ABSTRACT
The present study provides details of male meiosis on a population basis in Solanum seaforthianum Andrews of family Solanaceae from Mount Abu (Rajasthan). In total five populations are worked out from different localities of Mount Abu. Three accessions are reported at diploid level (n = 12) and two populations at tetraploid level (n = 24). The tetraploid cytotype is a new chromosome addition for the species worldwide. The tetraploid cytotype shows abnormal spindle behavior leading to the formation of unoriented bivalents, scattered chromosomes at anaphases and telophases, multipolarity, laggards and irregular microsporogenesis and high pollen sterility. Morphologically the two cytotypes are identical and do not show any significant difference.
Introduction
Meiosis is a sensitive process for sexual reproduction in plants and maintains diploid chromosome number generation after generation. Meiosis is controlled by large number of genes as is confirmed from many meiotic mutants studied in plants and other living organisms (Kaul Citation1988; Bhatt et al. Citation2001). For this phenomenon to occur normally, chromosomes segregate, regular cell division takes place and haploid gametes are produced. But this process is occasionally affected due to certain mutations (Jiang et al. Citation2003) by which normal meiotic course is disturbed, leading to abnormalities resulting in altered genetic constitution.
Solanum seaforthianum is commonly known as Brazilian nightshade as it is a native of Brazil and naturalized elsewhere. It is distributed in lower hills of the Sub-Himalayan tract, rather than the plains. The plant is an evergreen climber with pinnate leaves, elliptic ovate lobes, and oblique base. Flowers are in clusters of terminal dropping panicles, corolla bluish purple. Calyx lobes are very short. In Rajasthan, it is only found in hills of Mount Abu region. Regarding its ethnobotany, its extract is used by the Paliyan tribe to cure skin diseases and for treatment of boils. In other parts of India, it is used to cure bronchitis and asthma (ShaliSaheb Citation2014). This species has not been cytologically determined from this region. Thus, extensive meiotic studies have been carried out on a population basis from Mount Abu region of district Sirohi.
Materials and methods
Cytological investigations were carried out on five accessions of Solanum seaforthianum collected from Mount Abu region of Rajasthan, altitude varying from 1220 m to 1750 m. For meiotic studies, young inflorescences were fixed in Carnoy’s fixative (6:3:1 ethanol:chloroform:glacial acetic acid) for 40–48 h. Afterwards, these were transferred to 70% ethyl alcohol and stored in a refrigerator until use. For chromosomal preparations, anthers were crushed and tapped to prepare a smear of pollen mother cells (PMCs) in 1% acetocarmine. Large numbers of PMCs were observed at different stages of meiosis to look for the frequency of different abnormalities. Pollen fertility was observed by mounting the pollen grains in 50% glycerol-acetocarmine (1:1) solution. The photomicrographs of the PMCs and pollen grains were taken from the temporary slides by using the Nikon 80i digital imaging system(Tokyo, Japan). Voucher specimens of cytologically determined plants have been deposited in the Herbarium, Department of Botany, Punjabi University Patiala, India, and accession numbers are given in .
Table 1. Data showing accession number, meiotic chromosome number, ploidy level and localities with altitude of different populations of Solanum seaforthianum from Mount Abu.
