ABSTRACT
Objective
Generally, to map epidemiological and demographic features of patients with traumatic brain injury (TBI) in Umeå county, Sweden. Specifically, to evaluate the subjects needing a computed tomography (CT) of the head after suffering from TBI and frequencies of 1) intracranial lesions detected with CT, 2) need for neurosurgical intervention and 3) admission to hospital.
Methods
Patients with a suspected TBI, undergoing CT within 24 hours of arrival to hospital, were included in a database for evaluation.
Results
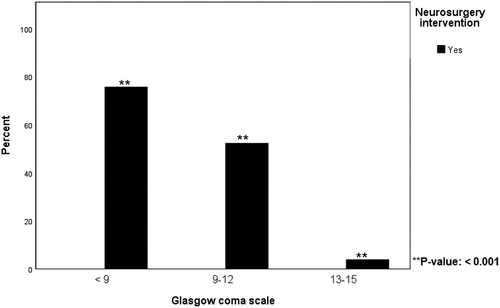
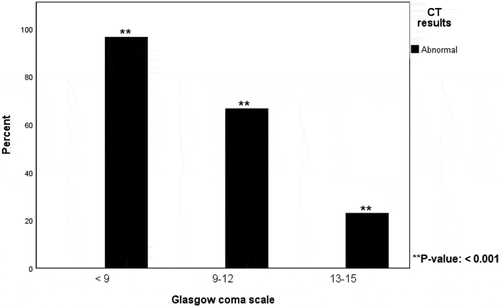
Out of 302 patients (63% male), 83% were GCS 13–15, 7% were GCS 9–12 and 10% were GCS <9. The frequency of abnormal CT findings was 23% in GCS 13–15, 67% in GCS 9–12 and 97% in GCS <9. Neurosurgical intervention was needed by 4% of those with GCS 13–15, 52% of those with GCS 9–12 and by 76% of those with GCS <9.
Conclusions
Subjects with GCS 13–15 had higher frequencies of abnormal CT findings, need for neurosurgical intervention and hospital admission than previously reported. A similar trend was observed for patients with GCS 9–12, which can be of serious nature, with a higher frequency of need for neurosurgical intervention than previously described.
Introduction
Traumatic brain injury (TBI) is a common cause of death and disability worldwide. In Europe, about 2.5 million people suffer from TBI every year, 1 million need hospitalization and 75,000 people die (Citation1,Citation2). In Sweden, the annual incidence of TBI is approximately 300 cases/100,000 citizens with roughly 10 deaths/100,000 citizens (Citation3). These figures are comparable to the European and American averages (Citation4,Citation5).
In the younger population, the most common cause of TBI is traffic-related injury, while fall injuries dominate in the elder population. TBI occurs twice as often in males as in females, but this gender difference declines with age (Citation4,Citation6–12).
The most commonly used classification of TBI is based on the Glasgow Coma Scale score (GCS) which is used at clinical examination and takes eye opening, verbal response and motoric reaction into account. GCS 13–15 corresponds to mild injury, 9–12 to moderate injury and < 9 to severe injury (Citation13). The risk of mortality increases with lower GCS. Severe TBI carries a mortality rate up to 40%, while the rate in mild TBI is 0.1% (Citation14,Citation15). For the past few decades, the research regarding TBI management has focused on severe TBI. However, severe TBIs make up only a few percent of all TBIs. A patient in the emergency room (ER) will usually have a mild (90%) or moderate (5%) TBI (Citation11,Citation16).
There is a dilemma regarding best practices on to perform a CT scan or hospitalize patients. Most patients will not develop any life-threatening consequences, but it is of the utmost importance to identify the minority who are at risk. There is no consensus globally on how to manage this matter (Citation8). Though there has been a positive influx of clinical strategies and guidelines in recent years, they are all based on an “one size fits all” approach and lack scope for personalization (Citation17–22). Furthermore, compliance with these guidelines and clinical strategies varies greatly between different hospitals and across the globe (Citation8).
