Abstract
The degree of variation exhibited within the 793/B serotype (also known as 4/91 and CR88 serotypes) was investigated with nine French and 10 British isolates, collected between 1985 and 1994. The S1 part (1644 nucleotides) of the spike protein gene of the first known isolate of this serotype, FR/CR85131/85, had 95.9% to 97% nucleotide identity with the other isolates. Partial sequencing of isolates from Iran and Saudi Arabia, isolated in 2000, revealed approximately 95% nucleotide identity with European isolates, including the two live 793/B vaccinal strains, showing that they were not re-isolations of vaccinal virus. The data indicates that strains within the 793/B serotype have ≥96% nucleotide identity within the whole S1 gene and ≥93% nucleotide identity within the first 560 nucleotides, and ≥92% and ≥86% amino acid identities in the corresponding protein regions. This is similar to the identities exhibited within the Massachusetts serotype.
Sequence analysis of a 793/B field isolate after passage in embryonated eggs, then in chickens and then again in eggs revealed selection for a serine and alanine at S1 amino acid position 95 in chicken-passaged and egg-passaged virus, respectively. There was no change in pathogenicity. This is the first demonstration at gene sequence level of host-driven selection for infectious bronchitis virus.
Variation de la protéine de spicule du virus de la bronchite infectieuse de type 793/B, sur le terrain et lors de passages alternés sur poulets et œufs embryonnés
Le degré de variation présenté au sein du sérotype 793/B (connu également comme sérotypes 4/91 et CR88) a été étudié avec neuf souches françaises et dix souches britanniques, collectées entre les années 1985 et 1994. La partie S1 (1644 nucléotides) du gène de la protéine de spicule du premier isolat connu de ce sérotype, FR/CR85131/85, avait une identité nucléotidique de 95,9 à 97% avec les autres souches. Le séquençage partiel des souches d'Iran et d'Arabie Saoudite, isolées en 2000, a révélé une identité nucléotidique d'approximativement 95% avec les souches européennes, incluant les deux souches vaccinales à virus vivant 793/B, montrant qu'elles n'étaient pas des ré-isolements de virus vaccinaux. Les données indiquent que les souches au sein du sérotype 793/B ont une identité nucléotidique ≥96% en ce qui concerne la totalité du gène S1 et une identité nucléotidique ≥93% concernant les 560 premiers nucléotides, ainsi qu'une d'identité en acides aminés de ≥92% et de ≥86% pour les régions protéiques correspondantes. Ceci est similaire aux niveaux d'identité observés au sein du sérotype Massachusetts.
L'analyse de la séquence d'une souche 793/B du terrain, après passages sur œufs embryonnés, puis sur poulets et à nouveau sur œufs a révélé la sélection d'un acide aminé en position 95 de la S1, à savoir la sérine pour le passage sur poulet et l'alanine pour le passage sur œufs. Il n'y a pas eu de changement en ce qui concerne la pathogénicité. Ceci est la première démonstration, au niveau de la séquence du gène, de la sélection induite par l'hôte pour l'IBV.
Variationen im Spikeprotein des 793/B-Typs des Virus der infektiösen Bronchtis in Feldisolaten und während alternierender Passagen in Hühnern und embryonierten Eiern
Die Variationsbreite innerhalb des 793/B-Serotyps (auch bekannt als 4/91- und CR88-Serotyp) des Virus der infektiösen Bronchitis (IBV) wurde an neun französischen und 10 britischen Stämmen, die zwischen 1985 und 1994 isoliert worden waren, untersucht. Der S1-Teil (1644 Nukleotide) des Spikeproteingens des ersten Isolats dieses Serotyps FR/CR85131/85, hatte eine 95,9–97%ige Nukleotidübereinstimmung mit den anderen Isolaten. Die Teilsequenzierung von im Jahr 2000 gewonnenen Isolaten aus dem Iran und Saudi-Arabien ließ eine Nukleotidübereinstimmung von ungefähr 95% mit den europäischen Isolaten, einschließlich der beiden 793B-Lebendvakzinestämme, erkennen, was belegte, dass es sich nicht um Reisolierungen von Vakzinevirus handelte. Die Daten zeigen, dass die Stämme innerhalb der 793B-Serotyps bezogen auf das gesamte S1-Gen eine 96 %ige Nukleotidübereinstimmung, bezogen auf die ersten 560 Nukleotide eine 93 %ige Nukleotididentität und bezogen auf die korrespondierenden Proteinregionen Aminosäurenübereinstimmungen von ≥92% bzw. ≥86% aufweisen. Dies entspricht den Übereinstimmungen, die innerhalb des Massachusetts-Serotyps vorkommen.