Results
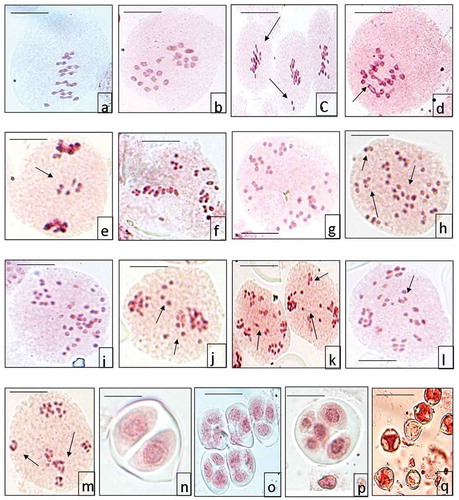
Cytological investigations have been made in five accessions of Solanum seaforthianum collected from different localities of Mount Abu. Three out of five accessions showed chromosome number n = 12, i.e. at diploid level as confirmed from 12 bivalents at metaphase I () and 12:12 equal distribution of chromosomes at anaphase I (). The population collected from Abu Road had normal meiotic course and high pollen fertility but populations from Guru Shikhar and Machgaon area had abnormal meiotic course. Meiotic abnormalities included chromosome stickiness and out of plate bivalents (), bridges (), laggards () at anaphase I of meiosis and presence of micronuclei during microsporogenesis. As a result of these abnormalities, pollen fertility decreases drastically (66%). The report of diploid cytotype is in conformity with the earlier reports from India by Madhavadian (Citation1968) from Tamil Nadu and Sarkar et al. (Citation1980) from central India. From outside India, Roe (Citation1967) from USA, and Gerasimenko, II. and Reznikova (Citation1968) from Russia reported the same number. However, Gottschalk (Citation1954) reported 2n = 32 from Australia.
Figure 1. (a) PMC at metaphase I 12 bivalents. (b) PMC at anaphase I 12:12. (c) PMC at metaphase I showing chromosomal stickiness and unoriented bivalents. (d) PMC at anaphase I showing multiple bridges. (e) PMC showing laggards at anaphase I. (f) PMC at metaphase I with the absence of equatorial plate and bivalents are scattered in the cytoplasm. (g) PMC at anaphase I showing 24:24. (h) PMC showing spindle irregularities. (i) PMC with unequal distribution of chromosomes at anaphase I. (j) PMC showing laggards at anaphase I. (k) PMC showing laggards at anaphase II. (l) PMC showing multipolarity. (m) Dyad. (n) Triads. (o) Polyad. (p) Sterile and fertile pollen grains.
Discussion
In the present study, two populations collected from Salgaon and Nakki Lake area of Mount Abu had a chromosome number of n = 24, which is clear from 24 bivalents at metaphase I () and 24:24 distribution of chromosomes at anaphase I (). This is the first ever report for the tetraploid cytotype in the species at the world level. A majority of the pollen mother cells (70%) depicted irregular spindle activity. Chromosomes in these pollen mother cells lack the ability of congregation of chromosomes at single pole and remained scattered in the cytoplasm (). This resulted in unequal distribution of chromosomes to poles at anaphase I () and laggards at anaphase I () and anaphase II (). These chromosomes fail to reach the respective poles leading to the formation of micronuclei during late telophases. Irregular spindle activity also leads to multipolarity at anaphase I in some PMCs or scattered chromosomes at anaphase I (). As a result of these abnormalities, microsporogenes is highly abnormal with the presence of dyads (), triads () and polyads (). Besides, pollen fertility also decreases as sterile pollen grains are formed (). This is not a case of asynaptic mutant as is clear from 24 bivalents () formation at metaphase I, but the arrangement of these bivalents on the equatorial plate is abnormal. These spindle abnormalities occur throughout the course of meiosis. As at most of the anaphases of the meiosis the chromosomes are unable to move to poles and remain scattered in the cells () and in other cases there is unequal distribution of the chromosomes at each pole; sometimes more than four poles are also seen () ( and ).
Table 2. Data on meiotic abnormalities at different stages observed in four populations of Solanum seaforthianum from Mount Abu.
Table 3. Data on abnormal microsporogenesis four different populations of Solanum seaforthianum from Mount Abu.
