?Mathematical formulae have been encoded as MathML and are displayed in this HTML version using MathJax in order to improve their display. Uncheck the box to turn MathJax off. This feature requires Javascript. Click on a formula to zoom.
?Mathematical formulae have been encoded as MathML and are displayed in this HTML version using MathJax in order to improve their display. Uncheck the box to turn MathJax off. This feature requires Javascript. Click on a formula to zoom.Abstract
In molecular ro-vibrational calculations, it is desirable to build a symmetry-adapted basis set where both the rotational and vibrational functions are each separately symmetrised. Enabling rotational symmetrisation requires the use of body-fixed frames whose rotations under molecular symmetry operations are constant. The familiar Eckart and Sayvetz frames are examples of such frames, as are bisector frames for certain molecules. In this work, we introduce alternative frames for CH3Cl-type molecules and C2H6 that can be used for rotational symmetrisation. These are defined by the vibrational coordinates without the need for a reference geometry. We also provide one method to find such frames. Finally, we state a straightforward transformation of the s-vectors when changing frames which facilitates the evaluation of kinetic energy operators in these non-standard frames.
1. Introduction
In ro-vibrational eigenfunction calculations, the choice of body-fixed (BF) frame is of great significance. A poorly chosen frame can result in large Coriolis coupling, leading to problems in the convergence of ro-vibrational energies. It is well known that the Eckart (for rigid molecules) and Sayvetz (for non-rigid molecules) frames [Citation1] are designed to eliminate the first-order Coriolis coupling and as such are a typical choice [Citation2–5]. Another common frame, defined by two vectors, has an axis parallel to one of the vectors and an axis perpendicular to the plane formed by the vectors (bond vector frames) [Citation6–12]. For symmetric triatomic (ABA) molecules and some polyatomics, a frame which bisects two vectors or two planes is a standard choice [Citation13–15].
A further consideration relates to the molecule's symmetry. It is often desirable to build a symmetry-adapted basis set as this reduces the corresponding Hamiltonian matrix to block diagonal form [Citation16] and permits the labelling of the ro-vibrational eigenfunction as an irreducible representation (irrep) of the molecular symmetry (MS) group [Citation17]. This label is crucial in intensity calculations because the eigenfunction has an associated nuclear spin weight dictated by its symmetry that in turn affects the transition intensity [Citation18].
One approach to symmetrisation is to independently classify functions of the rotational and vibrational coordinates as irreps of the MS group so that the combined functions have the form
(1)
(1) where (θ, ϕ, χ) are the rotational coordinates,
are the vibrational coordinates, while a and b number the rotational and vibrational functions. The functions are the
th and
th components of the irreps
and
, respectively. Multiplying all functions of irrep
with all functions of irrep
results in a list of functions which have a reducible representation
This can be reduced to a sum of irreps through the equation
(2)
(2) where
are the coefficients and
are the irreps [Citation18]. The result is a set of symmetrised functions of the form
(3)
(3) where
is a coefficient and k numbers the combined function. It is the
th component of the irrep
.
In this approach, to classify the rotational part, one needs to know how the Euler angles transform under the MS operation, or, in other words, how the BF-frame rotates under this operation. When the Eckart or Sayvetz frames are used, there are the so-called equivalent rotations [Citation18] of the BF-frame which occur during an MS operation. This procedure has enabled the successful construction of the symmetry-adapted eigenfunctions of several molecules [Citation19–25].
In general, however, the rotation associated with an MS operation depends on the vibrational coordinates. In particular, the χ Euler angle transforms as [Citation18]
(4)
(4) Because the rotational and vibrational coordinates are no longer separable, such a dependency will lead to a breakdown of the rotational basis set symmetrisation and prevent the construction of a symmetry-adapted rotational basis set [Citation15].
There are two possible solutions to this problem. The first is to relax the rotational and vibrational separation in Equation (Equation1(1)
(1) ) and instead symmetrise the combined rotational and vibrational functions together, rather than separately [Citation26]. This would essentially result in Equation (Equation3
(3)
(3) ) without Equation (Equation1
(1)
(1) ) as an intermediate. This has the advantage of greatly increasing the possible frames that can be used, including ones that are easy to define such as the bond vector frame. The other option, the one explored in this publication, is to use BF-frames where γ is a constant for all MS operations and all vibrational geometries. The ensuing rotational and vibrational separation simplifies the process because subsets of the coordinates can be treated independently during symmetrisation [Citation15].
As the Eckart and Sayvetz frames can only be solved analytically for triatomics [Citation27] and planar molecules [Citation28], and because they are suboptimal for molecules without a well-defined equilibrium, they are not always a suitable choice. Our goal is to find geometric frames – ones that can be expressed as an analytic function of the vibrational coordinates – for certain molecules where the frame's rotations are constants under symmetry operations.
The structure of this publication is as follows: Section 2 describes the action of the MS operations on the rotational and vibrational coordinates. The example of an ABA molecule illustrates frames where the rotation under MS operations depends on the vibrational coordinates. The transformation and invariance of the kinetic energy operator is stated and proved in Section 3. With the same ABA illustrative example, Section 4 demonstrates why rotational basis set symmetrisation is not possible for certain frames. Section 5 shows that the correct transformation of the kinetic energy operator was derived for an ABA molecule for both a bisector frame and a bond vector frame. It further proves that the rotations are constant under symmetry operations when using the Eckart frames. Section 6 largely follows the pattern of Section 5, but for the CH3Cl molecule. It also provides a geometric frame for which the rotations are constant under symmetry operations. Section 7 uses C2H6 as an example of Section 3 for a Sayvetz and a geometric frame. Section 8 shows how one can find a geometric frame for CH3Cl with which one can construct a symmetry-adapted rotational basis set and how to transform the kinetic energy operator between frames. Finally, Section 9 mentions the possible avenues for further research and Section 10 concludes the publication.
2. Coordinate transformations
To begin, some notation. The position vector of nucleus α from the centre of mass is denoted . Instead of a space-fixed (SF) frame, we use a ‘rotationally fixed’ (RF) frame whose origin is at the nuclear centre of mass but whose unit vectors are parallel to the unit vectors of the SF-frame. We will also partially follow Sørensen's [Citation29] notation in denoting the RF and BF unit vectors by
and
, that is, uppercase subscripts are RF unit vectors and lowercase are BF unit vectors. Then, the Cartesian coordinates of the position vectors in each frame are given by
(5)
(5) A list of coordinates
will be denoted by
, i.e. unbolded but without the subscript indicating the specific component.
Finally, a transformed object under MS operations has an apostrophe. For example, the transformed Cartesian coordinates of after a nuclear permutation is written as
. These must be the Cartesian coordinates of another atom
, thus
.
