Abstract
Over the past few years, nanoparticles and their role in drug delivery have been the centre of attraction as new drug delivery systems. Various forms of nanosystems have been designed, such as nanoclays, scaffolds and nanotubes, having numerous applications in areas such as drug loading, target cell uptake, bioassay and imaging. The present study discusses various types of nanoparticles, with special emphasis on ceramic nanocarriers. Ceramic materials have high mechanical strength, good body response and low or non-existing biodegradability. In this article, the various aspects concerning ceramic nanoparticles, such as their advantages over other systems, their cellular uptake and toxicity concerns are discussed in detail.
Introduction
The latest innovation of this decade in the field of science, involving the medical, technological and pharmaceutical fields, is the development of nanotechnology. Nanotechnology is a rapidly expanding field, encompassing the development of man-made materials in the 5–200 nm size range. Nanotechnology specifies a system whose structures and components impart novelty and significant properties at the nanoscale, specifically, < 100 nm/< 10–7 m. Indeed, their various advantages including size, structural advantages, highly active surfaces, unique physical and chemical properties and ease of modification serve as excellent platforms for drug transportation and controlled release (CitationRawat et al. 2006, CitationSingh et al. 2013). The application of nanotechnology to medicine has created an interdisciplinary research field, often referred to as nanomedicine, which has the potential to significantly treat many diseases (CitationFerrari 2005). The application of nanomaterials to medical problems has already demonstrated a clinical impact in terms of delivery strategies for a range of bioactive molecules, including therapeutic agents, nucleic acids and imaging contrast agents (CitationSakamoto et al. 2010).
There are already examples of nanomedicine in clinical use. Doxil®, a PEGylated liposomal doxorubicin formulation, was the first nanosized therapeutic in the market in 1995 and was used as an effective treatment for metastatic breast cancer and recurrent ovarian cancer (CitationBarenholz 2012). Numerous other systems are in various stages of preclinical and clinical advancement. In recent years, a targeted therapeutic nanoparticle, named BIND-014, which accumulates in tumours while avoiding uptake by the healthy cells, has shown promising results in an ongoing clinical trial (CitationHrkach et al. 2012). Another example is a lipid nanoparticulate delivery system (Oncoprex®) containing plasmid DNA encoding the TUSC2 tumour suppressor that is being studied in combination with erlotinib, a human epidermal growth factor receptor (EGFR) inhibitor, in lung cancer patients who were otherwise unresponsive to erlotinib or lacking the EGFR mutation (CitationZhang et al. 2012).
Nanotechnology: A technological boom in drug delivery
A drug delivery system must positively impact the rate of absorption, distribution, metabolism and excretion of the drug or other substances in the body. The drug delivery material must be compatible and bind easily with the drug, and be bioresorbable (i.e. degrade into fragments after use, which are either metabolized or eliminated via normal excretory routes).
Nanotechnology provides a wide range of new technologies for developing customized solutions that optimize the delivery of pharmaceutical products and has brought significant advancement in the diagnosis and treatment of disease (CitationSanvicens & Marco 2008). The medical applications include drug delivery, both in in vitro/in vivo diagnostics, improved biocompatible materials and nutraceuticals (CitationDuncan 2003, CitationWu et al. 2014). The major areas of focus associated with smart delivery systems are drug targeting, maintaining therapeutic efficacy and development of full-fledged safe medications. Furthermore, drugs need to be protected during their transit to the target action site in the body while maintaining their biological and chemical properties, to be therapeutically effective. The transit time and delivery challenges can be greatly different depending on the location of absorption of drug, like the colon or small intestine, and whether certain natural defense mechanisms need to be passed through such as the blood-brain barrier (CitationLaVan et al. 2003).
The basic pre-requisites for drug design of new materials comprise knowledge on drug incorporation and release, formulation stability and shelf life, biocompatibility, biodistribution, targeting, functionality and possible adverse effects. The miniature size of nanoparticles promotes penetration through cell membranes, stabilization of proteins, and lysosomal escape after endocytosis.
Types of nanocarriers
With the recent advancement in nanocarriers, various types of nanocarriers viz. ceramic, polymeric, liposomal, solid lipid, dendrimers, nanotubes, nanocrystals and nanobots are available. Polymeric nanoparticles are biodegradable and biocompatible, and are made from gelatin, chitosan, PLA, PLGA etc. Ceramic nanoparticles are typically composed of inorganic compounds such as silica and alumina. The solid lipid nanoparticle is yet another type, having a solid lipid core with controllable pharmacokinetic parameters and ease of biodegradation. The detailed description is listed in .
Table I. Classification of nanoparticles with specific characteristics.
Advantages of nanocarriers
Particulate drug carriers possess various advantages for use in drug delivery and are probably the most common ceramic drug delivery platforms today (CitationQuan et al. 2009, CitationBoris et al. 2013).
The advantages are as follows:
Particulate carriers could easily penetrate inside cells with large surface area: volume or surface area: mass ratio that allows high payload and a prolonged drug release profile.