For the development of new and current guidelines, the true incidence of consequences following mild and moderate TBI must be elucidated. The definitions of mild and moderate TBI may vary depending on the methods used, e.g., GCS, loss of consciousness or posttraumatic amnesia, which may complicate the comparison of studies. The overall aim of this study was to evaluate epidemiological and demographic features of cases with TBI with a CT scan of the head within 24 hours of arrival at the ER in a defined population in Umeå county. The specific aims of this study were to: 1) evaluate the incidence rates of intracranial lesions detected on CT in mild, moderate and severe TBI, 2) evaluate the need for neurosurgical intervention in mild, moderate and severe TBI, 3) evaluate the number of hospital admissions following mild, moderate and severe TBI.
Methods
Design and study population
This study was a longitudinal prospective observational consecutive cohort study of patients suffering mild to severe TBI. Between January 1, 2015 and December 31, 2016, data were sourced on patients who came to the ER of Umeå University Hospital (NUS) with a suspected TBI examined with CT during acute management. The catchment area was the city of Umeå, with a few exceptions. All patients with suspected TBI suffered an event of clear external mechanical force to the head within at most 24 hours before arrival. The CT examination had to be performed at most 24 hours after arrival.
Clinical and radiological assessment
In all cases, the clinical, demographic and epidemiological data were collected prospectively. The initial management of the TBI at the ER was based on the Scandinavian Neurotrauma Committee (SNC) guidelines on management of TBI in adults (Citation22) and children (Citation23). Only patients with a CT scan within 24 hours of arrival at the ER were included. S-100B was not considered as an inclusion criterion. The decision to perform a CT scan was made by the ER physician and thus reflected everyday clinical practice. Severe TBI was treated in accordance with a modified Lund concept (Citation24). Radiological assessment was performed by neuroradiologists. CT images were reviewed by at least two medical specialists, one being a senior doctor. Mortality was assessed at discharge from the hospital or ER.
Definition of variables
Mild TBI was defined as GCS 13–15, moderate as 9–12 and severe < 9, as evaluated at the ER at admission. As the mild group, defined as GCS 13–15, can be negatively influenced by the worst affected, GCS 13, we also report the results for the GCS 14–15 group and compare these groups with each other. CT results were assessed as abnormal if there was any acute intracranial injury or evidence thereof: compressed basal cisterns, 0–4 mm, > 5 mm, > 10 mm midline shifts, epidural hematoma (EDH), acute subdural hematoma (ASDH), traumatic subarachnoid hemorrhage (tSAH) and/or contusion. If there was an already known abnormality on the CT scan, the result was classified as normal. Hospital admission was defined as admission to either a ward or an intensive care unit. Need for neurosurgical intervention was defined as need for craniotomy, craniectomy, ventricular drainage or insertion of an intraparenchymal pressure (ICP) monitoring device. Each patient could receive several interventions. This was counted as one intervention. Injury mechanism was divided into road traffic accident (RTA), fall accident, assault, sport/recreational and self-harm. RTA included all types of high-energy violence associated with traffic, e.g., car and motorcycle accidents affecting driver, occupant, opposing driver, pedestrian or cyclist. Snowmobile accidents were also included in this category. Fall accidents included falls from both standing and height. Injury type was divided into penetrating, blunt/closed and crush. Place injury occurred was divided into home, street/traffic, work, public place and sports field/hall. The patients were divided into age groups: < 18, 18–29, 30–49, 50–64 and 65–99 years.
Statistical analysis
Statistical analysis was performed using the software Statistical Package for the Social Sciences (IBM SPSS, v. 25). Qualitive variables were presented as frequencies or percentages. Statistical significance was evaluated using the chi-squared test (p<0.001).
Ethical considerations
All data have been anonymized. No future detriment to the individuals can be identified. Ethical approval has been granted for research of human subjects, by the Regional Ethical Board at Umeå University Hospital (dnr 2014/1473-31/4).
Results
General, demographic and epidemiological data
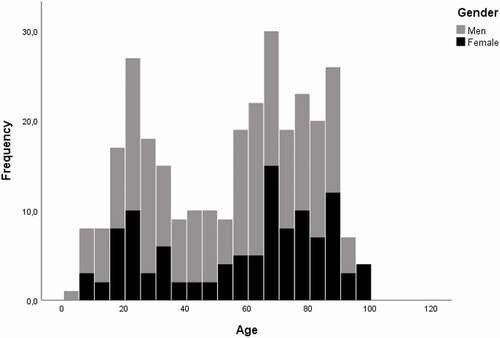
In all, approximately 2,000 patients with TBI seek care at the NUS ER annually. Most have mild TBI (GCS 13–15), with few clinical symptoms. During the study period (2015–2016), 302 patients fulfilled the inclusion criteria of having a CT scan of the head within 24 hours after arrival to the ER. In , demographic, epidemiological and clinical characteristics are presented. In total, 83% (n = 252) suffered from mild injuries, 7% (n = 21) from moderate injuries and 10% (n = 29) from severe injuries. shows the age distribution of the total study group.
