Abstract
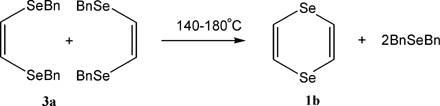
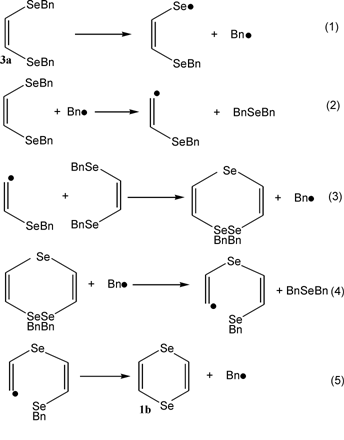
Heating (Z)-1,2-bis(benzylseleno)ethene at 140–180 °C leads to 1,4-diselenin (79% yield) and dibenzyl selenide. This unusual reaction represents a convenient method for preparing 1,4-diselenin.
Acknowledgement
The authors gratefully acknowledge support of this work by the U.S. National Science Foundation (CHE-0201555).
Notes
1A mixture of Bn2Se2 (6.8 g), KOH (8 g), DMSO (40 ml) and water (4 ml) was rotated at room temperature in a 1 l rotating autoclave under an atmosphere of acetylene (12 atm) for 5 h. The mixture was then diluted with water and extracted with ether. The organic phase was washed with water and dried over CaCl2. The ether was distilled off by heating on a warm water bath at ordinary pressure and then in vacuum at room temperature. The residue is crude 3a (6.98 g, 95%), which was used to prepare 1b without further purification. 1H NMR of the crude 3a (CDCl3, δ, ppm): 7.23 (m, 10H, C6H5), 6.75 (s, 2H, SeCH˭), 3.93 (s, 4H, SeCH2). MS, m/z (rel. int., %): 368 (50), 291 (19), 210 (100), 184 (30), 160 (17), 105 (44), 80 (17). Attempted distillation of 3a at 1 mmHg gave crude 1b, which was distilled in vacuo.
21,4-Diselenin 1b, bp 60–62 °C/1 mmHg. 1H NMR (CDCl3, δ, ppm): 7.02 (s). 13C NMR (CDCl3, δ, ppm): 120.11. MS, m/z (rel. int., %): 212 (55), 186 (19), 160 (100), 132 (60), 105 (33), 80 (43). Anal. Calcd for C4H4Se2: C, 22.88; H, 1.92; Se 75.20. Found: C, 23.21; H, 1.98; Se, 74.65.