Die Sequenzanalyse eines 793/B-Feldisolats nach Passagierung in embryonierten Eiern, dann in Hühnern und nachfolgend wieder in Eiern ließ eine Selektion auf Serin bzw. Alanin auf der Aminosäurenposition 95 im Hühner- bzw. Ei-passagiertem Virus erkennen. Es gab keine Veränderungen in der Pathogenität. Dies ist die Erstbeschreibung einer wirtsgesteuerten Selektion auf der Ebene der Genomsequenz beim IBV.
Variación de la proteína de la espícula del tipo 793/B de virus de bronquitis infecciosa, en el campo y durante pases alternativos en pollos y huevos embrionados
El grado de variación dentro del serotipo 793/B (también conocido como serotipo 4/91 y CR88) fue investigado con nueve aislados franceses y 10 aislados británicos, recogidos entre 1985 y 1994. La parte S1 (1644 nucleótidos) del gen de la proteína de la espícula del primer aislado conocido de este serotipo, FR/CR85131/85, presentó de un 95.9 a un 97% de identidad nucleotídica con otros aislados. La secuenciación parcial de aislados de Irán y Arabia Saudí, aislados en el 2000, reveló aproximadamente un 95% de identidad nucleotídica con los aislados europeos, incluídos las dos cepas vacunales vivas de 793/B, lo cual demostró que no eran reaislamientos del virus vacunal. Estos datos indican que las cepas dentro del serotipo 793/B presentan ≥96% de identidad nucleotídica en relación a todo el gen S1 y ≥93% de identidad nucleotídica teniendo en cuenta los primeros 560 nucleótidos, y ≥92% y ≥86% de identidad aminoacídica en las correspondientes regiones de la proteína. Esto es similar a las identidades exhibidas dentro del serotipo Massachusetts.
Los análisis de la secuencia de un aislado de campo de 793/B tras el pase en huevos embrionados, pollos y posteriormente otra vez en huevos embrionados reveló la selección de serina y alanina en la posición aminoacídica 95 de la S1 en virus pasado por pollo y huevo, repectivamente. No hubo cambios en patogenicidad. Esta es la primera demostración a nivel de secuencia génica de una selección relacionada con el huésped de IBV.
Introduction
Infectious bronchitis virus (IBV), together with Turkey coronavirus (Guy, Citation2000; Cavanagh, Citation2001a,Citationb; Cavanagh et al., Citation2001) and Pheasant coronavirus (Cavanagh et al., Citation2002; Welchman et al., Citation2002), form Group 3 within the genus Coronavirus, family Coronaviridae (Cavanagh, Citation1997; Cavanagh, Citation2001a; Enjuanes et al., Citation2000). Although usually associated with respiratory disease (Matthijs et al., Citation2003) and drops in egg production, some strains also cause nephritis (Cook et al., Citation2001b; Li & Yang, Citation2001).