They are cheap to manufacture especially in mass production. Advancement in nanotechnology has strengthened these nanoparticles to be of high purity and high surface area-to-volume ratios as well as developed affordable fabrication processes with a high control of particle size, morphology or porosity. Li et al. developed a nanofibrous system having ultrafine morphology of polyvinyl alcohol as a filament forming polymer for the delivery of caffeine and riboflavin; 100% release of caffeine and more than 40% of riboflavin has been found (CitationLi et al. 2013).
Nanoparticles are highly advantageous because they facilitate deeper penetration into capillaries and through fenestrations to enhanced cellular uptake. These systems have been also widely used for topical delivery of drug for treatment of various topical issues. Paveer et al. reported effective wound and burn treatment by a polymeric nanofibrous system prepared by electrospinning of soluplus (polyvinyl caprolactam polyvinyl acetate polyethylene glycol) polymer. These systems possess an ultra-fine structure, large surface are to volume ratio and high porosity with small pore size which makes them more effective for topical application of abrasion treatment (CitationPaveer et al. 2014).
High surface area-to-volume ratios of nanoparticles and surface activity aid in adsorption of high amount of drugs in them. This implies better drug control and sustained release.
Moreover, novel approaches lead to precise, targetable drug release patterns. For example, Quan et al. demonstrated a thermosensitive nanogel with the ability to target tumours. The poly (N-isopropylacrylamide- co-propyl acrylic acid) nanogel, conjugated with an arginine-glycine-aspartic acid (RGD) containing peptide and transferrin, has a dual ability: it can target tumour cells and release embedded doxorubicin–a DNA interacting anticancer drug, in response to temperature changes above 34.4°C.
Ceramic nanoparticles
There are various types of nanocarriers which have shown promising output in drug delivery such as polymeric nanoparticles, nanotubes, fullerenes, dendrimers, micelles etc (CitationShi et al. 2013). Among these, a newer class of nanoparticles have evolved- ceramic nanoparticles which are highly resistant to environmental changes. Ceramics can be described as having a definite solid core, structured by application of heat or both heat and pressure, comprising a combination of metal and nonmetal – for instance, at least one metal and a non-metallic elemental solid or a non-metal, a combination of at least two non-metallic elemental solids or a combination of at least two non-metallic elemental solids and a non-metal (CitationWu et al. 2010). Ceramic nanoparticles are basically comprised of inorganic compounds such as silica or alumina. However, the nanoparticle core is not limited to just these two materials; rather, metals, metal oxides (CitationArmatas and Kanatzidis 2006, CitationZou et al. 2005) and metal sulphides can be used to produce nanosystems of varying size, shape, and porosity. Mostly, inorganic nanoparticles are designed to evade the reticulo-endothelial system by altering size and surface composition. The porous nature of nanoparticles gives them physical protection from degradation and degranulation. Hollow silica nanoparticles have been reported with surface pores and calcium phosphate-based nanoshells leading to a central reservoir. Ceramics comprised of calcium phosphates, silica, alumina, zirconium, iron oxides, carbonates and titanium dioxide are nowadays used for various medical applications due to their positive interactions with human tissues. For instance, in the field of dentistry, where calcium phosphate and calcium hydroxide-based materials are used as endodontic filling materials and metal–ceramic alloys are applied for crowns (CitationWhitters et al. 1999). Also in the field of orthopaedics (invertebral disks, joints) and plastic surgery (cranial defects) ceramics are frequently employed. Characteristics of these ceramic materials are often high mechanical strength, good body-response and low or non-existing biodegradability (CitationBlock and Thorn 2000, CitationGladstone et al. 1995). The basic concept behind the preparation of a nanosystem is discussed in .
Table II. Methodology for nanoceramic preparation.
Classification of nanophase ceramics
Nanophase ceramics can be classified on the basis of their architectural differences into three general categories: nanoparticles, nano scaffolds and nanoclays ().
Figure 1. Classification of ceramic nanoparticulate systems on the basis of their composition and construct.
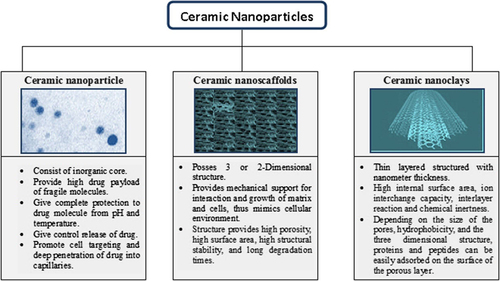
Ceramic nanoparticles
These are the nanoparticles made up of inorganic (ceramic) compounds such as silica, titania and alumina (CitationRawat et al. 2008). These particles provide the complete protection to the entrapped molecules such as proteins, enzymes and drugs against the denaturing effects of external pH and temperature, as they involve no swelling and porosity changes with the change in pH (CitationSingh et al. 2013). The efficient sensitivity and high selectivity for donating and detecting a nitric oxide system by hybrid nanospheres with cadmium-selenide quantum dots has been reported (CitationLiu 2014).