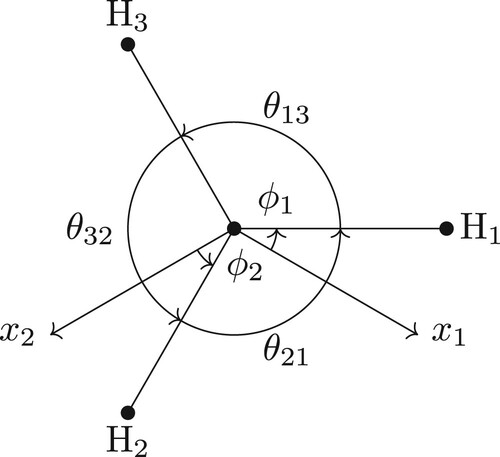
Typically, the Cartesian coordinates are expressed in terms of the vibrational and angular coordinates: e.g. where the latter three coordinates are the Euler angles which specify the orientation of the BF-frame with respect to the RF-frame [Citation30]. We use the standard definition of the Euler angles which involves first rotating the BF-frame by ϕ about the z-axis of the BF-frame (which is also the z-axis of the RF-frame initially), then rotating the BF-frame by θ about the y-axis of the BF-frame, and finally by χ about the z-axis of the BF-frame. All rotations are counter-clockwise (right handed). See Figure . With this definition the coordinates
in the RF-frame are given by
(6)
(6) where
(7)
(7) and
are the coordinates of
in the BF-frame.
Figure 1. A visual representation of the Euler angles. (a) The first two Euler angles ϕ and θ which are essentially angles defining the spherical coordinates. The first rotation of ϕ is about the z-axis of both the RF- and BF-frame which initially coincide. The BF-frame is then rotated by θ about the y-axis of the BF-frame or the -axis of the figure. (b) The final Euler angle χ is due to a rotation about the z-axis of the BF-frame or the
-axis of the figure.

The transformation of the RF Cartesian coordinates – the left-hand side of Equation (Equation6(6)
(6) ) – under MS operations is well-defined and explained in [Citation18]. For the transformation of the BF Cartesian coordinates, however, the definition is not so clear-cut as only the transformed Euler matrix multiplied by the transformed BF Cartesian coordinates – the right-hand side of Equation (Equation6
(6)
(6) ) – must equal the transformed RF Cartesian coordinates. There is thus an ambiguity for the transformation of the BF Cartesian coordinates as the rotation of the frame can be defined to compensate. See Appendix 2 of [Citation31] for an example of this.
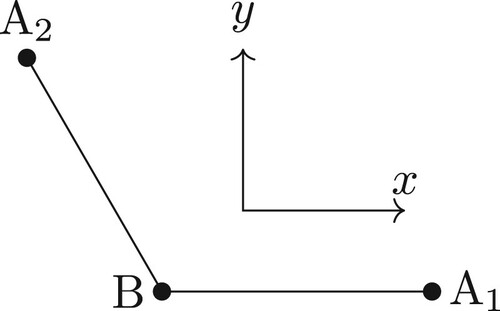
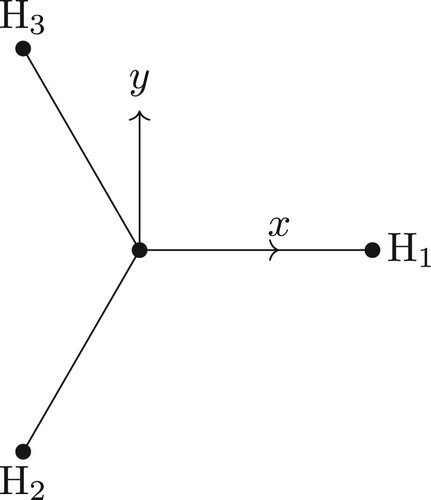
Figure 2. The bond vector BF-frame for the ABA molecule with the x-axis parallel to the A–B bond and the y-axis in the plane of the three atoms. The axes are offset for clarity.
Our approach defines the transformation of the BF Cartesian coordinates from the transformation of the vibrational coordinates. The latter can be determined if their definition in terms of the Cartesian coordinates is known. See Appendix 3 from [Citation32]. Since the vibrational coordinates fully specify the BF Cartesian coordinates, their transformation encodes the transformation of the BF Cartesian coordinates. The change in the left-hand side of Equation (Equation6
(6)
(6) ) then can be used to find the Euler angle transformation.
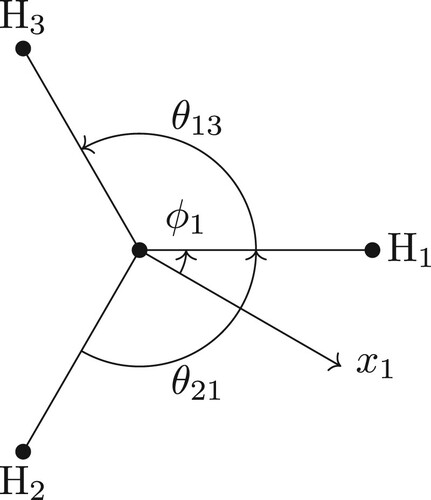
As an example, consider a triatomic molecule of type ABA and let the A-type atoms (called A1 and A2) have coordinates and
. In this representation,
and
are the AB bonds while
is the ABA angle.
Throughout this work, the origin is chosen for convenience rather than at the centre of mass. Since the vector from the chosen origin to the centre of mass is the same for all atoms, the desired transformation properties will trivially be satisfied. In the case of the ABA molecule, the origin will be at the B atom.
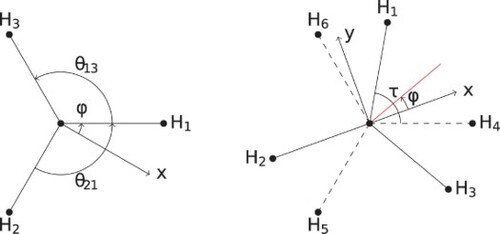
For the BF-frame, one possible choice is with the x-axis parallel to the bond from atom B to A1 (i.e. parallel to the vector) and the z-axis perpendicular to the plane of ABA (see ). With this choice, the BF Cartesian coordinates for the ABA molecule are
(8)
(8) The operation (12) permutes
and
resulting in new BF Cartesian coordinates given by
(9)
(9) where
is the result after transforming the vibrational coordinates in the expression for
. In general, the Euler angles also change, and we call the Euler matrix with the transformed angles as
. As we have
and
now defines a rotated BF-fame, we see that the transformed
are the BF Cartesian coordinates of
in the rotated BF-frame and after the transformation. The BF-frame is rotated in such a way so as to ensure that
and vice versa. In this case, the new RF Cartesian coordinates are
(10)
(10) In performing this operation, we now have three frames: the RF-frame and the two BF-frames. Denoting the new BF-frame unit vectors as
, i.e. with an asterisk, the components of
are then
(11)
(11) It is of interest to note what the transformed Cartesian coordinates in the original BF-frame are (denoted by
). Since the Euler matrix of this frame is unchanged, if
, we have,
(12)
(12) and so
. We also have
(13)
(13) where
is the rotation matrix relating the coordinates of
in the new BF-frame to that of the old BF-frame. In this case, it is
(14)
(14)
3. Kinetic energy transformation
3.1. Kinetic energy operator
For completeness, we state the MS transformation properties of the objects used in constructing the kinetic energy operator (KEO), namely, the s and t vectors [Citation29]. These are labelled as, for example, , which is the s-vector for atom α and vibrational or rotational coordinate a. The vibrational coordinates will be labelled by i, j and k, and their primed versions; the rotational coordinates by g and h, and their primed versions. The Hamiltonian
is then given by
(15)
(15) where V is the potential energy while
is the KEO given by
(16)
(16) where
is the mass of atom α,
is the vibrational momentum operator for vibrational coordinate
,
is the g-axis BF component of the SF angular momentum operator, and U is the pseudo-potential. The generic terms in brackets are known as the G-matrix elements, defined by
(17)
(17) The terms of Equation (Equation16
(16)
(16) ) contain both vibrational and rotational s-vectors, specified by their subscript, which are inverses of the rotational and vibrational t-vectors. We have the relations
(18)
(18) We shall consider all four in turn and then evaluate the transformation of the KEO, demonstrating its invariance.