Chromosome stickiness plays an important role as it prevents the chromosomal separation and produces unreduced gametes as suggested by Dewitti et al. (Citation2010) in Begonia species. During the cell division, the function of the spindle is to separate and reorganize chromosomes. But if due to some genetic factors the spindle activity is abnormal, then bivalents are unable to line up in the equator and then separate at anaphase, leading to abnormal meiotic course. There are at least 14 proven or putative proteins which are identified in plants which are necessary for chromosomal segregation (Jiang et al. Citation2003). According to recent research on Arabidopsis thaliana, a specific protein called Multipolar Spindle 1 is tangled in spindle organization in meiocytes (Jiang et al. Citation2003) and AtPS1 gene is responsible for diploid gamete (2n) formation due to irregular spindle activities (D’ Erfurth et al. Citation2008). Earlier, a number of plants have been reported with abnormalities such as irregular spindle activity, cytomixis, and chomatin stickiness leading to abnormal microsporogenesis (Baum et al. Citation1992; Risso-Pascotto et al. Citation2003; Kumar and Singhal Citation2008; Singhal and Kaur Citation2009).
It has been concluded that spindle irregularities give rise to various other abnormalities like laggards, bridges, multipolarity and irregular microsporogenesis resulting in dyads, triads, polyads and ultimately low pollen fertility. Tetraploid cytotype is more abnormal, compared to the diploid cytotype.
Acknowledgments
The first author is highly thankful to DBT-IPLS for providing Senior Research Fellowship (ref. no. BT/PR 4548/NF/22/146/2012) and Head, Department of Botany, Punjabi University, Patiala for providing necessary laboratory facilities. We are also thankful to Director of Botanical Survey of India, Jodhpur for their invaluable help in identification of plant species.
Disclosure statement
No potential conflict of interest was reported by the authors.
Additional information
Funding
References
- Baum ME, Lagudah ES, Appels R. 1992. Wide crosses in cereals.annual review of plant. Phyiology Mol Biol. 43:117–143.
- Bhatt AM, Canales C, Dickison HG. 2001. Pant meiosis: the means to 1N. Trends in Plant Sciences. 6:114–121.
- D’ Erfurth I, Jolivet S, Froger N, Catrice O, Novatchkova M, Simon M, Jenczewski E, Mercier R. 2008. Mutations in AtPS1 (Arabidopsis thaliana Parallel Spindle 1) lead to the production of diploid pollen grains. Genetics. 4:11.
- Dewitti A, Eeckhaut T, Van Huylenbroeck J, Van Bockstaele E. 2010. Meiotic aberrations during 2n pollen formation in Begonia. Heredity. 104:215–223.
- Gerasimenko, II. and Reznikova. 1968. A cytological investigation of genus Solanum. BotanicheskiiZhurnal. 53:505–513.
- Gottschalk W. 1954. Die chromosomenstruktur der Solanacee nunter Berucksichtigung phlogenetischer Fragestellungen. Chromosoma. 6(6–7):539–626.
- Jiang J, Birchler JA, Parrot WA, Dawe RK. 2003. A molecular view of plant centromeres. Trends Plant Sci. 8:570–575.
- Kaul MLH. 1988. Male sterility in higher plants. New York: Springer, Berlin Heidelberg.
- Kumar P, Singhal VK. 2008. Cytology of Calthapalustris L. (Ranunculaceae) from cold regions of Westen Himalayas. Cytologia (Tokyo). 73:137–143.
- Madhavadian P. 1968. Chromosome number in South Indian Solanaceae. Caryologia. 21:343–347.
- Risso-Pascotto C, Pagliarini MS, Valle CB. 2003. A mutation in the spindle checkpoint arresting meiosis II in Brachiaria ruziziensis. Genome. 46:724–728.
- Roe KE. 1967. Chromosome size in Solanum and Cyphomandra: taxonomic and phylogenetic implications. Am Naturalist. 101:295–297.
- Sarkar AK, Datta N, Chatterjee. 1980. In chromosome number report CXVII. Taxon. 29: 358–360.
- ShaliSaheb T. 2014. A study on medicinal climbers of Nallamalais, AndraPardesh.International. J Multidisciplinary Res Dev. 1(5):172–176.
- Singhal VK, Kaur D. 2009. Spontaneous occurrence of meiotic spindle abnormalities in ‘Long Headed Poppy’ (Papaverdubium L.) from Indian cold regions. Cytologia (Tokyo). 74:385–389.