Table 1. Characteristics of the study cohort
The most common injury type was blunt/closed, followed by crush. Crush injury was more common in younger ages, while blunt/closed injuries were more often observed among the elderly. The most common injury mechanism was fall accidents, followed by RTA. The highest percentages of fall accidents occurred in the older age groups and the highest percentages of RTA were found in the younger age groups. The most common place injuries occurred was at home, followed by street/traffic. Injury at home was more common in older ages.
CT results, neurosurgical intervention and hospital admission
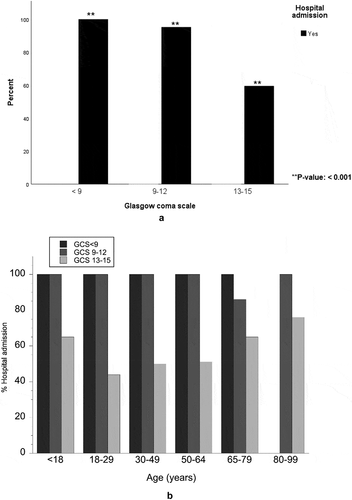
shows the frequencies of abnormal CT findings grouped by head injury severity. The frequency was 23%, 67% and 97% in mild, moderate and severe TBI, respectively, i.e., the frequency of abnormal CT findings increased with lower GCS. The types of neurosurgical intervention needed are described in . shows the characteristics of the CT results. Twenty-two percent of the total number of patients with an abnormal CT had a compressed basal cistern and fifty-six percent had a tSAH. Isolated tSAH was only seen in 17 patients, all within GCS 13–15. Thirty-five percent had a midline shift of > 0 mm. There was almost no difference between the GCS 13–15 and 14–15 as shown in , and definitely no significant difference. shows the proportion of subjects who had to undergo a neurosurgical intervention. presents the frequency of hospital admissions caused by each severity of TBI. shows the same outcome but grouped by age and severity of TBI. Admittance was nearly 100% for moderate and severe TBI, in all age groups. An admission frequency of approximately 65% was seen among children and elderly with mild TBI.
Table 2. Characteristics of the required neurosurgical interventions
Table 3. Characteristics of the CT results
Figure 2. The percentage of patients with abnormal CT-results within each severity of TBI. The difference between each group was statistically significant using chi-squared test.
Discussion
The study has analyzed all patients suffering from mild, moderate and severe TBI, on whom CT was performed at the NUS ER within 24 hours after arrival, during a period of two years. The total annual number of patients with TBI seeking care at the NUS ER was approximately 2,000. A similar selected cohort, prospectively and consecutively measured, has not to our knowledge previously been reported from our region. Overall, we demonstrate that in such a selected cohort the incidence of pathological CT-findings of the brain in the group of GCS ,13–15 and 9–12 is not negligible. Further, acute brain injuries defined at the ER as mild or moderate are not always that simple.
Overall aim
Our findings were consistent with those of earlier studies performed in Sweden, as well as internationally. Demographically, there are more males across all severities of TBI, there is a dominance of males in younger age groups and the difference in gender distribution of cases with TBI decreases with age (Citation4,Citation5,Citation11,Citation12,Citation25). Epidemiologically, road traffic accidents are more common in young ages and fall accidents are more common among elderly. Males, especially in young ages, are more likely to suffer severe head injury (Citation4,Citation5,Citation11,Citation12). As regards the distribution of different severities presenting at the ER, our findings diverged from those of others. The frequency of mild TBI was low, at 83%, and the occurrence rates of moderate and severe TBI (7% and 10%, respectively) were high (Citation5,Citation11–13,Citation25,Citation26). It cannot be ruled out that more severe TBI might be more common in the northern part of Sweden. In theory, more vehicle-associated injuries (car and bicycle accidents) could occur because of poor local road conditions during the long winter and more high-energy trauma could result from snowmobile accidents (Citation27,Citation28). It is important to note that the reported cohort did not include referrals from the entire neurosurgical catchment area for the hospital, but only patients with TBI who arrived at the ER and fulfilled the inclusion criteria.