3.2. s and t vector transformations
In the so-called s-vector approach [Citation33], one needs the vibrational and rotational s-vectors as well as the rotational t-vectors (to calculate the rotational s-vectors). For the transformation and
, the definition and transformation of the vectors and operators are summarised in Table . Table shows the transformation for the vibrational s-vectors and vibrational momentum operators for more complex vibrational transformations. The transformation of the rotational s-vectors and angular momentum operators is unchanged.
Table 1. The transformation of the t and s vectors.
Table 2. The transformation of in the left column and the corresponding transformed vibrational s-vector and vibrational momentum operator.
Table 3. The change in the Euler angles for a given rotation of the BF-frame.
The vibrational s-vector is defined
(19)
(19) whose components are in the RF-frame. In practical applications, however, we would prefer to have the components of
in the BF-frame.
In Appendix 1, it is shown that to calculate the components of in another frame one simply must evaluate
at the Cartesian coordinates of that frame [Citation35].
In the BF-frame, the Cartesian coordinates are functions of the vibrational coordinates. In Appendix 1, it is also shown that the transformed vibrational s-vectors can also be expressed in the new BF-frame by performing the vibrational coordinate transformation (which we shall denote as
) on the original BF components
. The same procedure also applies to the other vectors.
3.3. G-matrix transformation
From the transformation properties of the s-vectors, we can obtain the transformation of the G-matrix. Assuming the vibrational coordinates and
transform to
and
, respectively, the vibrational, rotational and Coriolis components of the G-matrix transform as
(20)
(20) where the rotation matrix N is that of Equation (Equation13
(13)
(13) ) and relates the directions of the original and rotated BF-frames and
defined in Table . The left-hand side of the above equations are of the form
, where P is the MS operation which transforms the coordinates first, after which the G matrix element
is applied to the transformed coordinates. The right-hand side is in terms of
which is the matrix element
applied to the un-transformed coordinates.
From the definition of the G matrix, we have
(21)
(21) where the s-vector components are in the RF, old BF and new BF-frame, respectively. Since the new BF-frame components of the transformed s-vectors are given by
, to transform the G matrix, one can also perform the vibrational coordinate transformation on the original G matrix.
Action of the full Hamiltonian on a function is of the form
(22)
(22) where implicit summation of repeated indices is assumed. In Appendix 2, it is shown that the Hamiltonian is invariant under the transformations of Table .
4. Rotational symmetrisation
As described in the Introduction, typical eigenfunctions in ro-vibrational calculations have the form
(23)
(23) If one applies the full Hamiltonian to the function
, then the rotational and Coriolis parts drop out, and we can find the transformation of
under MS operations. The vibrational operators then transform as
, i.e. without the second term, and the Hamiltonian is invariant, showing the vibrational eigenfunctions also are irreps of the same group [Citation18].
To symmetrise the rotational part, however, a standard choice of basis functions are the rigid rotors functions [Citation36]. These have definite transformation properties under the rotations defined in Table , given by
(24)
(24) and
(25)
(25) If we refer back to our ABA example and instead use the principal axis system at the vibrational equilibrium, shown in Figure , with the z-axis pointing out of the plane and the y-axis bisecting the ABA angle, then one can follow the prescription of Section. 12.1 of [Citation18] and find the ‘equivalent rotations’ of the axes during an MS operation. There are four symmetry operations for the MS group
(M):
, and they have the equivalent rotations of
, respectively. This results in the transformation properties of
being
(26)
(26) which is a representation of the MS group
(M) of the ABA molecule. Irreducible representations can be formed through standard projection operator techniques [Citation18]. The result is that the symmetrised functions
have the form
(27)
(27) where
. The values of p and the symmetry are summarised in Table .
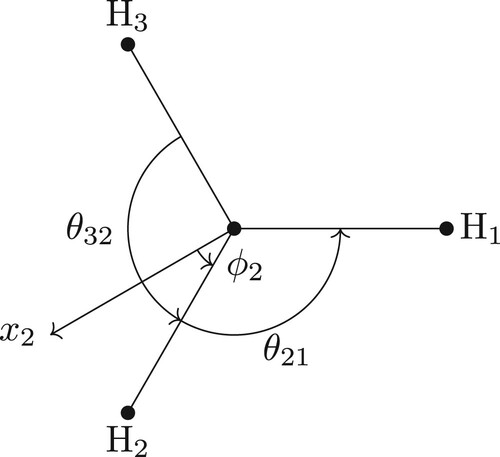
Figure 3. The principal axis system for the ABA molecule in vibrational equilibrium with the z-axis pointing out of the plane and the y-axis bisecting the A1–B–A2 bond. The axes are offset for clarity.
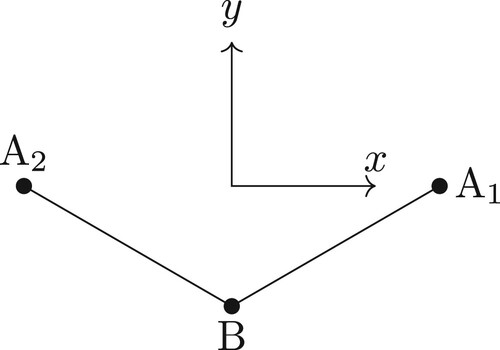
Table 4. The irrep of the function for the MS group
(M) depending on the values of J,
and p.
The key aspect of these transformations that enable this is that the transformation matrices are constant (up to J and k) which allow standard combinations of the rigid rotors (dependant on J and k) that transform irreducibly under the MS group transformations [Citation15].
In our original example, however, we saw that the rotations associated with the MS group were not the equivalent rotations, nor were they constant. The operation (12), for example, included the rotations whose effect on the rigid rotors is
(28)
(28) With this transformation, the representation of the group now depends on the angle
, and one can no longer form
independent combinations of the rigid rotors that transform as the irreps of the group. For example, if J and k are both even, the A
irrep has the form
(29)
(29) Thus, one cannot have separate symmetry-adapted rotational functions used in Equation (Equation1
(1)
(1) ) using this choice of BF-frame. A separate rotational basis set can still be used, but it cannot be assigned a symmetry label of the group in question. To do this, a different BF-frame is required. Alternatively, one could use the
dependent symmetry-adapted rotational functions and take them into account when performing further calculations.
The upshot of this result is that to keep the rotational and vibrational coordinates separate, and thus for the rotational functions to transform with matrices that do not depend on the vibrational coordinates, the rotation angle must be constant and its axis of rotation relative to the BF-frame must also be constant. This will ensure N is constant and . Whatever the rotation axis is, the angle must be
where n is the order of the MS operation.