Ceramic nano-scaffold
A scaffold is defined as a structure that allows cells and extracellular matrices to interact, and provide the mechanical support for growing cells and tissues. A scaffold can have two types of porosity: macroporosity (pore size > 50 nm) and microporosity (pore size < 10 nm).The ceramic nano-scaffolds are advantageous in term of high porosity, high surface area, high structural stability and long degradation times. These properties suit them for the storage and controlled release of drug, thus solving the in-situ purpose (CitationSingh et al. 2013). Lo et al. reported the success of nano-scaffold dendrimers as a potential system for theranostics of prostate cancer; it employed an imaging agent for the diagnostic purpose and a therapeutic agent for the target treatment of prostate cancer. Similarly, many more scaffold systems have been designed (CitationLo et al. 2013).
Nanoclay
Nanoclay architecture resembles thin layers with each layer having a thickness of a few nanometers and a length from a few hundred to a few thousand nanometers. There has been an increased surge of interest for clay minerals in pharmaceutical applications due to their high adsorption ability, high internal surface area, high cation exchange capacity, interlayer reaction, chemical inertness, and low or null toxicity. Some of the widely used nanoclays are based on porous silica, zeolites, halloysite nanotubes, montmorillonite, titanium dioxide, etc. (Yuri et al. 2013). CitationKevadiya and Bajaj (2013) reported controlled delivery of encapsulated drug from layered nanostructures and material at high concentration to the target site by crossing the cell membrane in a specific period of time.
Mechanism of cellular uptake
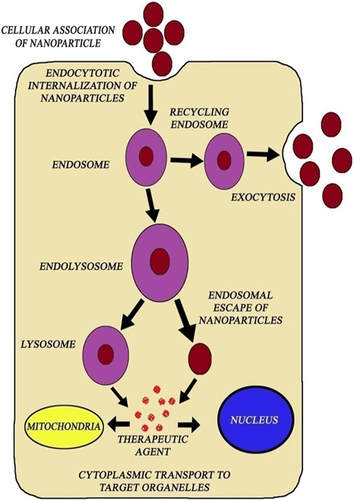
For effective drug delivery, only organ or cell targeting is not sufficient, as the fate of the nanoparticles within the cells is also important. Intracellularly, particles are engulfed by endosomes or lysosomes, followed by degradation. For activity to happen, release of drug into the cytosol is needed. Surface modifications of nanoceramics serves possibilities of applications like drug targeting in terms of cellular binding, uptake and intracellular transport. The mechanism of cellular uptake has been shown in .
Figure 2. Cytosolic delivery of therapeutic agents through nanoparticle carriers: The uptake of nanoparticles is mediated by clathrin endocytosis, by the formation of endosomes. In the presence of organelle lysosomes, endosomes get degraded and nanoparticles freely release the drug into cytoplasm at the targeted site.
Nanoparticle uptake by tissues
Nanoparticles act just like other cell structures or antigens. Nanoparticles face an obstacle due to several successive membrane layers, in attempting to target intracellular structures. During this process, the compound is lost due to ineffective partitioning across biological membranes. The extent of partition across a membrane is related directly to the polarity of a molecule; nonpolar or lipophilic molecules easily bypass this obstacle with greater membrane penetration, generally via diffusion. Endocytosis is the process by which cells envelop and absorb materials, and involves three subtypes: phagocytosis, pinocytosis, and receptor-mediated endocytosis. Phagocytosis involves the ingestion of materials up to 10 μm in diameter and can be accomplished by few cell types of the reticuloendothelial system, such as macrophages, neutrophils, and dendritic cells (CitationKohane 2007). Larger microparticles provide selective access to phagocytic cells, while smaller nanoparticles provide access to virtually all cell types. Pinocytosis is an uptake mechanism that can be conducted by virtually all cell types, and normally involves ingestion of sub-micron material and substances in solution.
Cellular phagocytosis/endocytosis
Receptor-mediated endocytosis affords the potential for even greater selectivity in cellular targeting. The cell membrane consists of various receptors that are specific in their functions, and binding to receptors generates signals. This signal can trigger a multitude of biochemical pathways; however it may also cause internalization of the ligand and its appended nanoparticle via endocytosis. Typically, clathrin coats generate a membrane indentation with a radius of curvature as small as approximately 50 nm, and invaginate further upon binding of the ligand. Cross-linking of receptors via ligands attached to nanoparticles results in a more pronounced membrane crater with subsequent enfolding and reunification of the cellular membrane to form an endosome (CitationGao et al. 2005) It has been shown that nanoparticles sized between 25 and 50 nm are a requisite for optimal endocytosis and intracellular localization (CitationChithrani et al. 2006).