CT results and neurosurgical intervention
Among patients suffering mild TBI (GCS 13–15), 23% had abnormal CT results and 4% needed neurosurgical intervention. These results are at the higher end of what has been reported previously. Rates of abnormal CT findings have been reported in the range 8.5–16% (Citation14,Citation18,Citation29–31). Need for neurosurgical intervention has been reported in 0.4–8.8% of cases (Citation14,Citation31–33). The differences might be due to the fact that we used the most common clinical definition of mild TBI, GCS 13–15, used by over 60% of neurotrauma centers worldwide (Citation33), while many earlier studies have used GCS 14–15 or only GCS 15 as the definition of mild TBI (Citation14,Citation18,Citation29). As GCS 13 subjects may be clearly worse injured compared to the rest in the mild traumatic injury group (GCS 13–15), the overall results of the mild group may be biased or driven by the GCS 13 subjects (Citation34,Citation35). We found no such effect in our study. However, it is worthwhile to recognize this in future studies and report these groups separately for comparison.
In moderate TBI, the frequency of abnormal CT findings was 67%, with a need for neurosurgical intervention in 52% of cases. The observed abnormality rate was consistent with earlier studies, where rates of 61–85% have been reported (Citation13,Citation29,Citation36,Citation37). However, the need for intervention was significantly higher than earlier reports of 12–18% (Citation20,Citation37,Citation38). This could also be explained by methodological differences. Several past studies have used the definition of moderate TBI as GCS 9–13, which would include some TBI categorized as mild in this study, thus presumably giving a better outcome. One should also have in mind that we report all neurosurgical interventions as a group which may render in higher numbers. However, in previously reported results it is not always clear how neurosurgical interventions are defined.
Hospital admission and cost
In the study, admission rates to hospital for mild, moderate and severe TBI were 60%, 95% and 100%, respectively. The high admission rate of more severe and moderate head injury is correct management under SNC guidelines (Citation22,Citation23). However, the high percentage of admission of mild TBI is not. We found no comparable studies reporting the percentage of patients needing hospital admission using the SNC 2013 guidelines. There is a meta-analysis reporting hospital admission to be necessary for 8% of patients with mild TBI (Citation14). Again, methodological differences must be considered when comparing these numbers. The lower admission rate is based on mild TBI defined as GCS 15 and does not exclude patients on the basis of no CT scan having been performed. Thus, the population studied herein was more specific, having a clinical indication for CT, which would suggest a more severe type of TBI and could explain our higher numbers. Another possible explanation for the high admission rate is that patients admitted to NUS had traveled long distances and lived alone. Admission is sometimes justified on a practical or moral basis. When these results for mild and moderate TBI are taken together, it seems obvious that the triage of patients at the ER is focused on correct indicators for performing CT of the brain and admission for patient care.
Every effort to correctly select patients for radiological evaluation and hospital admission is important, as the hospital cost for this patient group is very high. There is also a need for decreasing unnecessary radiological examinations to decrease the burden of radiation to the subject. Though the individual impact is less in mild TBI than in severe TBI, the group as a whole has significant impact on the global burden of disability and generates high costs for health care and society (Citation1,Citation39). When all severities are included, TBI costs the global economy up to $US400 billion annually (Citation1). There has been no global estimates on the impact of mild TBI alone. However, a population-based study from New Zealand reported that, for each new case with TBI, the cost over a lifetime was around $US5,000 for mild TBI compared with approximately $US40,000 for moderate/severe TBI (Citation39). However, as 95% of cases were mild TBI, they added up to three times the cost of the cases with moderate and severe TBI.
Strengths and limitations
The method used in this study has several strengths. First, this patient population has never been explored in a prospective manner before, only retrospectively (Citation5,Citation40). By using a prospective approach, we eliminated the risk of recall bias, which is always present in retrospective studies. Second, our inclusion criteria – requiring a performed CT examination and a clear mechanism of TBI – resulted in an interesting patient population. Third, we made no exclusions based on the age of the subjects. Therefore, we gathered a large, randomized patient material, with high generalizability, including representatives from all age groups. This study suffers from a few limitations. First, we need to review our inclusion criteria. The patients who lacked clinical indications for a CT examination were excluded. This may have disqualified the mildest forms of TBI, creating a selection bias toward more severe types of mild TBI and moderate TBI. This would explain our higher frequencies of interventions and hospital admissions. However, this indicates correct triage of patients. Second, medication with anticoagulants and other risk factors was not considered. This could influence the outcome. Future studies need to be conducted to explore these interesting aspects further.