5. ABA example
5.1. Bond vector frame
To demonstrate the MS transformation of the KEO and its invariance, we return to the ABA molecule in more detail. For our original BF-frame we know that matrix N for the operation (12) is Equation (Equation14(14)
(14) ). The components of the G matrix are as follows [Citation37]: the rotational part is
(30)
(30) where
is the reduced mass
(31)
(31) and
and
are the masses of A and B, respectively; the Coriolis part is
(32)
(32) where the rows correspond to the vibrational coordinates and the columns to the axes; the vibrational part of the G matrix
(33)
(33) If we list the vibrational momentum and angular momentum operators as
(34)
(34) then the full kinetic energy operator (besides the pseudo-potential) is given by
During the MS operation, we transform
in each of the G matrix elements. The operator
transforms to
(35)
(35) where C and D are given by
(36)
(36) The rotational operators transform as
. The transformed kinetic energy operator is then
(37)
(37) where the expressions in brackets can be confirmed to be the original G matrix elements. The other generator for the MS group of the ABA molecule is
. For this operation, the vibrational coordinates are unchanged so that
. For
to be satisfied, the BF-frame rotates by π about the z-axis. The rotation
is then
(38)
(38) and we have
, as required. The corresponding operation
changes the rigid rotors by the second line of Equation (Equation26
(26)
(26) ) and thus an irreducible representation for the subgroup
can be formed. In other words, we can use a product basis set with this BF-frame to symmetrise the rotation basis functions by parity (the subgroup
), but not of the full MS group
(M).
5.2. Bisector frame
Another obvious BF-frame choice for the ABA molecule is the y-axis bisecting the angle and the z-axis pointing out of the plane. The equivalent rotations for this frame were given in Equation (Equation26(26)
(26) ). There is a subtle difference, however. In that frame, the y-axis bisects ABA angle as this is the orientation of the principal axis system at vibrational equilibrium. In this case, the y-axis bisects the ABA angle for all geometries. The BF Cartesian coordinates in this frame are given by
(39)
(39) This time, when we apply the (12) operation, the matrix
given by
(40)
(40) has exactly the property that
, and likewise for
. In this case, we can build a symmetry adapted rotational basis set because the rotation angle is
and the axis of rotation is constant relative to the BF-frame for this operation.
For this frame, the vibrational G matrix is [Citation37] Equation (Equation33(33)
(33) ).
The rotational G matrix is
(41)
(41) The Coriolis G matrix is
(42)
(42) In this BF-frame, D = 0 for operation (12). One can again use Equation (Equation37
(37)
(37) ) to confirm the invariance of the G matrix.
5.3. Frame conditions
A more qualitative way to understand these results, which will prove important when studying the Eckart frames, is to examine the three conditions which define the BF-frame in the Sørensen approach. These conditions are, in general, functions of the BF Cartesian components which must be zero at all times. In the case of the bisector frame of the ABA molecule
(43)
(43) where the subscript refers to the atom the coordinate refers to. The first two conditions specify that the z-axis is perpendicular to the A1-B-A2 plane and the last that the y-axis bisects A1-B-A2 angle. It should be pointed out that these do not totally define the BF-frame orientation, as we can rotate the BF-frame by π about any of the BF-frame axes and the conditions would still hold. One must supplement these conditions with the BF Cartesian coordinates defined in terms of the vibrational coordinates. These implicitly assume the BF-frame the conditions define. In general, the conditions also do not define the handedness of the axes as one can change all the BF Cartesian coordinates
to
(i.e. make the axes left handed) and the conditions would still hold.
An MS operation applied to each condition relabels the BF Cartesian coordinates. The result is the transformed BF Cartesian coordinates in the original BF-frame. For example, for the (12) operation, they become
(44)
(44) which are actually still zero, a manifestation of the possible choice we had in the BF-frame that satisfies these conditions. We know that we must apply a π rotation about the z-axis, after which they become
(45)
(45) which are the original conditions and thus zero.
In the bond vector frame choice of the molecule, the conditions were
(46)
(46) which after a transformation of
and a rotation by π are
(47)
(47) The last condition is in general not zero.
5.4. Eckart frame
Approaching the problem from this point of view provides us with an alternative way to check if the BF-frame undergoes a constant rotation under MS operations. This is especially useful when the BF Cartesian coordinates as functions of the vibrational coordinates are not available analytically. In particular, we can check that the Eckart frame also satisfies the aforementioned rotation condition.
The conditions for the Eckart frame are defined as [Citation38]
(48)
(48) where the equilibrium frame is defined to be the principal axis system (PAS). The
are the BF Cartesian components of the nuclei at equilibrium and enter the condition as constants. As stated above, the PAS for the ABA molecule in vibrational equilibrium is the bisector. We thus have
(49)
(49) where
,
and
are of the form
(50)
(50) In particular, if
, then
and
. Once the operation (12) is applied, we have
(51)
(51) where we reiterate that the components
are in the original BF-frame. If we then rotate the frame the new conditions are
(52)
(52) where in the second line we applied
to the conditions and in the third line used the property that, if N is a rotation,
. The last line is N applied to the original conditions and therefore zero.
The operation is even easier and can be trivially shown to work as the operation changes the
to
while
. We have therefore shown that the Eckart frames are applicable for ABA molecules if one desires a symmetry-adapted rotational basis set.
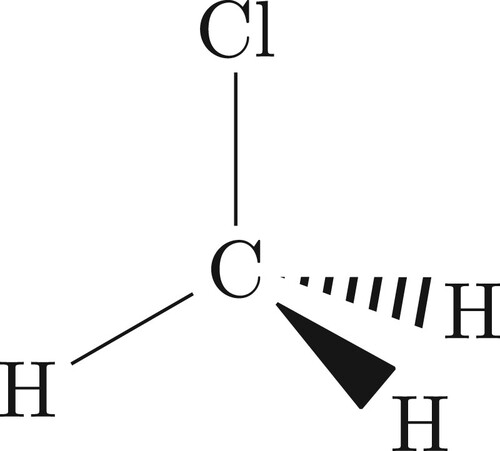
6. CH3Cl example
6.1. Eckart frame
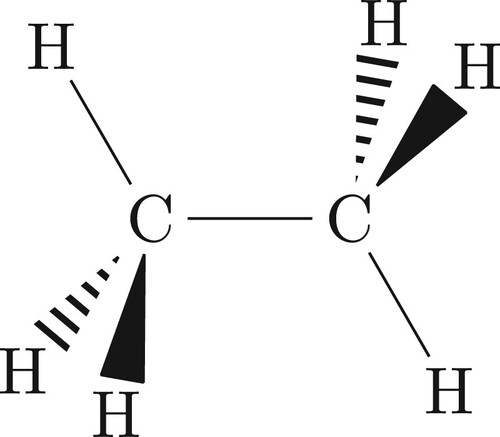
For ABA molecules, it was relatively straightforward to choose alternative (to Eckart) axes with which one could symmetrise the rotational basis functions. For other molecules the choice is not so simple. We introduce the example of CH3Cl (shown in Figure ) and again demonstrate the Eckart frame (whose equilibrium orientation is shown in Figure ) can be employed to build a symmetry-adapted rotational basis set and also describe an alternative and geometric frame which one can also use.