Interaction of nanomaterials with a biological system
There is a complex relationship between the physicochemical properties of nanomaterials (e.g., size, charge, surface properties) and their interactions with a biological system. Minute variance related to size, surface modification, charge and chemical composition can lead to radically active interactions with living systems (CitationHarper et al. 2008). The active interactions of nanomaterials thus affect the biocompatibility, stability, biological performance and side effects of the nanomaterial. Major aspects of the interactions between nanomaterials and proteins are generally protein-binding, ligand-mediated interactions, and interactions during intracellular processing (CitationRoy et al. 2014).
Binding of nanocarriers with protein components
Whenever a drug is taken in any form, the first interaction occurs with the blood or systemic circulation before it reaches its target site. Blood contains various proteins which interact with the drug carrier's protein or other moiety, forming new complexes. The protein that binds significantly to particulates like liposomes, polymeric nanoparticles, iron oxide nanoparticles and carbon nanotubes are basically albumin, immunoglobulins, fibrinogen, apolipoproteins and proteins (CitationNel et al. 2009, CitationZhou et al. 2014).
Ligand- directed nanocarrier-receptor interaction
Nanomaterials are designed specifically to recognize a target with a surface ligand. The ligands used can include antibodies, engineered antibody fragments, proteins, peptides, small molecules, and aptamers (CitationPeer et al. 2007).
The surface modification could be utilized to make the drug concentration appropriate in the target area or to detect a biomarker for diagnostic purposes. The presence of a ligand at the target site of nanoparticles initializes receptor-mediated endocytosis by cells expressing the right target on their membrane, leading to targeted delivery (CitationFarokhzad and Langer 2009).
Cellular internalization, further processing and interaction
Once the nanoparticles are engulfed, they are entrapped and transported by vesicles. Intracellular trafficking and the fate of nanomaterials are linked to their physicochemical properties and endocytic pathways (CitationMiller et al. 2009, CitationUlbrich and Subr 2004). For example, nanoparticles taken up by clathrin-dependent receptor mediated endocytosis (RME) are typically destined for lysosomal degradation; whereas clathrin-independent RME internalization leads to endosomal accumulation and sorting to a non-degradative path. While some drug delivery systems aim to avoid lysosomal degradation, recent studies have utilized delivery to this environment for the enzymatic release of therapeutics (CitationDuncan 2006). Appropriate design and engineering of nanocarriers could therefore allow for controlled intracellular delivery of therapeutics to individual intracellular compartments, which provides benefits to therapies associated with these unique organelles, including cancer therapy, gene therapy, and lysosomal storage disease (LSD) treatments.
Intracellular drug release
The nanosized drug delivery systems are based on the covalent conjugation of chemotherapeutics to hydrophilic polymers, which markedly improves solubility as well as alters drug biodistribution and pharmacokinetics. Conjugates have longer half-life (typically > 1 h) than free drug when circulating in the blood, leading to significantly increased drug concentrations in tumors (CitationSinger et al. 2005). A wide range of delivery systems have been developed, for example dendrimers, liposomes, cationic lipid compounds, cyclodextrin and others, to facilitate endosomal escape and ensure cytosolic delivery of bioactives. Nano ceramic constructs can be further engineered with specific ligands for the targeting of therapeutic agents to specific organelles. Muro et al. demonstrated that the specific delivery of recombinant ASM to lysosomes, by nanocarriers coated with an antibody against the intercellular adhesion molecule-1 (ICAM-1), could improve the efficacy of enzyme replacement therapy (CitationMuro et al. 2006, CitationJinjin et al. 2013).
Toxicity concerns
Nanoparticles are associated with enormous advantages which overwhelm their disadvantages. The most concerning factor preventing their universal use is the toxicity concerns due to the health hazard potential of nanomaterials and the consequent hurdles for regulatory approval and commercialization of nanomedical products.
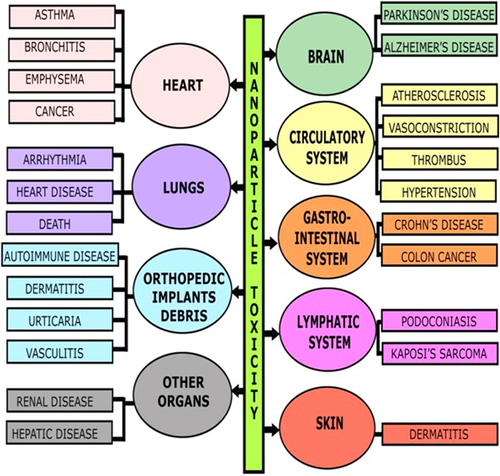
The properties of nanomaterials, like their miniature size, increased reactivity, and high surface-to-volume ratio, are likely to provide health benefits, along with associated hazardous effects on cells and tissues (CitationMarchant 2009). These hazardous effects of nanomaterials result due to the size of the nanoparticles being similar to organelles found in the cell, thus causing interference in vital cellular functions, resulting in potential toxicity (CitationShvedova et al. 2010). Some researchers have even revealed the fact that most of the nanoparticles cause oxidative stress and inflammation by the RES (reticuloendothelial system). The toxic effect of ceramic nanoparticles varies from tissue to cell. The effects on inflammatory and immunological systems may include oxidative stress/cytotoxic activity in the lungs, liver, heart, and brain. The effects can also include prethrombosis in heart function, genotoxicity, carcinogenicity, and teratogenicity. Many a times it has been reported that nanoparticles pass the blood–brain barrier and cause brain toxicity. The toxic effects of nanoparticles on various organs are shown in (Muhlfeld et al. 2007, CitationYacobi et al. 2007).
