJDo HPMA copolymer conjugates have a future as clinically useful nanomedicines? A critical overview of current status and future opportunitiesAdv Drug Deliv Rev201062227228220005271
- NicolettiSSeifertKGilbertIHN-(2-hydroxypropyl)methacrylamide-amphotericin B (HPMA-AmB) copolymer conjugates as antileishmanial agentsInt J Antimicrob Agents200933544144819097763
- DosioFArpiccoSStellaBFattalEHyaluronic acid for anticancer drug and nucleic acid deliveryAdv Drug Deliv Rev20169720423626592477
- MicaleNPipernoAMahfoudhNA hyaluronic acid-pentamidine bioconjugate as a macrophage mediated drug targeting delivery system for the treatment of leishmaniasisRSC Adv201551169554595550
- Le PapePDevelopment of new antileishmanial drugs – current knowledge and future prospectsJ Enzyme Inhib Med Chem200823570871818671165
- MishraBBKaleRRSinghRKTiwariVKAlkaloids: future prospective to combat leishmaniasisFitoterapia2009802819019015012
- EspuelasSPlanoDNguewaPInnovative lead compounds and formulation strategies as newer kinetoplastid therapiesCurr Med Chem201219254259428822834813
- PenãSScaroneLSerraGMacrocycles as potential therapeutic agents in neglected diseasesFuture Med Chem20157335538225650721
- Castillo-GaritJAAbadCRodríguez-BorgesJEMarrero-PonceYTorrensFA review of QSAR studies to discover new drug-like compounds actives against leishmaniasis and trypanosomiasisCurr Top Med Chem201212885286522352913
- TariqAAdnanMAmberRPanKMussaratSShinwariZKEthnomedicines and anti-parasitic activities of Pakistani medicinal plants against Plasmodia and Leishmania parasitesAnn Clin Microbiol Antimicrob20161515227647140
- PolonioTEfferthTLeishmaniasis: drug resistance and natural products (review)Int J Mol Med200822327728618698485
- NagleASKhareSKumarABRecent developments in drug discovery for leishmaniasis and human African trypanosomiasisChem Rev201411422113051134725365529
- SangshettiJNKalam KhanFAKulkarniAAAroteRPatilRHAntileishmanial drug discovery: comprehensive review of the last 10 yearsRSC Adv20155413237632415
- MarrAKMcGwireBSMcMasterWRModes of action of leishmanicidal antimicrobial peptidesFuture Microbiol2012791047105922953706
- SundarSChakravartyJInvestigational drugs for visceral leishmaniasisExpert Opin Investig Drugs20152414359
- PenicheAGRensloARMelbyPCTraviBLAntileishmanial activity of disulfiram and thiuram disulfide analogs in an ex vivo model system is selectively enhanced by the addition of divalent metal ionsAntimicrob Agents Chemother201559106463647026239994
- FortinAHendrickxSYardleyVCosPJansenHMaesLEfficacy and tolerability of oleylphosphocholine (OLPC) in a laboratory model of visceral leishmaniasisJ Antimicrob Chemother201267112707271222782488
- HernándezLGálvezRMontoyaAFirst study on efficacy and tolerability of a new alkylphosphocholine molecule (oleylphosphocholine – OlPC) in the treatment of canine leishmaniosis due to Leishmania infantumParasitol Res2014113115716424192865
- FortinADorloTPCHendrickxSMaesLPharmacokinetics and pharmacodynamics of oleylphosphocholine in a hamster model of visceral leishmaniasisJ Antimicrob Chemother20167171892189827084920
- RoyPDasSBeraTMondolSMukherjeeAAndrographolide nanoparticles in leishmaniasis: characterization and in vitro evaluationsInt J Nanomedicine2010511113112121270962
- MondalSRoyPDasSHalderAMukherjeeABeraTIn Vitro susceptibilities of wild and drug resistant Leishmania donovani amastigote stages to andrographolide nanoparticle: role of vitamin e derivative TPGS for nanoparticle efficacyPLoS One2013812e8149224339938
- GhoshSKarNBeraTOleanolic acid loaded poly lactic co-glycolic acid-vitamin E TPGS nanoparticles for the treatment of Leishmania donovani infected visceral leishmaniasisInt J Biol Macromol20169396197027645930
- TavaresJOuaissiASilvaAMLinPKTRoyNCordeiro-da-SilvaAAnti-leishmanial activity of the bisnaphthalimidopropyl derivativesParasitol Int201261236036322155672
- Costa LimaSAResendeMSilvestreRCharacterization and evaluation of BNIPDaoct-loaded PLGA nanoparticles for visceral leishmaniasis: in vitro and in vivo studiesNanomedicine20127121839184922812711
- YangDMLiewFYEffects of qinghaosu (artemisinin) and its derivatives on experimental cutaneous leishmaniasisParasitology199310617118479804
- WantMYIslamuddinMChouhanGDasguptaAKChattopadhyayAPAfrinFA new approach for the delivery of artemisinin: formulation, characterization, and ex-vivo antileishmanial studiesJ Colloid Interface Sci201443225826925086720