It has been known for many years that IBV exists as dozens of serotypes (Cavanagh & Naqi, Citation2003). The serotype is defined by the virus neutralization test, the defining antibodies being against the amino-terminal half, S1, of the large spike glycoprotein (S) (Koch et al., Citation1990; Kant et al., Citation1992). In keeping with the large number of IBV serotypes, the S1 protein is very variable; serotypes commonly differ by 20% to 25%, and up to 50%, of amino acids. There have been few in-depth analyses of the degree of variation within IBV serotypes (Cavanagh et al., Citation1988, Citation1992; Lee & Jackwood, Citation2001). In this paper we report the intra-serotype variation exhibited by the 793/B serotype (Gough et al., Citation1992), also known as 4/91 (Parsons et al., Citation1992) and CR88 (Picault et al., Citation1995). Virus of this serotype has been detected in several European countries, although at different frequencies (Cook et al., Citation1996; Capua et al., Citation1999; Cavanagh et al., Citation1999; Picault et al., Citation1995; Meulemans et al., Citation2001), although not in some others (Farsang et al., Citation2002). This serotype may have entered the UK in the winter of 1990/91, when it was sometimes associated with deep pectoral muscle myopathy in layers, in addition to the more usual manifestations of infectious bronchitis (Gough et al., Citation1992; Parsons et al., Citation1992). Subsequently it was discovered that this serotype had been present in France for several years. Indeed, the first known strain of this serotype was isolated there in 1985 (Picault et al., Citation1995).
Jackwood and colleagues have compared the S1 gene sequence of a 793/B-type field virus and its egg-passaged derivative and reported a difference of amino acid at residue 95 (Callison et al., Citation2001). We have investigated this further to ascertain whether this difference is host-associated.
Materials and methods
Isolates of the 793/B serotype
The nomenclature of the isolates used here is that proposed by Cavanagh (Citation2001a): country/isolate number/year of isolation. The names of the countries of origin in this report have been abbreviated thus: France (FR), Iran (IR), Saudi Arabia (SA) and United Kingdom (UK). The UK isolates isolated in 1991 and 1993 have been described by Adzhar et al. (Citation1997). These and the names of the other isolates investigated are shown later (‘Databank gene sequence accession numbers’).
Extraction of RNA
RNA was extracted as described previously, using guanidinium isothiocyanate, and dissolved in 20 μl water (Cavanagh et al., Citation2002).
Reverse transcription-polymerase chain reaction
The reverse transcription-polymerase chain reactions (RT-PCRs) were performed as previously described (Cook et al., Citation2001a; Cavanagh et al., Citation2002). The whole of the S1 part of the S gene was amplified using oligonucleotides IBP1− and S1Uni2+ (Adzhar et al., Citation1996, Citation1997). Nucleotides 92 to 570 of S1 were amplified using oligonucleotides B1+ (92 5′-AAAGTGCCTTTAGGCCTGG-3′ 110) and B88− (557 5′-GCCCACGTCCGCAAA-3′ 570), where the numbers refer to the positions of the first and last nucleotides in relation to the S1 gene sequence of 793/B-type isolates (Adzhar et al., Citation1997). Oligonucleotides B1 and B88 were designed to be specific to the 793/B serotype. The more general oligonucleotides XCE1– and XCE2+ (Capua et al., Citation1999; Cavanagh et al., Citation1999), corresponding to nucleotides 709 to 1166 in the S1 gene of the 793/B serotype, were used to amplify part of S1 of the Iranian and Saudi Arabian isolates.
Nucleotide sequencing
Sequencing of PCR products was performed using either the Sequenase PCR product sequencing kit (USB; Amersham, Little Chalfont, Bucks., UK) or the Thermosequenase dye terminator cycle sequencing kit, version 2.0 (Amersham). The PCR products complementary to the whole of S1 were sequenced using a number of oligonucleotides as sequencing primers (Adzhar et al., Citation1997) while the smaller PCR products were sequenced using the same oligonucleotides as had been used to amplify them.