Important factors affecting toxicity of nanoparticles
There are various factors that are crucial determinants of the toxicity of nanoparticles. These factors are discussed below.
Size
Toxicity effects are highly associated with two basic factors, namely the size and chemical components. The particle size plays a more crucial role than its chemical properties (CitationFubini 1997). The size of any particle is crucial, as the smaller the size, the greater will be its surface area available to adsorb chemical molecules on its surface, which enhances its interaction and results in increased toxic effects (CitationLinkov et al. 2008). After absorption, nanoparticles reach the blood stream and then spread through the tissue. Hyuk et al. reported that 33% (50 nm), 26% (100 nm), and 10% (500 nm) particles were discovered in mucosal and lymphatic tissues of the intestine (CitationSuh et al. 2009). Nanoparticles larger than 1 μm were weakly observed and nanoparticles larger than 3 μm were occasionally seen in lymphatic tissues. Researchers have concluded that nanoparticles in the range of 100–300 nm are absorbed by intestinal cells whereas nanoparticles of 100 nm are absorbed greatly in the lymphatic tissue, more than in intestinal cells. Intestinal cells cannot absorb nanoparticles larger than 400 nm and nanoparticles smaller than 500 nm can enter the circulatory system. In vitro studies have shown that very small particles demonstrate more pathological and destructive potential over the lungs rather than the particles of smaller size, due to the larger surface area (CitationOberdorster et al. 2010).
Surface chemistry
The various surface related factors are important in toxicity studies. The degree of hydrophobicity and hydrophilicity of a nanocarrier surface is the major feature used to estimate the toxicity. The absorption of nanoparticles produced by hydrophobic polymers is greater than that of nanoparticles produced by hydrophilic polymers. This absorption difference can alter their concentration in the cell and thus toxicity due to overdose can occur. In addition, the presence of a reactive group on the surface modifies their biological effects. For example, it has been shown that surface modification of quartz affects its cytotoxicity, inflammogenicity, and fibrogenicity. These differences are mainly due to particle surface characteristics (CitationSchins et al. 2002). The toxicity of silica is due to its interaction with ROS, causing cancer in the lungs.
Chemical components
Another important factor is the chemical component present on the particle's surface. It can react with metals like iron. Iron can be affected by nanoparticles, which increases the induction of ROS in the free cell system. Researchers have also shown that the toxicity of super paramagnetic iron oxide nanoparticles could be reduced by coating them with pullulan (CitationClift et al. 2008). This implies that surface modification can alter structure toxicity.
Dosage
This factor is well known in the conventional system of drug delivery, and has similar effects in the case of nanoparticles. Overdose of nanoparticles is a serious threat to the human body. Research has shown that a high dose of nanoparticles, whether they are small or big particles, could be harmful to health (CitationSingh et al. 2007).
Free radical production
Most or all pathogenic particles produce free radicals in the free cell system and this ability causes oxidative stress, which gives rise to inflammation, cell destruction, and genotoxicity. The particle surface of free radicals can activate the redox cycle and cause particle toxicity (CitationHussain et al. 2009).
Cytotoxicity
Ceramic nanoparticles are able to enter cells due to their small size. Cellular uptake, subcellular localization, and the ability to catalyze oxidative products, depend on the nanoparticle's chemistry, size, and shape (CitationXia et al. 2006). The mechanism by which nanoparticles penetrate cells without specific receptors on their outer surface is assumed to be by passive uptake or adhesive interaction. This uptake may be initiated by Van der Waals forces, electrostatic charges, steric interactions, or interfacial tension effects, and does not result in the formation of vesicles (CitationGeiser et al. 2005). After this type of uptake, the nanoparticles are not necessarily located within a phagosome (which offers some protection to the rest of the cellular organelles from the chemical interaction with the nanoparticle). For example, C60 molecules enter cells and can be found along the nuclear membrane and within the nucleus (CitationPorter et al. 2006). This type of uptake and free movement within the cell makes them very dangerous by giving them direct access to cytoplasmic proteins and organelles. Upon non-phagocytic uptake, nanoparticles can be found in various locations inside cell, such as the outer-cell membrane cytoplasm, mitochondria, lipid vesicles along the nuclear membrane or within the nucleus. Depending on their localization inside the cell, the nanoparticles can damage organelles or DNA, or cause ultimately cell death (CitationStefani et al. 2005).