Figure 5. The equilibrium Eckart frame for CH3Cl. The z-axis is along the C–Cl bond and the x-axis in the plane formed by Cl–C–H.
The Eckart conditions for CH3Cl are
(53)
(53) where the PAS frame for the equilibrium geometry is shown in Figure . Because of this, both
and
only have a z component.
The MS group of CH3Cl is (M) and can be constructed from two generating operations (123) and
with equivalent rotations of
and
, respectively.
For the operation (123), we can relate the equilibrium coordinates as and
where
from Equation (Equation7
(7)
(7) ). The Eckart conditions can thus be can be expressed as
(54)
(54) Applying the operation (123), the Eckart conditions become
(55)
(55) As usual, we rotate the frame, in this case by
. This transforms
to
. Using the same procedure as for the ABA case we can rewrite this as
(56)
(56) where we used that A applied to the equilibrium coordinates of Cl or C does not change them. The final result is precisely the same as in the case of ABA.
For the operation , the rotation matrix is
. We can again relate the equilibrium coordinates by
and vice versa. Also,
and likewise for C and Cl. The Eckart conditions can thus be written
(57)
(57) The result after applying
is
(58)
(58) Rotating the frame by N, multiplying the conditions by
, and using the same steps as before, we have
(59)
(59) The technique used in this proof can be applied to many different molecules where the Eckart frame is used as it relied on relations between the equilibrium Cartesian coordinates of the molecule. In vibrational equilibrium, the Cartesian coordinates of the nuclei are related to each other by constant rotations. These rotations can be found, for example, through graphical means, which is the technique used to find the equivalent rotations explained in [Citation18]. Alternatively, although the form of the BF Cartesian coordinates as a function of vibrational coordinates for all geometries is not known, their equilibrium ‘structure’ (à la Equation (Equation50
(50)
(50) )) is. The required rotation for an MS operation can then be found by satisfying
, where
are the equilibrium BF Cartesian coordinates. These do not change under the vibrational coordinate transformation because the vibrational coordinates are effectively frozen. Since the matrix N should be the same regardless of the geometry, if it is found in vibrational equilibrium, it is found in general. As demonstrated above, if one uses the PAS system at the vibrational equilibrium to define the reference geometry, then the equivalent rotations are the correct rotations of the BF-frame undergoing MS operations.
6.2. Geometric CH3Cl frame
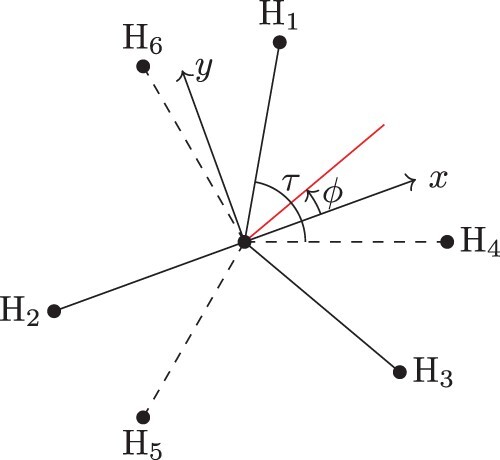
We introduce a frame definition which can be solved analytically. In this case, the z-axis always points parallel the C–Cl bond and the direction of the x-axis is defined by ϕ, given by
(60)
(60) where
is the dihedral angle from the plane formed by Cl–C–H
to the plane Cl–C–H
and likewise for
. This is illustrated in Figure . Had we decided to start from H2, the choice of x-axis direction (
) would be determined by
(61)
(61) where Figure illustrates this choice. A feature of these choices is the angle between the x-axes, as illustrated in Figure , can be found as follows:
(62)
(62) Thus the angle between them is always
, irrespective of the coordinates of the atoms. It is this property that allows the frame to be used when symmetrising the rotational basis set.
To demonstrate that, with this frame, the Cartesian coordinates transform correctly under MS operations if the BF-frame rotates by constant angles, let us first parametrise the vibrational coordinates by the Cl–C stretch (), the 3 C–H stretches (
to
), the three Cl–C–H bends (
to
), and the symmetrised coordinates defined by
(63)
(63) Note that
is the angle
.
The BF Cartesian coordinates in the bond vector frame are given by (with the origin at the C atom)
(64)
(64) Solving Equation (Equation63
(63)
(63) ) for the angles
and
by using the substitution
, we obtain
(65)
(65) which can be substituted into Equation (Equation64
(64)
(64) ). Applying the rotation matrix
(the matrix S of Equation (Equation90
(90)
(90) )) to the BF Cartesian coordinates and using the relations
and
, the final result is
(66)
(66) The operation (123) changes the stretches and angles in the usual manner and changes the dihedrals according to
(67)
(67) or
(68)
(68) The transformed coordinates are then
(69)
(69) These are in the new BF-frame. If we now consider them in the old BF-frame via the rotation matrix
(70)
(70) and apply the first form to
and
and the second form to
, we obtain the original coordinates but permuted by (123).
For the operation , the stretches and angles transform in an obvious way while the dihedrals transform as
(71)
(71) Performing this transformation and also applying the matrix
to the Cartesian coordinates, we obtain
(72)
(72) which are the original Cartesian coordinates with 2 and 3 permuted and then inverted.
7. C2H6 example
7.1. Sayvetz frame
The Sayvetz condition [Citation39] is an addition to the Eckart conditions for non-rigid molecules, defined by
(73)
(73) where now the equilibrium Cartesian coordinates
are functions of a non-rigid vibrational coordinate τ where the BF-frame is set to the PAS frame for every value of τ while all other coordinates are in equilibrium. The prime on
signifies that it is a derivative.
We shall prove these conditions work for the C2H6 molecule, shown in Figure . In the PAS (shown in Figure ), the x-axis bisects the dihedral formed by H–C–C and H
–C–C while the z-axis is parallel to the C–C bond. The origin is the middle of the C–C bond. The PAS coordinates of H1 and H4 are given by
(74)
(74) for some c and d. The coordinates of the other atoms are
(75)
(75) where
. The carbons only have a z-component and are constants. The Eckart conditions are thus given by
(76)
(76) and the Sayvetz condition is given by
(77)
(77) The MS group of C2H6 is
(EM) which has generators given by Table .
Figure 10. The equilibrium Sayvetz frame for C2H6. The z-axis is parallel to the C–C bond and the x-axis bisects the dihedral between the planes formed by C–C–H and C–C–H
.
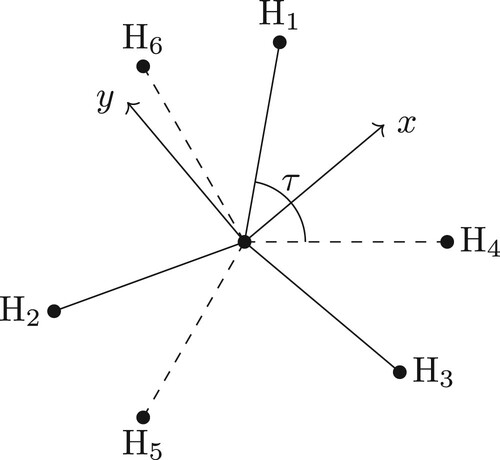
Table 5. The generators of the extended group (EM) of C2H6 and their effect on the torsional angle (τ) and the equivalent rotation of the generator [Citation32].