Sequence analysis
Sequences were aligned using ClustalX version 1.64b (Thompson et al., Citation1994) and compared using GeneDoc Multiple Sequence Alignment Editor and Shading Utility version 2.5.000 (http://www.psc.edu/biomed/genedoc). Aligned sequences were analysed using PHYLIP version 3.57C (http://evolution.genetics.washington.edu/phylip.html; Felsentein, Citation1993). The trees were constructed using the neighbour-joining programme and viewed using Treeview (http://taxonomy.zoology.gla.ac.uk/fod/treeview.html).
Databank gene sequence accession numbers
First region in S1 (up to nucleotide 561): FR/CR85131/85 (AJ619606); FR/CR88061/88 (AJ619607); FR/CR88099/88 (AJ619608); FR/CR88121/88 (AJ619609); FR/CR94007/94 (AJ619610); FR/CR94047/94 (AJ619611); FR/CR94048/94 (AJ619612); FR/CR94247/94 (AJ619613); FR/CR94279/94 (AJ619614); FR/CR94364/94 (AJ619615); UK/1/91 (AJ619616); UK/2/91 (AJ619617); UK/3/91 (AJ619618); UK/5/91 (AJ619619); UK/6/91 (AJ619620); UK/7/93 (AJ619621); UK/8/93 (AJ619622); UK/9/93 (AJ619623); UK/10/93 (AJ619624); UK/11/93 (AJ619625); UK/12/93 (AJ619626); UK/42/96 (AJ619627); UK/159/93 (AJ619628); UK/186/96 (AJ619629); UK/236/95 (AJ619630); UK/574/96 (AJ619631); UK/1233/95 (AJ619632).
Complete S1 sequences: UK/1233/95 (AJ618984); FR/CR85131/85 (AJ618985); FR/CR88061/88 (AJ618986); FR/CR94047/94 (AJ618987). Previously published complete S1 sequences, included in the analyses: UK/7/91 (Z83975); UK/2/91 (Z83976); UK/3/91 (Z83977); UK/5/91 (Z83978); UK/7/93 (Z83979).
Results
Mid-1990s UK isolates were very similar to earlier 793/B-type strains
Each isolate that we analysed was probably a quasispecies (i.e. there would have been heterogeneity at some nucleotide positions). Our sequencing approach was to sequence PCR products. The sequence obtained reflects the nucleotides that were possessed by the majority of the viral RNA molecules at each nucleotide position. All the British and French strains in this study were isolated before vaccines of the 793/B serotype were used in the field.
Following sequencing of nucleotides 92–557 of the S1 gene, the UK isolates of the 793/B serotype isolated between 1991 and 1996 had nucleotide identity of ≥94.1%, compared with approximately 70% identity with many other serotypes.
Sequence analysis of French isolates of the 793/B serotype
Virus neutralization tests in the mid-1990s had shown that many IBVs isolated in France since 1988 were serologically related to the 793/B-type isolates of the UK (Picault et al., Citation1995). Our retrospective analysis has further revealed that another 793/B-serotype French isolate, FR/CR85131/85, isolated in 1985, is the oldest known isolate of this serotype. There is no evidence to suggest that this isolate was the progenitor of the others. Notwithstanding, FR/CR85131/85 had 95.3% to 97.4% nucleotide identity with the other French isolates.
Comparison of the French and British isolates
Clustal analysis of the first 560 or so S1 nucleotides of the French and British isolates showed that most, but not all, of them tended to segregate along geographical lines (). A similar, although not identical, distribution was evident when the amino acid sequences were compared (). The degree of identity of FR/CR85131/85 with the British isolates (≥94.2%) was essentially the same as that among the French isolates. This is reflected not only in the Clustal diagram based on the first 560 nucleotides (), but also that based on complete S1 sequences (data not shown).
Figure 1. Relationships between the UK and French isolates of the 793/B serotype illustrated by a maximum likelihood phylogeny unrooted tree, based on nucleotides 1 to 561. (a) Nucleotide sequences. (b) Amino acid sequences. The French isolates are shown in bold. The oldest isolate, FR/CR85131/85, has been boxed.