Conclusion
Ceramic nanoparticles, with their superiority over synthetic counterparts, have proved to be a good alternative for drug delivery compared to traditional systems. The foremost advantage of ceramic nanoparticles is that they are unaffected by pH and temperature. Moreover, they can be moulded into the desired size, shape and form. These characteristics propose them as ideal delivery systems but lack of research is the biggest hurdle in the way of their clinical use. The major issue preventing their use is their toxicity concerns. Even though they possess many advantages, their side effects are still in scrutiny, mainly the toxic effects they may possibly have in the body. Although quality research has been carried out in this field, the road ahead is still long, till full and adequate knowledge about ceramic nanoparticles will be available. However, with the positive direction that research is following, and with so many privileges offered by ceramic nanoparticles, they could be the future prospect of drug delivery.
Acknowledgement
The authors are thankful to Director, University Institute of Pharmacy, Pt. Ravishankar Shukla University, Raipur (C.G.) India for this study and UGC-MRP-41-748-2012, UGC-RA 70-371/2012, DST-FIST for financial assistance relating to this work.
Declaration of interest
The authors report no declarations of interest. The authors alone are responsible for the content and writing of the paper.
References
- Armatas GS, Kanatzidis MG. 2006. Mesostuctured germanium oxide with cubic pore symmetry. Nature. 441:1122–1125.
- Alyautdin RN, Reichel A, Lobenberg R, Ramge P, Kreuter J, Begley DJ. 2001. Interaction of poly(butylcyanoacrylate) nanoparticles with the blood-brain barrier in vivo and in vitro. J Drug Target. 9: 209–221.
- Barenholz Y. 2012. Doxil® - The first FDA-approved nano-drug: lessons learned. J Control Release. 160:117–134.
- Block JE, Thorn MR. 2000. Clinical indications of calcium-phosphate biomaterials and related composites for orthopedic procedures. Calcif Tissue Int. 66:234–238.
- Boris MH, Reichert JC, Schantz JT, Gbureck U, Rackwitz L, Nöth U, et al. 2013. How smart do biomaterials need to be? A translational science and clinical point of view. Adv Drug Del Rev. 65:581–603.
- Chithrani BD, Ghazani AA, Chan WCW. 2006. Determining the size and shape dependence of gold nanoparticle uptake into mammalian cell. Nano Lett. 6:662–668.
- Clift MJ, Rutishauser BR, Brown DM. 2008. The impact of different nanoparticle surface chemistry and size on uptake and toxicity in a murine macrophage cell line. Toxicol Appl Pharmacol. 232:418–427.
- LaVan DA, McGuire T, Langer R. 2003. Small scale systems for in vivo drug delivery. Nat Biotechnol. 21:1184–1191.
- Dubertret B, Skourides P, Norris DJ, Noireaux V, Brivanlou AH, Libchaber A. 2002. In vivo imaging of quantum dots encapsulated in phospholipid micelles. Science. 298:1759–1762.
- Duncan R. 2003. The dawning era of polymer therapeutics. Nat Rev Drug Disc. 2:347–360.
- Duncan R. 2006. Polymer conjugates as anticancer nanomedicines. Nat Rev Cancer. 6:688–701.
- Farokhzad OC, Langer R. 2009. Impact of nanotechnology on drug delivery. ACS Nano. 3:16–20.
- Ferrari M. 2005. Cancer nanotechnology: opportunities and challenges. Nat Rev Cancer 5:161–171.
- Fubini B. 1997. Surface reactivity in the pathogenic response to particulates. Environ health perspect 105:1013–1020.
- Gao H, Shi W, Freund LB. 2005. Mechanics of receptor-mediated endocytosis. Proc Natl Acad Sci USA. 102:9469–9474.
- Geiser M, Rothen-Rutishauser B, Kapp N, Schurch S, Kreyling W, Schultz H, et al. 2005. Ultrafine particles cross cellular membranes by nonphagocytotic mechanisms in lungs and in cultured cells. Environ Health Perspect. 113:1555–1560.
- Gash AE, Tilltson TM, Satcher JH, Hrubesh LW, Simpson RL. 2001. New sol-gel synthetic route to transition and main group metal oxide aerogels using inorganic salt precursors. J Non-cryst Solids. 285:22–28.
- Gladstone HB, Mc Dermott MW, Cooke DD. 1995. Implants for cranioplasty, Otolaryngol. Clin North Am. 28:381–400.
- Harper S, Usenko C, Hutchison JE, Maddux BLS, Tanguay RL. 2008. In vivo biodistribution and toxicity depends on nanomaterial composition, size, surface functionalization and route of exposure. J Exp Nanosci. 3:195–206.
- Hrkach J, Von Hoff D, Ali MM, Andrianova E, Auer J, Campbell T, et al. 2012. Preclinical development and clinical translation of a PSMA-targeted docetaxel nanoparticle with a differentiated pharmacological profile. Sci Transl Med. 4:128–139.
- Hussain S, Boland S, Baeza-Squiban A, Hamel R, Thomassen LC, Martens JA. 2009. Oxidative stress and proinflammatory effects of carbon black and titanium dioxide nanoparticles: role of particle surface area and internalized amount. Toxicology. 260:142–149.