The operation (123)(456) changes the hydrogen labels in the usual way. However, due to the change in the τ angle, the PAS equilibrium coordinates change according to for hydrogens 1, 2 and 3 and
for hydrogens 4, 5 and 6. The carbons are unchanged. We thus have
(78)
(78) which is the original equation. The derivatives of the PAS equilibrium coordinates transform the same way under a change in τ and thus the Sayvetz condition is trivially satisfied.
For the operation (132)(456), the angle τ does not change, but the rotation is applied and thus the instantaneous Cartesian coordinates change by
. We thus have
(79)
(79) We apply
to the conditions and using the same technique as before, the result is
applied to the original conditions, and this is zero. For the Sayvetz condition, we use that
and pre-apply A to both terms of all dot products. The original condition is re-obtained.
The operation has no rotation and changes
and vice versa. The result is
(80)
(80) which is the original set of conditions and thus is zero. The Sayvetz condition is satisfied in the same way.
The element does not transform τ but has the equivalent rotation
. Thus, it transforms the instantaneous coordinates to
, where
, and changes the conditions to
(81)
(81) The matrix N changes the equilibrium Cartesian coordinates of the carbons by Na = −a and H1's by
. Also,
and
. Thus, in applying I = NN to the transformed condition, we have
(82)
(82) which is zero. The Sayvetz condition works the same way as in the previous operations.
Finally, the operation is peculiar. It is defined to change the torsional coordinate τ by
through a rotation of only one of the CH3 groups, ensuring the Cartesian coordinates in a non-rotating frame do not change. The Euler angle χ changes by
with an associated equivalent rotation
. Due to the τ change, the equilibrium as transform as
; the frame rotation changes the
s by
. Applying I = NN, where
, to the transformed condition then results in
applied to the original condition and is thus zero. The Sayvetz condition again works the same way.
7.2. Geometric C2H6 frame
As with the case of CH3Cl, we define a geometric frame which can be used to produce a symmetry-adapted rotational basis set. The valence coordinates used are for the C–C bond,
to
for the C–H bonds,
to
for the H–C–C angles, and the symmetrised dihedrals given by
(83)
(83) and, finally, the symmetrised torsional angle given by
(84)
(84)
The geometric frame has the x-axis pointing at an angle
(85)
(85) from the bisector between H
and H
. See Figure .
Figure 11. The geometric frame for C2H6. The z-axis is parallel to the C–C bond. The x-axis is at an angle from the bisector of the dihedral angle between the planes formed by C–C–H
and C–C–H
(colour online).
In the bisector frame, the Cartesian coordinates are given by, with the origin between and
(86)
(86) where the dihedral angle combination results from the Z-matrix definition of the Coordinates. The atoms of the Z-matrix are, in turn, a, b, 1, 4, 2, 6, 3 and 5. The un-symmetrised dihedral angles are defined by
(87)
(87) Solving the symmetrised Coordinates for the dihedrals in a manner similar to CH3Cl, we get
(88)
(88) Substituting this into Equation (Equation86
(86)
(86) ) and applying the rotation
, we obtain
(89)
(89) In Appendix 3 it is demonstrated that this frame can be used for rotational symmetrisation.
8. Transforming between BF-frames
As explored above, a bond vector BF-frame is not always suitable if one requires a symmetry-adapted rotational basis set. However, the KEO is quite straightforward to define and compute in this frame. In the Sørensen approach, for example, the three rotational conditions of Equation (Equation46(46)
(46) ) are very simple. In contrast, defining a condition in terms of
of Equation (Equation60
(60)
(60) ) is trickier and involves using trigonometric functions of the Cartesian coordinates rather than simple differences.
In this section, we shall explain how to find the angle in the case of CH3Cl and also how to transform the s-vector components defined in a bond vector BF-frame to the desired frame.
For a general symmetry operation, if , we required that
or
. We then stipulate that N is a constant matrix, for example
for CH3Cl. The Cartesian coordinates
are in the old BF-frame. In the new BF-frame, they will be of the form
for some rotation matrix S which is a function of the vibrational coordinates. We thus want
(90)
(90) for every operation. In the case of CH3Cl we see that the z-axis along the Cl–C bond should be maintained. Only a rotation about the z-axis is required. Since motion of the atoms in this plane is parametrised by the dihedral angles, we will assume the simplest rotation of the form
where a and b are constants. The position of H1 in this, yet, undetermined frame is given by
(91)
(91) while
is given by
(92)
(92) Thus we want to equate the transformed
to
, or
(93)
(93) from which we obtain
(94)
(94) For the operation
,
in this frame is given by
(95)
(95) and
is
(96)
(96) Equating the transformed Equation (Equation95
(95)
(95) ) to Equation (Equation96
(96)
(96) ), we have
(97)
(97) From these we obtain a = 0 and
as stated previously.
For the second step, that of transforming the s-vectors to the new BF-frame, first one starts with the original BF vibrational and rotational s-vector components and
, respectively. It can be shown that the vibrational s-vector components transform to
(98)
(98) where S is the matrix which transformed the Cartesian coordinates.
The rotational s-vector components can be written as a matrix
where g signifies the row. It then transforms as
(99)
(99) where
is defined by
(100)
(100) For multiple rotations, these steps are applied sequentially.
These results are a generalisation of the results [Citation40] for rotations which are not linear functions of the vibrational coordinates, and also a transform of the s-vectors rather than the G matrix. This enables their form to be simplified further with software which performs analytic computations on the smaller s-vectors compared to the G matrix. It is ideal to calculate the vibrational G matrix and the pseudo-potential with the original s-vectors as these do not depend on the choice of frame. The Coriolis and rotational G matrix must be calculated with the new frame.
For the t-vector approach, if and
are the original BF vibrational and rotational t-vector components, respectively, then the transformed vibrational t-vector components in the new BF-frame are
(101)
(101) and the transformed rotational t-vectors are
(102)
(102) where
is the matrix formed from
, where g signifies the column.
9. Further applications
In the above sections, the focus was on valence coordinates as these are the simplest to define and use to determine the appropriate frames. However, this does not preclude the use of other coordinates. For example, the Jacobi [Citation41] and Radau [Citation42] coordinates offer alternative parametrisations. These have advantages due to the simplification of the Hamiltonian.
It is feasible that a similar procedure could be applied for these coordinates for rotational basis symmetrisation. Indeed, the s-vector approach stipulates that the vibrational coordinates are independent of the translation and rotation of the molecule. It does not specify the form of the coordinates. For CH3F, for example, the kinetic energy operator was derived in [Citation43] where the canonical point was the centre of mass of the CH3 group and the vectors were the distances from that point to the hydrogen and fluorine nuclei. The frame chosen was the bond vector using vectors from the canonical point to two of the hydrogens. Using vibrational coordinates analogous to the valence ones for CH3Cl of Section 6.2, it is clear that the structure of the Cartesian coordinates as a function of the vibrational is identical, and hence the same process should work.