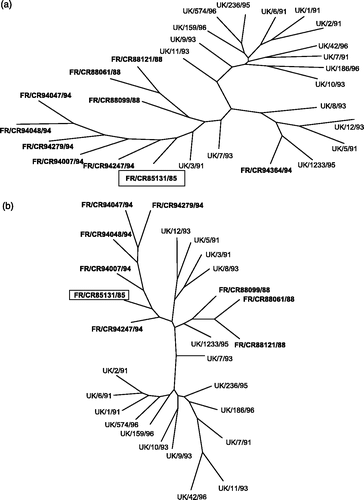
The results show that within the 793/B serotype, nucleotide identity in the first 560 nucleotides was ≥93%, compared with about 70% when one compares the nucleotides of most serotypes with other serotypes. At the deduced amino acid level, FR/CR85131/85 had ≥86.5% identity with the French and British isolates in the first 560 nucleotides, compared with approximately 65% when comparing different serotypes.
The S1 genes of three of the French isolates, FR/CR85131/85, FR/CR88061/88 and FR/CR94047/94, were sequenced in their entirety. The S1 gene of the 1985 French isolate had 95.9% and 97.0% nucleotide identity with the other two French isolates, respectively, and 96.1% to 96.9% identity with the five UK isolates for which the complete S1 gene sequence had been established. Thus, nucleotide identity within the whole of the S1 gene for 793/B-serotype isolates was ≥96%, compared with generally ≤80% between many serotypes. The deduced amino acid sequences of the complete S1 proteins had ≥92.4% identity with the oldest known 793/B isolate, FR/CR85131/85, compared with 75% to 80% between many serotypes.
Synonymous (s) and non-synonymous (ns) genetic changes are those that do not and do, respectively, cause amino acid differences. The average ratio d s/d ns for the whole S1 sequences of the 793/B serotype was 1.7. The d s/d ns ratios following comparisons of whole S1 sequences of representative strains of different serotypes (Massachusetts M41 strain, D274 strain D207 and 793/B strain CR85131/85) were in the range 3.4 to 5.5 (mean, 4.4).
Sequence analysis of isolates from Iraq and Saudi Arabia
Two isolates from Iran and one from Saudi Arabia, isolated in 2000, had been characterized as being of the 793/B serotype by the virus neutralization test. Corroborative evidence was sought by sequencing S1 nucleotides 802 to 1081 after RT-PCR using general IBV oligonucleotide primers XCE1− and XCE2+ (Capua et al., Citation1999; Cavanagh et al., Citation1999). The two Iranian isolates had 97.5% identity with each other, and had 94.7% and 96.4% nucleotide identity, respectively, with UK/7/91 (the latter strain was isolated from the same chickens as the UK/4/91 strain that was subsequently used to make an attenuated vaccine; Parsons et al., Citation1992). The Saudi isolate had 94.7% identity with UK/7/91. These figures are very similar to the comparison of one of the most recent French isolates examined, FR/CR94047/94, with UK/7/91 (96.4% identity). Thus the Iranian and Saudi isolates were clearly of the 793/B type by sequence as well as by serotype.
The Iranian and Saudi isolates had approximately 95% identity in the sequenced region with the 4/91 live vaccinal strain and with FR/CR88061/88, from which the CR88 live vaccine was derived. Thus the Iranian, Saudi Arabian, French and UK isolates all differed from the two vaccinal strains to a similar extent: the Iranian and Saudi isolates were not re-isolations of vaccinal virus.
Furthermore our analysis shows that 793/B-serotype strains isolated 15 years apart still had >94% nucleotide identity in this region.