- JinJin S, Wang Z, Wang L, Wang H, Li L, Yu X, Zhang J, Ma R, Zhang Z. 2013. Photodynamic therapy of a 2-methoxyestradiol tumor-targeting drug delivery system mediated by Asn-Gly-Arg in breast cancer. Int J Nanomedicine. 8:1551–1562.
- Kohane DS. 2007. Microparticles and nanoparticles for drug delivery. Biotechnol Bioeng. 96:203–209.
- Kevadiya BD, Bajaj HC. 2013. The layered silicate montmorillonite as a drug delivery. Key Eng Mat. 571:111–132.
- Lade M, Mays H, Schmidt J, Willumeit R, Schomacker R. 2000. On the nanoparticle synthesis in microemulsions: detailed characterization of an applied reaction mixture. Colloids Surf A Phys Chem. 163:3–15.
- Li X, Kanjwal MA, Lin L, Charonakis IS. 2013. Electrospun polyvinyl-alcohol nanofibres as oral fast-dissolving delivery system of caffeine and riboflavin. Key Eng Mat. 103:182–188.
- Linkov I, Satterstrom FK, Corey LM. 2008. Nanotoxicology and nanomedicine: making hard decisions. Nanomedicine Nanotech Bio Med. 4:167–171.
- Liu J. 2014. Hyperbranched polyether hybrid nanospheres with CdSe quantum dots incorporated for selective detection of nitric oxide. Mater Lett. 123:104–106.
- Lo S, Kumar A, Hsieh JT, Sun X. 2013. Dendrimers nanoscaffold for potential theranostics of prostate cancer with a focus on radiochemistry. Mol Pharm. 10:793–812.
- Marchant GE. 2009. Small is beautiful: what can nanotechnology do for personalized medicine? Curr Pharmacogenomics Person Med. 7:231–237.
- Medina SH, El-Sayed Mohd EH. 2009. Dendrimers as carriers for delivery agents. Chem Rev. 109:3141–3157.
- Miller K, Erez R, Segal E, Shabat D, Satchi-Fainaro R. 2009. Targeting bone metastases with a bispecific anticancer and antiangiogenic polymer–alendronate–taxane conjugate. Angew Chem Int Ed. 48:6387–6392.
- Muhlfeld C, Gehr P, Rothen-Rutishauser B. 2008. Translocation and cellular entering mechanisms of nanoparticles in the respiratory tract. Swiss Med Wkly. 138:387–391.
- Muller RH, Mader K, Gohla S. 2000. Solid lipid nanoparticles (SLN) for controlled drug delivery a review of the state of the art. Eur J Pharm Biopharm. 50:161–177.
- Muller RH, Radtke M, Wissing SA. 2002. Solid lipid nanoparticles (SLN) and Nanostructured lipid carriers (NLC) in cosmetic and dermatological preparation. Adv Drug Deliv Rev. 54:5131–5155.
- Muro S, Schuchman EH, Muzykantov VR. 2006. Lysosomal enzyme delivery by ICAM-1-targeted nanocarriers bypassing glycosylation- and clathrin-dependent endocytosis. Mol Ther. 13: 135–141.
- Nel AE, Madler L, Velegol D, Xia T, Hoek EMV, Somasundaran P, et al. 2009. Understanding biophysicochemical interactions at the nano- bio interface. Nat. Mater. 8:543–557.
- Oberdorster G. 2010. Safety assessment for nanotechnology and nanomedicine: concepts of nanotoxicology. J Int Med. 267:89–105.
- Paveer U, Ingrid T, Lust A. 2014. Soluplus Graft copolymer: potential novel carrier polymer in electrospinning of nanofibrous drug delivery systems for wound therapy. Biomed Res Int. 2014:789765. Article ID 789765
- Peer D, Karp JM, Hong S, Farokhzad OC, Margalit R, Langer R. 2007. Nanocarriers as an emerging platform for cancer therapy. Nat Nanotechnol. 2:751–760.
- Porter AE, Muller K, Skepper J, Midgley P, Welland M. 2006. Uptake of C60 by human monocyte macrophages, its localization and implications for toxicity: studied by high resolution electron microscopy and electron tomography. Acta Biomater. 2:409–419.
- Porter AE, Gass M, Muller K, Skepper JN, Midgley PA, Welland M. 2007. Direct imaging of single-walled carbon nanotubes in cells. Nat. Nanotechnol. 2:713–717.
- Quan CY, Chang C, Wei H, Chen CS, Xu XD, Cheng SX, et al. 2009. Dual targeting of a thermosensitive nanogel conjugated with transferrin and RGD-containing peptide for effective cell uptake and drug release. Nanotechnology. 20:335101.
- Rawat M, Singh D, Saraf S, Saraf S. 2006. Nanocarriers: Promising vehicle for bioactive drugs. Biol Pharm Bull. 29:1790–1798.
- Rawat M, Singh D, Saraf S, Saraf S. 2008. Development and in vitro evaluation of alginate gel-encapsulated, chitosan-coated ceramic nanocores for oral delivery of enzyme. Drug Dev Ind Pharm. 34: 181–188.