Conclusion
In a defined population in Umeå county, we have strengthened several international findings on epidemiological and demographic features of TBI. The specific findings of this study suggest that mild TBI might not be as mild as previously reported. Rather, mild TBI might sometimes be of a more severe nature, resulting in a greater need for CT scans, neurosurgical intervention and hospital admission. Further, moderate TBI may require neurosurgical intervention more frequently than has been described previously. These findings should be considered in future development of guidelines for the management of TBI.
Disclosure statement
No potential conflict of interest was reported by the author(s).
References
- Maas AIR, Menon DK, Adelson PD, Andelic N, Bell MJ, and Belli A, Bragge P, Brazinova A, Buki A Chesnut RM, et al. Traumatic brain injury: integrated approaches to improve prevention, clinical care, and research. Lancet Neurol. 2017;16:987–1048. doi:https://doi.org/10.1016/S1474-4422(17)30371-X.
- Maas AI, Menon DK, Steyerberg EW, Citerio G, Lecky F, Manley GT, Hill S, Legrand V, Sorgner A. Collaborative european neurotrauma effectiveness research in traumatic brain injury (CENTER-TBI): a prospective longitudinal observational study. Neurosurgery. 2015;76(1):67–80. doi:https://doi.org/10.1227/NEU.0000000000000575.
- Socialstyrelsen: The national board of health and welfare. Statistikdatabas. Stockholm: Socialstyrelsen. [ accessed 2019 February 10] https://www.socialstyrelsen.se/statistik/statistikdatabas.
- Kleiven S, Peloso PM, von Holst H. The epidemiology of head injuries in Sweden from 1987 to 2000. Inj Control Saf Promot. 2003;10(3):173–80. doi:https://doi.org/10.1076/icsp.10.3.173.14552.
- Styrke J, Stalnacke BM, Sojka P, Bjornstig U. Traumatic brain injuries in a well-defined population: epidemiological aspects and severity. J Neurotrauma. 2007;24(9):1425–36. doi:https://doi.org/10.1089/neu.2007.0266.
- Brazinova A, Rehorcikova V, Taylor MS, Buckova V, Majdan M, and Psota M, Peeters W, Feigin V, Theadom A, Holkovic L, et al. Epidemiology of traumatic brain injury in europe: a living systematic review. J Neurotrauma. 2021;38(10):1411–1440. doi: https://doi.org/10.1089/neu.2015.4126.
- Chan V, Zagorski B, Parsons D, Colantonio A. Older adults with acquired brain injury: a population based study. BMC Geriatr. 2013;13(1):97. doi:https://doi.org/10.1186/1471-2318-13-97.
- Coronado VG, Thomas KE, Sattin RW, Johnson RL. The CDC traumatic brain injury surveillance system: characteristics of persons aged 65 years and older hospitalized with a TBI. J Head Trauma Rehabil. 2005;20(3):215–28. doi:https://doi.org/10.1097/00001199-200505000-00005.
- Koskinen S, Alaranta H. Traumatic brain injury in Finland 1991-2005: a nationwide register study of hospitalized and fatal TBI. Brain Inj. 2008;22(3):205–14. doi:https://doi.org/10.1080/02699050801938975.
- Majdan M, Plancikova D, Brazinova A, Rusnak M, Nieboer D, and Feigin V, Maas A. Epidemiology of traumatic brain injuries in Europe: a cross-sectional analysis. Lancet Public Health. 2016;1:e76–e83. doi:https://doi.org/10.1016/S2468-2667(16)30017-2.
- Peeters W, van den Brande R, Polinder S, Brazinova A, Steyerberg EW, Lingsma HF, Maas A. Epidemiology of traumatic brain injury in Europe. Acta Neurochir (Wien). 2015;157(10):1683–96. doi:https://doi.org/10.1007/s00701-015-2512-7.