Host-selected amino acid difference within the S1 protein
Callison et al. (Citation2001) sequenced the S1 gene of the 793/B serotype field isolate UK/4/91 (called UK/4f/91 in this paper) and its embryo-passaged, attenuated derivative (UK/4e/91). They reported nucleotide differences at three positions: 283, 1522 and 1589 (where position 1 is the start of the open reading frame, i.e. inclusive of the signal sequence that is not present in the mature spike protein). We were unable to confirm the differences at positions 1522 and 1589. At position 283, Callison et al. (Citation2001) reported a T residue in the field strain and a G residue in the egg-passaged virus. Our sequencing of PCR products derived from UK/4e/91 reproducibly revealed both G and T at nucleotide position 283 (), suggesting that there were two subpopulations in the embryo-passaged virus with respect to this position—one a T residue as in the initial field virus, the other with a G residue as detected by Callison et al. (Citation2001). Our finding suggested that growth in chickens or embryos favoured a T or G at nucleotide 283, respectively.
Table 1. Host-dependent variation at nucleotide position 283 (encoding amino acid residue 95) of the S1 part of the S gene of UK/4/91
The egg-adapted virus had been back-passaged in chickens to establish whether the attenuated phenotype was stable (i.e. still attenuated); it was. Notwithstanding, it was still possible that back-passage in chickens would favour UK/4/91 virus with a T residue at 283, like the wild-type strain, even though that was not related to pathogenicity. We therefore sequenced this part of the S gene for back-passages 1 (BP/C1) and 10 (BP/C10) in chickens of UK/4e/91. BP/C1 still had a mixture of T and G at this position but in BP/C10 only a T residue was detected. This indicated that either UK/4e/91 had reverted, during passage in chickens, to a T residue (as in the UK/4f/91 field isolate; ), or that a subpopulation within UK/4e/91 with a T residue had been selected for.
The nucleotides T and G were the first bases in codon 95, encoding serine (field virus, UK/4f/91) and alanine (embryo-adapted virus, UK/4e/91), respectively. This suggested that growth in chickens and growth in embryonated eggs was favoured by serine and alanine, respectively, at this position. When BP/C10 was back-passaged in embryonated eggs, to give BP/C10/E10, position 283 reverted again, to G, the same as the initial embryo-passaged virus (). Codon 95 again encoded alanine, supporting the proposition that UK/4/91 with serine and alanine at this position was somehow better fitted to replication in chickens and in embryonated eggs, respectively.
Re-inspection of 10 French and 10 UK 793/B-type isolates for which we had sequence data in this region showed that 19 of them had a T residue at position 283, encoding serine, indicating this to be a characteristic of field virus, replicating in chickens, although one UK isolate had a leucine residue at this position.
Discussion
Our first evaluation of the degree of variation exhibited within the spike protein genes of a single serotype was with eight strains of the Massachusetts serotype, collected over a three-decade period (Cavanagh et al., Citation1988). The maximum difference between them was 4% of nucleotides and 6% of amino acids of the S1 protein. Our analysis of S1 of seven isolates of the D274 group, from The Netherlands and the UK over an 8-year period (Cavanagh et al., Citation1992) revealed maximum differences within the group of 3% of nucleotides and 5% of amino acids. In our current investigation of the 793/B serotype there were maximum differences within the serotype of 4% of nucleotides and 8% of amino acids. The ratio d s/d ns of synonymous to non-synonymous (resulting in amino acid changes) nucleotide changes for the whole S1 sequences of the 793/B serotype was 1.7, compared with ratios of approximately 4.4 when Massachusetts, D274 and 793/B serotypes were compared with each other. For comparison, the d s/d ns ratio for the IBV nucleocapsid protein, not expected to be under selective pressure to change, was 6.5 (Adzhar et al., Citation1997). d s/d ns<1.0 is often cited as evidence for the presence of positive Darwinian selection. Clearly all the ratios already stated were >1.0, suggesting that some of the changes were not related to selection pressures. That the d s/d ns ratio within the 793/B serotype (1.7) was nearer to 1.0 than that between other serotypes (mean, 4.4) might indicate that when a serotype starts to diversify, a high proportion of the small number of S1 mutations are non-synonymous, whereas subsequent genetic changes involve predominantly synonymous changes.