- Riman RE, Suchanek WL, Lencka MM. 2002. Hydrothermal crystallization of ceramics. Ann Chim Sci Mat. 27:15–36.
- Roy R, Kumar S, Tripathi A, Das M, Dwivedi PD. 2014. Interactive threats of nanoparticles to the biological system. Immunol Lett. 158:79–87.
- Sakamoto JH, van de Ven AL, Godin B, Blanco E, Serda RE, Grattoni A, et al. 2010. Enabling individualized therapy through nanotechnology. Pharmacol Res. 62:57–89.
- Sanvicens N, Marco MP. 2008. Multifunctional nanoparticles– properties and prospects for their use in human medicine. Trends Biotechnol. 26:425–433.
- Schins RP, Duffin R, Hohr D, Knaapen AM, Shi T, Weishaupt C, et al. 2002. Surface modification of quartz inhibits toxicity, particle uptake, and oxidative DNA damage in human lung epithelial cells. Chem Res Toxicol. 15:1166–1173.
- Shi J, Yu X, Wang L, Liu Y, Gao J, Zhang J, et al. 2013. PEGylated fullerene/iron oxide nanocomposites for photodynamic therapy, targeted drug delivery and MR imaging. Biomaterials. 34: 9666–9677.
- Shibahara A, Hoshino A, Hanaki K, Suzuki K, Yamamoto K. 2004. On the cyto-toxicity caused by quantum dots. Microbiol Immunol. 48:669–675.
- Shvedova AA, Kagan VE, Fadeel B. 2010. Close encounters of the small kind: adverse effects of man-made materials interfacing with the nano-cosmos of biological systems. Annu Rev Pharmacol Toxicol. 50:63–88.
- Singh D, Dubey P, Pradhan M, Rawat M. 2013. Ceramic nanocarriers: versatile nanosystem for protein and peptide delivery. Expert Opin Drug Deliv. 10:241–259.
- Singer JW, Shaffer S, Baker B, Bernareggi A, Stromatt S, Nienstedt D, Besman M. 2005. Paclitaxel poliglumex (XYOTAX; CT-2103): an intracellularly targeted taxane. Anticancer Drugs. 16:2223–228.
- Singh S, Shi T, Duffin R, Albrecht C, van Berlo D, Hohr D, et al. 2007. Endocytosis, oxidative stress and IL-8 expression in human lung epithelial cells upon treatment with fine and ultrafine TiO2: role of the specific surface area and of surface methylation of the particles. Toxic Appl Pharmacol. 222:141–151.
- Stefani D, Wardman D, Lambert T. 2005. The implosion of the Calgary General Hospital: ambient air quality issues. J Air Waste Manag Assoc. 55:52–59.
- Suh WH, Suslick SK, Stucky Galen D, Suh YH. 2009. Nanotechnology, nanotoxicology, and neuroscience. Pro Neurobiol. 87:133–170.
- Ulbrich K, Subr V. 2004. Polymeric anticancer drugs with pH-controlled activation. Adv Drug Deliv Rev. 56:1023–1050.
- Whitters CJ, Strang R, Brown D, Clarke RL, Curtis RV, Hatton PV, et al. 1999. Dental materials: 1997 literature review. J Dent. 27: 401–435.
- Wu C, Zreiqat H. 2010. Porous bioactive diopside (CaMgSi2O6) ceramic microspheres for drug deliver. Acta Biomater. 6:820–829.
- Xia T, Kovochich M, Brant J, Hotze M, Sempf J, Oberley T, et al. 2006. Comparison of the abilities of ambient and manufactured nanoparticles to induce cellular toxicity according to an oxidative stress paradigm. Nano Lett. 6:1794–1807.
- Wu X, Wu M, Zhao JX. 2014. Recent development of silica nanoparticles as delivery vectors for cancer imaging and therapy. Nanomedicine. 10:297–312.
- Yacobi NR, Phuleria HC, Demaio L, Liang CH, Peng CA, Sioutas C, et al. 2007. Nanoparticle effects on rat alveolar epithelial cell monolayer barrier properties. Toxicol In Vitro. 21:1373–1381.
- Zhang L. 2004. Preparation of multi-component ceramic nanoparticles. CISM. 8–21.
- Zhang XQ, Xu X, Bertrand N, Pridgen E, Swami A, Farokhzad OC. 2012. Interactions of nanomaterials and biological systems: Implications to personalized nanomedicine. Adv Drug Delivery Rev. 64:1364–1384.
- Zhou X, Porter LA, Robinson KRD, Shim SM, Guo Y. 2014. Nano-enabled drug delivery: a research profile. Nanomedicine. 10: 889–896.
- Zou XD, Conradsson T, Klingstedt M, Dadachov MS, O’ Keeffe M. 2005. Mesostuctured germanium oxide with crystalline pore walls and its chiral derivative. Nature. 437:716–719.