- Thompson HJ, McCormick WC, Kagan SH. Traumatic brain injury in older adults: epidemiology, outcomes, and future implications. J Am Geriatr Soc. 2006;54(10):1590–95. doi:https://doi.org/10.1111/j.1532-5415.2006.00894.x.
- Teasdale G, Jennett B. Assessment of coma and impaired consciousness. A practical scale. Lancet. 1974;2(7872):81–84. doi:https://doi.org/10.1016/S0140-6736(74)91639-0.
- Af Geijerstam JL, Britton M. Mild head injury - mortality and complication rate: meta-analysis of findings in a systematic literature review. Acta Neurochir (Wien). 2003;145(10):843–50. discussion 850. doi:https://doi.org/10.1007/s00701-003-0115-1.
- Rosenfeld JV, Maas AI, Bragge P, Morganti-Kossmann MC, Manley GT, Gruen RL. Early management of severe traumatic brain injury. Lancet. 2012;380(9847):1088–98. doi:https://doi.org/10.1016/S0140-6736(12)60864-2.
- Cassidy JD, Carroll LJ, Peloso PM, Borg J, von Holst H, and Holm L, Kraus J, Coronado VG. Incidence, risk factors and prevention of mild traumatic brain injury: results of the WHO collaborating centre task force on mild traumatic brain injury. J Rehabil Med. 2004;36:28–60. doi:https://doi.org/10.1080/16501960410023732.
- Haydel MJ, Preston CA, Mills TJ, Luber S, Blaudeau E, DeBlieux PM. Indications for computed tomography in patients with minor head injury. N Engl J Med. 2000;343(2):100–05. doi:https://doi.org/10.1056/NEJM200007133430204.
- Ibanez J, Arikan F, Pedraza S, Sanchez E, Poca MA, Rodriguez D, Rubio E. Reliability of clinical guidelines in the detection of patients at risk following mild head injury: results of a prospective study. J Neurosurg. 2004;100(5):825–34. doi:https://doi.org/10.3171/jns.2004.100.5.0825.
- Mower WR, Hoffman JR, Herbert M, Wolfson AB, Pollack CV Jr., and Zucker MI. Developing a decision instrument to guide computed tomographic imaging of blunt head injury patients. J Trauma. 2005;59:954–59. doi:https://doi.org/10.1097/01.ta.0000187813.79047.42.
- Smits M, Dippel DW, Steyerberg EW, de Haan GG, Dekker HM, Vos PE, Kool DR, Nederkoorn PJ, Hofman A, Twijnstra A, et al. Predicting intracranial traumatic findings on computed tomography in patients with minor head injury: the CHIP prediction rule. Ann Intern Med. 2007;146(6):397–405. doi:https://doi.org/10.7326/0003-4819-146-6-200703200-00004.
- Stiell IG, Wells GA, Vandemheen K, Clement C, Lesiuk H, Laupacis A, McKnight RD, Verbeek R, Brison R, Cass D, et al. The Canadian CT head rule for patients with minor head injury. Lancet. 2001;357(9266):1391–96. doi:https://doi.org/10.1016/S0140-6736(00)04561-X.
- Unden J, Ingebrigtsen T, Romner B. Scandinavian guidelines for initial management of minimal, mild and moderate head injuries in adults: an evidence and consensus-based update. BMC Med. 2013;11(1):50. doi:https://doi.org/10.1186/1741-7015-11-50.
- Astrand R, Rosenlund C, and Unden J. Scandinavian guidelines for initial management of minor and moderate head trauma in children. BMC Med. 2016;14:33. doi:https://doi.org/10.1186/s12916-016-0574-x.
- Koskinen LO, Olivecrona M, Grande PO. Severe traumatic brain injury management and clinical outcome using the Lund concept. Neuroscience. 2014;283:245–55. doi:https://doi.org/10.1016/j.neuroscience.2014.06.039.
- Andersson EH, Bjorklund R, Emanuelson I, Stalhammar D. Epidemiology of traumatic brain injury: a population based study in western Sweden. Acta Neurol Scand. 2003;107(4):256–59. doi:https://doi.org/10.1034/j.1600-0404.2003.00112.x.
- Prince C, Bruhns ME. Evaluation and treatment of mild traumatic brain injury: the role of neuropsychology. Brain Sci. 2017;7(12):105. doi:https://doi.org/10.3390/brainsci7080105.