- WantMYIslamuddinMChouhanGTherapeutic efficacy of artemisinin-loaded nanoparticles in experimental visceral leishmaniasisColloids Surf B Biointerfaces201513021522125936561
- FouladvandMBarazeshATahmasebiREvaluation of in vitro antileishmanial activity of curcumin and its derivatives “Gallium curcumin, Indium curcumin and Diacethyle curcumin”Eur Rev Med Pharmacol Sci201317243306330824379060
- CouvreurPStellaBHarivardhan ReddyLSqualenoyl nanomedicines as potential therapeuticsNano Lett20066112544254817090088
- MaksimenkoADosioFMouginJA unique squalenoylated and nonpegylateddoxorubicin nanomedicine with systemiclong-circulating properties and anticancer activityProc Natl Acad Sci U S A20141112E217E22624385587
- Cheikh-AliZCaronJCojeanS“Squalenoylcurcumin” nanoassemblies as water-dispersible drug candidates with antileishmanial activityChemMedChem201510241141825523035
- TiwariBPahujaRKumarPRathSKGuptaKCGoyalNNanotized curcumin and miltefosine, a potential combination for treatment of experimental visceral leishmaniasisAntimicrob Agents Chemother2017613e011691628031196
- PaloqueLVerhaeghePCasanovaMDiscovery of a new antileishmanial hit in 8-nitroquinoline seriesEur J Med Chem201254758622608675
- Costa DuarteMdos Reis LageLMLageDPAn effective in vitro and in vivo antileishmanial activity and mechanism of action of 8-hydroxyquinoline against Leishmania species causing visceral and tegumentary leishmaniasisVet Parasitol2016217818826827866
- DuarteMCLageLMDRLageDPTreatment of murine visceral leishmaniasis using an 8-hydroxyquinoline-containing polymeric micelle systemParasitol Int201665672873627425599
- NanACroftSLYardleyVGhandehariHTargetable water-soluble polymer-drug conjugates for the treatment of visceral leishmaniasisJ Control Release200494111512714684276
- SaudagarPDubeyVKCarbon nanotube based betulin formulation shows better efficacy against Leishmania parasiteParasitol Int201463677277625086374
- CrommelinDJAFlorenceATTowards more effective advanced drug delivery systemsInt J Pharm2013454149651123415662
- de MenezesJPGuedesCEPetersenALFragaDBVerasPSAdvances in development of new treatment for leishmaniasisBiomed Res Int2015201581502326078965
- ZhangW-WLypaczewskiPMatlashewskiGOptimized CRISPR-Cas9 genome editing for Leishmania and its use to target a multigene family, induce chromosomal translocation, and study DNA break repair mechanismsmSphere201721e003401628124028
- AntillónADe VriesAHEspinosa-CaballeroMAn amphotericin B derivative equally potent to amphotericin B and with increased safetyPLoS One2016119e016217127683101
- OlliaroPLShamsuzzamanTAMarasiniBInvestments in research and surveillance are needed to go beyond elimination and stop transmission of Leishmania in the Indian subcontinentPLoS Negl Trop Dis2017111e000519028125596
- HiemenzJWWalshTJLipid formulations of amphotericin B: recent progress and future directionsClin Infect Dis199622suppl 2S133S1448722841
- ThakurCPSinghRKHassanSMKumarRNarainSKumarAAmphotericin B deoxycholate treatment of visceral leishmaniasis with newer modes of administration and precautions: a study of 938 casesTrans R Soc Trop Med Hyg199993331932310492770
- Sánchez-BruneteJADeaMARamaSTreatment of experimental visceral leishmaniasis with amphotericin B in stable albumin microspheresAntimicrob Agents Chemother20044893246325215328080
- Sánchez-BruneteJADeaMARamaSInfluence of the vehicle on the properties and efficacy of microparticles containing amphotericin BJ Drug Target200513422523316051534
- World Health OrganizationNeglected tropical diseases: WHO and Gilead Sciences extend collaboration against visceral leishma-niasis Available from: http://www.who.int/neglected_diseases/news/WHO_and_Gilead_Sciences_extend_collaboration/en/
- Drugs for Neglected Diseases InitiativeFexinidazole/Miltefosine combination (VL) Available from: https://www.dndi.org/diseases-projects/portfolio/completed-projects/fexinidazole-vl/
- iCo Therapeutics IncPipeline Available from: http://www.icothera-peutics.com/pipeline/
- Matinas BiopharmaLearn more about cochleate technology Available from: http://www.matinasbiopharma.com
- American Pharmaceutical ReviewFDA’s regulatory science program for generic PLA/PLGA-based drug products Available from: http://www.americanpharmaceuticalreview.com/Featured-Articles/188841-FDA-s-Regulatory-Science-Program-for-Generic-PLA-PLGA-Based-Drug-Products/