The size of the Coriolis coupling was not considered in this work and was beyond its scope, but it is arguably the most important factor when comparing these frames. Previously research into this has been undertaken; see, for example, Sarka et al.[Citation44] who compared the Radau bisector, the valence bisector, and the Eckart frame for symmetric triatomics and found that the Radau bisector is superior in reducing this coupling and should lead to better ro-vibrational separation. Comparing the frames described in this publication is a possible avenue for further research.
Moving beyond the molecules chosen, the question of the possibility of finding non-Eckart/Sayvetz frames where rotational symmetrisation is possible still remains. For certain molecules, such as ammonia, a trisector can define the z-axis. Then, once again, two symmetrised coordinates based on the dihedrals could be used to rotate about this z-axis as the structure is the same as the CH3Cl molecule. For other molecules, such as CH4, it is remarkably more challenging. For Equation (Equation90(90)
(90) ), one can replace
with
, where B is the rotation matrix relating the old and rotated bond vector frame (i.e. it is the matrix N of Equation (Equation13
(13)
(13) ) for the bond vector frame rather than the desired frame). Then, the equation that must be solved is
(103)
(103) for each generator. For CH4, the matrix S would be parametrised by five symmetrised angles and the coefficient values could only be found numerically, if they could be found at all. In general, finding the frame for a given molecular structure and coordinate choice would have to be done on a case-by-case basis.
We aim to use the results in this study on the ro-vibrational calculations of C2H6. An analytic KEO in valence coordinates and using this frame has been derived. Moreover, we have a Taylor expanded KEO based on linearised coordinates in the Sayvetz frame. A comparison of the two approaches would be instructive.
10. Conclusion
The main result in this study was the determination of geometric BF-frames for a certain classes of molecules – with the CH3Cl and C2H6 molecules used as defining examples – where rotational symmetrisation is possible. The Eckart and Sayvetz frame were also shown to be suitable for these molecules, with the form of the proofs extendable to other molecules as they relied on the correspondence between the MS group of the molecule and the point group of the molecule when in vibrational equilibrium.
Furthermore, one approach to find such frames is described for CH3Cl, and a method to check frames is also given, provided one has the BF Cartesian coordinates as a function of the vibrational coordinates. Finally, a simple transformation of the s-vectors due to a change of frame is stated, allowing them to be calculated in a straightforwardly defined frame first and then transformed to another, more appropriate, frame.
Acknowledgments
T. Mellor would like to thank S. Yurchenko and J. Buldyvera for their suggested improvements in the structure of the manuscript.
Disclosure statement
No potential conflict of interest was reported by the author.
References
- V. Szalay, J. Chem. Phys. 140 (23), 234107 (2014). doi:10.1063/1.4883195
- D. Lauvergnat, J.M. Luis, B. Kirtman, H. Reis and A. Nauts, J. Chem. Phys. 144 (8), 084116 (2016). doi:10.1063/1.4942172
- K. Sadri, D. Lauvergnat, F. Gatti and H. Meyer, J. Chem. Phys. 141 (11), 114101 (2014). doi:10.1063/1.4895557
- S.N. Yurchenko, M. Carvajal, H. Jensen, P. Lin, J. Zheng and W. Thiel, Mol. Phys. 103 (2-3), 359–378 (2005). doi:10.1080/002689705412331517255
- E. Mátyus, J. Šimunek and A.G. Császár, J. Chem. Phys. 131 (7), 074106 (2009). doi:10.1063/1.3187528
- A.E. Protasevich and A.V. Nikitin, Atmos. Ocean Opt. 35 (1), 14–18 (2022). doi:10.1134/S1024856022010110
- F. Gatti and C. Iung, Phys. Rep. 484 (1), 1–69 (2009). doi:10.1016/j.physrep.2009.05.003
- X. Chapuisat and C. Iung, Phys. Rev. A 45, 6217–6235 (1992). doi:10.1103/PhysRevA.45.6217
- A.V. Nikitin, M. Rey and V.G. Tyuterev, J. Chem. Phys. 142 (9), 094118 (2015). doi:10.1063/1.4913520
- C. Fábri, M. Quack and A.G. Császár, J. Chem. Phys. 147 (13), 134101 (2017). doi:10.1063/1.4990297
- C. Fábri, E. Mátyus and A.G. Császár, J. Chem. Phys. 134 (7), 074105 (2011). doi:10.1063/1.3533950
- X. Wang and T. Carrington, J. Chem. Phys. 123 (15), 154303 (2005). doi:10.1063/1.2043148
- B.T. Sutcliffe and J. Tennyson, Intern. J. Quantum Chem. 39 (2), 183–196 (1991).doi:10.1002/qua.560390208.
- S.M. Colwell and N.C. Handy, Mol. Phys. 92 (2), 317–330 (1997). doi:10.1080/002689797170527
- M.J. Bramley, W.H. Green Jr. and N.C. Handy, Mol. Phys. 73 (6), 1183–1208 (1991). doi:10.1080/00268979100101871
- M. Tinkham and G. MacKay, Group Theory and Quantum Mechanics (Dover, 2003).
- H.C. Longuet-Higgins, Mol. Phys. 6 (5), 445–460 (1963). doi:10.1080/00268976300100501
- P.R. Bunker and P. Jensen, Molecular Symmetry and Spectroscopy (2nd ed.. NRC Research Press, Ottawa, 1998).
- X. Wang and T. Carrington, J. Chem. Phys. 138 (10), 104106 (2013). doi:10.1063/1.4793474
- C. Fábri, E. Mátyus and A.G. Császár, Spectrochim. Acta A Mol. Biomol. Spectrosc.119, 84–89 (2014). doi:10.1016/j.saa.2013.03.090
- S.N. Yurchenko, R.J. Barber, A. Yachmenev, W. Thiel, P. Jensen and J. Tennyson, J. Phys. Chem. A 113 (43), 11845–11855 (2009). doi:10.1021/jp9029425
- A.I. Pavlyuchko, S.N. Yurchenko and J. Tennyson, J. Chem. Phys. 142 (9), 094309 (2015). doi:10.1063/1.4913741
- A.F. Al-Refaie, A. Yachmenev, J. Tennyson and S.N. Yurchenko, Mon. Not. R. Astron. Soc. 448 (2), 1704–1714 (2015). doi:10.1093/mnras/stv091
- A.F. Al-Refaie, R.I. Ovsyannikov, O.L. Polyansky, S.N. Yurchenko and J. Tennyson, J. Chem. Phys. 318, 84–90 (2015). doi:10.1016/j.jms.2015.10.004.