Interestingly, the seven/eight isolates in the D274 group were not all of the same serotype, as defined by the virus neutralization test (Cavanagh et al., Citation1992). Our conclusion then, as now, was that most of the neutralizing antibody was induced by a small number of dominant epitopes, a small number of amino acid changes in these regions being sufficient to change the serotype. Serotypes within the D274 group differed by as few as 10 residues in the S1 protein. It may even be that not all of these few changes were required for the change in serotype.
Do a small number of amino acid changes in S1, sufficient to change the serotype, affect cross-protection? This may sometimes be the case. Isolates UK/123/82 and UK/6/82, of different serotype, differ by only 3% of S1 amino acids (Cavanagh et al., Citation1992). When chickens were inoculated with UK/6/82 and later challenged with UK/123/82, protection against the latter was poor (Cavanagh et al., Citation1997). The S1 proteins of the Beaudette and M41 strains of IBV have 95% amino acid identity. When chickens were inoculated with a recombinant IBV Beaudette expressing the spike protein of the M41 strain, good protection was induced against challenge with M41, as assessed by ciliary activity and snicking (Hodgson et al., Citation2004). In contrast, inoculation with Beaudette induced very poor protection against challenge with M41, suggesting that some of the few amino acids that differed between the two strains were associated with protection-inducing epitopes.
Some isolates that had ≥97% S1 nucleotide identity and ≥95% S1 amino acid identity with the Dutch D274 strain were assessed as being of different serotypes by neutralization tests using tracheal organ cultures (Cook, Citation1984; Cook & Huggins, Citation1986; Cavanagh et al., Citation1992). It may be that a given serotype is always on the brink of becoming a new serotype, as only a small number of changes are required to bring this about. This is not in conflict with the observation that serotypes exhibit only little variation in S1 (up to 6% or so) over long periods of time; a sequence investigation of isolates limited to a particular serotype has, by definition, eliminated isolates that may differ a little more in protein sequence and that might have, consequently, become a different serotype.
Comparison of the amino acid differences among the UK and French isolates of the 793/B serotype has not revealed any fixation of changed amino acids (cumulative changes) as time progressed. Also, some isolates obtained several years apart were more similar than some of those isolated within a given year. In contrast, Lee & Jackwood (Citation2001) have shown cumulative changes in their study of isolates related to the Delaware serotype. A contributory factor to the different findings might be that the 793/B isolates in our study had been obtained before the introduction of 793/B-type vaccines, whereas the Delaware type was in use before and during the period when Lee and Jackwood collected their isolates.
The change that we observed at residue 95 in the S1 protein of the UK/4/91 strain during passages and back-passages in embryonated eggs and chickens was not associated with pathogenicity; the attenuated UK/4e/91 remained non-pathogenic after reversion from an alanine to serine residue had occurred at position 95. There were two possible explanations for our observation of a switch from a T residue in the field virus to a G residue in the egg-passaged virus at nucleotide position 283. First, that some of the field strain had mutated at this position during passage in embryonated eggs. Second, the field strain itself was a mixture at this position; the ratio of G to T being low enough to make the G residue not detectable by sequencing of the RT-PCR product. Notwithstanding, replication in chickens and embryonated eggs was favoured by a serine or alanine residue, respectively, at amino acid position 95.
This situation is reminiscent of that for human influenza virus, in that passage of clinical isolates in embryonated chicken eggs results in the selection of variants with one, sometimes more, amino acid substitutions in the haemagglutinin spike protein (Katz & Webster, Citation1992). The substitutions are in the vicinity of the receptor binding site. Virus from clinical specimens has an affinity for cell surface glycoproteins and/or gangliosides with terminal sialyloligosaccharides in which the sialic acid residue is linked to galactose by an α2,6 linkage. In contrast, influenza virus passaged in embryonated eggs has an affinity for sialoligosaccharides with terminal α2,3 linkages. Ito et al. (Citation1997) have shown that sialic acid residues of endodermal cells of the chorioallantoic membrane (CAM) of chicken eggs have α2,3 linkages. The authors concluded that a lack of α2,6 linkages in the CAM of embryonated eggs selected influenza virus mutants with an haemagglutinin that had a high affinity for α2,3 linkages.