- Authority for traffic analysis in Sweden. Annual report 2018. Stockholm/Östersund: Trafikanalys. [ accessed 2019 May 10] https://www.trafa.se/vagtrafik/vagtrafikskador/.
- Bjornstig J, Bylund PO, and Bjornstig U. Vehicle-related injuries in and around a medium sized Swedish City - bicyclist injuries caused the heaviest burden on the medical sector. Inj Epidemiol. 2017;4:4. doi:https://doi.org/10.1186/s40621-016-0101-8.
- Isokuortti H, Iverson GL, Silverberg ND, Kataja A, Brander A, Ohman J, Luoto TM. Characterizing the type and location of intracranial abnormalities in mild traumatic brain injury. J Neurosurg. 2018;129(6):1588–97. doi:https://doi.org/10.3171/2017.7.JNS17615.
- Iverson GL, Lovell MR, Smith S, Franzen MD. Prevalence of abnormal CT-scans following mild head injury. Brain Inj. 2000;14(12):1057–61. doi:https://doi.org/10.1080/02699050050203559.
- Stiell IG, Clement C, Rowe B, Schull M, Brison R, Cass D, Eisenhauer M, McKnight D, Bandiera G, Holroyd B, et al. Comparison of the Canadian CT head rule and the new orleans criteria in patients with minor head injury. JAMA. 2005;294(12):1511–18. doi:https://doi.org/10.1001/jama.294.12.1511.
- Sweeney TE, Salles A, Harris OA, Spain DA, Staudenmayer KL. Prediction of neurosurgical intervention after mild traumatic brain injury using the national trauma data bank. World J Emerg Surg. 2015;10(1):23. doi:https://doi.org/10.1186/s13017-015-0017-6.
- Foks KA, Cnossen MC, Dippel DWJ, Maas A, Menon D, van der Naalt J, Steyerberg E, Lingsma H, Polinder S. Management of mild traumatic brain injury at the emergency department and hospital admission in Europe: a survey of 71 neurotrauma centers participating in the CENTER-TBI study. J Neurotrauma. 2017;34(17):2529–35. doi:https://doi.org/10.1089/neu.2016.4919.
- Culotta VP, Sementilli ME, Gerold K, Watts CC. Clinicopathological heterogeneity in the classification of mild head injury. Neurosurgery. 1996;38(2):245–50. doi:https://doi.org/10.1097/00006123-199602000-00002.
- Pandor A, Harnan S, Goodacre S, Pickering A, Fitzgerald P, Rees A. Diagnostic accuracy of clinical characteristics for identifying CT abnormality after minor brain injury: a systematic review and meta-analysis. J Neurotrauma. 2012;29(5):707–18. doi:https://doi.org/10.1089/neu.2011.1967.
- Einarsen CE, van der Naalt J, Jacobs B, Follestad T, Moen KG, Vik A, Haberg AK, Skandsen T. Moderate traumatic brain injury: clinical characteristics and a prognostic model of 12-month outcome. World Neurosurg. 2018;114:e1199–e1210. doi:https://doi.org/10.1016/j.wneu.2018.03.176.
- Jacobs B, Beems T, van der Vliet TM, Diaz-Arrastia RR, Borm GF, Vos PE. Computed tomography and outcome in moderate and severe traumatic brain injury: hematoma volume and midline shift revisited. J Neurotrauma. 2011;28(2):203–15. doi:https://doi.org/10.1089/neu.2010.1558.
- Fearnside M, McDougall P. Moderate head injury: a system of neurotrauma care. Aust N Z J Surg. 1998;68(1):58–64. doi:https://doi.org/10.1111/j.1445-2197.1998.tb04638.x.
- Te Ao B, Brown P, Tobias M, Ameratunga S, Barker-Collo S, Theadom A, McPherson K, Starkey N, Dowell A, Jones K, Feigin V. Cost of traumatic brain injury in New Zealand: evidence from a population-based study. Neurology. 2014;83(18):1645–52. doi:https://doi.org/10.1212/WNL.0000000000000933.
- Jacobsson LJ, Westerberg M, Lexell J. Demographics, injury characteristics and outcome of traumatic brain injuries in northern Sweden. Acta Neurol Scand. 2007;116(5):300–06. doi:https://doi.org/10.1111/j.1600-0404.2007.00896.x.