- C. Fábri, E. Mátyus, T. Furtenbacher, L. Nemes, B. Mihály, T. Zoltáni and A.G. Császár, J. Chem. Phys. 135 (9), 094307 (2011). doi:10.1063/1.3625404
- X. Wang and T. Carrington, J. Chem. Phys. 119 (1), 94–100 (2003). doi:10.1063/1.1559479
- H. Wei and T.T. Carrington, J. Chem. Phys. 107 (8), 2813–2818 (1997). doi:10.1063/1.474639
- H. Wei, J. Chem. Phys. 118 (16), 7202–7207 (2003). doi:10.1063/1.1562624
- G.O. Sørensen, A new approach to the hamiltonian of nonrigid molecules, in Large Amplitude Motion in Molecules II, edited by M. J. S. Dewar et al., Vol. 82, of Topics in Current Chemistry, pp. 97–175 (Springer Berlin Heidelberg, Heidelberg, 1979).
- H. Goldstein, C. Poole and J. Safko, Classical Mechanics (Addison Wesley, 2002).
- X. Wang and T. Carrington, J. Chem. Phys. 146 (10), 104105 (2017). doi:10.1063/1.4977179
- T.M. Mellor, S.N. Yurchenko, B.P. Mant and P. Jensen, Symmetry 11 (7), 862 (2019). doi:10.3390/sym11070862.
- E. Mátyus, G. Czakó and A.G. Császár, J. Chem. Phys. 130 (13), 134112 (2009). doi:10.1063/1.3076742
- A. Nauts and X. Chapuisat, Mol. Phys. 55 (6), 1287–1318 (1985). doi:10.1080/00268978500102031
- J.K.G. Watson, J. Mol. Spectrosc. 228 (2), 645–658 (2004). doi:10.1016/j.jms.2004.07.003 Special Issue Dedicated to Dr. Jon T. Hougen on the Occasion of His 68th Birthday.
- S.N. Yurchenko, A. Yachmenev and R.I. Ovsyannikov, J. Chem. Theory Comput. 13 (9), 4368–4381 (2017). doi:10.1021/acs.jctc.7b00506
- S.N. Yurchenko and T.M. Mellor, J. Chem. Phys. 153 (15), 154106 (2020). doi:10.1063/5.0019546
- C. Eckart, Phys. Rev. 47, 552–558 (1935). doi:10.1103/PhysRev.47.552
- A. Sayvetz, J. Chem. Phys. 7 (6), 383–389 (1939). doi:10.1063/1.1750455
- K.L. Mardis and E.L. Sibert III, J. Chem. Phys. 106 (16), 6618–6621 (1997). doi:10.1063/1.473658
- V Aquilanti and S Cavalli, J. Chem. Phys. 85, 1355–1361 (1986). doi:10.1063/1.451223
- B.R. Johnson and W.P. Reinhardt, J. Chem. Phys. 85 (8), 4538–4556 (1986). doi:10.1063/1.451775
- Z. Zhao, J. Chen, D.H. Zhang, Z. Zhang, D. Lauvergnat and F. Gatti, J. Chem. Phys. 144 (20), 204302 (2016). doi:10.1063/1.4950028
- J. Sarka, B. Poirier, V. Szalay and A.G. Császár, Spectra Chimica Acta A 250, 119164 (2021). doi:10.1016/j.saa.2020.119164
Appendices
Appendix 1 Vibrational s-vector components
In this section we demonstrate that the components of the vibrational s-vectors in any frame are found by evaluating in the Cartesian coordinates of that frame. To show this, we use the property that the value of
does not depend on which frame it is evaluated in, i.e.
(A1)
(A1) where
are the coordinates of
when the frame is rotated and translated. Calling
, we want to determine
in terms of
. Let us define the function
as
(A2)
(A2) Then, the translational and rotational invariance can be expressed as
(A3)
(A3) Differentiating both sides, and using the chain rule, we obtain
(A4)
(A4)
(A5)
(A5) Here
is the
component of the derivative of
, which is non-zero only when
and is given by
. We thus have
(A6)
(A6) Thus
(A7)
(A7) where
. Since the Cartesian components in the new frame are given by
,
are the unit vectors in the same frame, which tells us the component of
is the derivative of
evaluated at the Cartesian coordinates of that frame. This gives a computational way to determine the components of
in any frame that does not involve first evaluating the components in the RF-frame and applying the rotation matrix.
For the transformed s-vector, we have that
(A8)
(A8) as
are the coordinates of atom α after the MS operation is performed and in the new BF-frame. As we have seen, this is given by
, so that to transform the BF components
we simply perform the vibrational coordinate transformation (which we shall denote as
). This will give us the transformed s-vector components in the new BF-frame.
Appendix 2
Kinetic energy operator invariance
If and
then the KEO (without the pseudo-potential) transforms as
(A9)
(A9) Terms under-braced with the same number cancel. What remains when all is summed together is the original Hamiltonian, showing invariance.
The pseudo-potential U consists of the four terms
(A10)
(A10) Table provides the s-vector transforms. One can show that the BF-frame unit vectors transform as
while the derivatives of the s-vectors transform as, if
and
,
(A11)
(A11) where
and
is z-axis vector of the BF-frame. We assume a rotation N of the first column of Table . Due to this, the term containing
.
With these expressions, we shall express the transformation of the pseudo-potential terms (temporarily ignoring the summation over the masses) by subscripting each U term with the atom it corresponds to. For , it is given by
(A12)
(A12) For
, it is
(A13)
(A13) where in the second line we use
. For
, we have
(A14)
(A14) Finally, for
, we have
(A15)
(A15) The first term of each of these corresponds to the original U elements, so summing over them as in Equation (EquationA10
(A10)
(A10) ), we obtain the original U. Thus we must show the other terms sum to zero. The difference between the transformed and original terms,
, are given by
(A16)
(A16) The terms under-braced by the same number cancel each other. Thus we must show the remaining terms cancel. We have
(A17)
(A17) The last line exactly cancels the remaining two terms.
Appendix 3
Geometric C2H6 frame proof
For simplicity we shall drop the bond and angle contributions of the hydrogens since they transform trivially.
The first operation (123)(456) transforms the dihedrals as
(A18)
(A18) where
represents the pairs
and
. The torsional angle transforms as Table . With these transformations, the (abridged) transformed BF Cartesian coordinates are
(A19)
(A19) which are precisely the original BF Cartesian coordinates permuted according to (123)(456). There is no rotation.
For the operation (132)(456), the dihedrals transform as the transpose of Equation (EquationA18
(A18)
(A18) ) and the torsional angle does not change. The transformed Cartesian coordinates are
(A20)
(A20) With this construction of the Cartesian coordinates, the corresponding BF-frame rotation is
. Thus we have
(A21)
(A21) Applying the first form to H1, H3, H4, and H5, and the second form to H2 and H6, we recover the transformed Cartesian coordinates in the original BF-frame.
The operation changes the dihedrals according to
(A22)
(A22) and likewise for
. The torsional angle changes as
. The result is
(A23)
(A23) which are exactly the original BF Cartesian coordinates after the operation
is applied, which means no rotation is required.
Next, the operation transforms the dihedrals as
(A24)
(A24) and likewise for
and does not change the torsional angle. With the BF-frame rotation of
applied, the transformed Cartesian coordinates in the original frame are
(A25)
(A25) which is the desired permutation.
Finally, the operation is not an MS operation. The nuclei should not be permuted or inverted. With
, the x and y components of all the nuclei become negative, while the rotation
transforms them back, so the operation has no effect on the Cartesian coordinates, as desired.