The switching of serine to alanine at residue 95 of the IBV spike protein following passage in embryonated eggs may or may not be related to interaction with receptors in the CAM of embryonated eggs. Schultze et al. (Citation1992) showed that attachment of embryo-grown IBV to, and subsequent agglutination of, chicken erythrocytes required sialic acid that was α2,3-linked to galactose—the linkage present on CAM cells (Ito et al., Citation1997). Thus it is conceivable that egg-grown IBV attaches preferentially to permissive cells that have sialoligosaccharides with terminal α2,3 linkages and that replication in chickens might favour the selection of spike protein with a higher affinity for α2,6 linkages.
This work was supported by the British Chicken Association/British Poultry Council, the Department of the Environment, Food and Rural Affairs (grant OD0712), Intervet UK, and the Biotechnology and Biological Sciences Research Council, UK.
Additional information
Notes on contributors
Michael Hess Footnote†
Present address: University of Veterinary Medicine, Clinic for Avian, Reptile and Fish Medicine, Veterinärplatz 1, A-1210 Wien, Austria.Notes
Present address: University of Veterinary Medicine, Clinic for Avian, Reptile and Fish Medicine, Veterinärplatz 1, A-1210 Wien, Austria.
References
- Adzhar , A , Shaw , K , Britton , P and Cavanagh , D . (1996) . Universal oligonucleotides for the detection of infectious bronchitis virus by the polymerase chain reaction . Avian Pathology , 25 : 817 – 836 .
- Adzhar , A , Gough , RE , Haydon , D , Shaw , K , Britton , P and Cavanagh , D . (1997) . Molecular analysis of the 793/B serotype of infectious bronchitis virus in Great Britain . Avian Pathology , 26 : 625 – 640 .
- Callison , SA , Jackwood , MW and Hilt , DA . (2001) . Molecular characterization of infectious bronchitis virus isolates foreign to the United States and comparison with United States isolates . Avian Diseases , 45 : 492 – 499 .
- Capua , I , Minta , Z , Karpinska , E , Mawditt , K , Britton , P , Cavanagh , D and Gough , RE . (1999) . Co-circulation of four types of infectious bronchitis virus (793/B, 624/I, B1648 and Massachusetts) . Avian Pathology , 28 : 587 – 592 .
- Cavanagh , D . (1997) . Nidovirales: a new order comprising Coronaviridae and Arteriviridae . Archives of Virology , 142 : 629 – 633 .
- Cavanagh , D . (2001a) . Commentary. A nomenclature for avian coronavirus isolates and the question of species status . Avian Pathology , 30 : 109 – 115 .
- Cavanagh , D . (2001b) . Innovation and discovery: the application of nucleic acid-based technology to avian virus detection and characterization . Avian Pathology , 30 : 581 – 598 .
- Cavanagh D Naqi S (2003) Infectious bronchitis In Y.M. Saif, H.J. Barnes, J.R. Glisson, A.M. Fadly, L.R. McDougald & D.E. Swayne (Eds.) Diseases of Poultry 11th edn (pp. 101–119) Ames Iowa State University Press
- Cavanagh , D , Davis , PJ and Mockett , APA . (1988) . Amino acids within hypervariable region 1 of avian coronavirus IBV (Massachusetts serotype) spike glycoprotein are associated with neutralization epitopes . Virus Research , 11 : 141 – 150 .
- Cavanagh , D , Davis , PJ , Cook , JKA , Li , D , Kant , A and Koch , G . (1992) . Location of the amino-acid differences in the S1 spike glycoprotein subunit of closely related serotypes of infectious-bronchitis virus . Avian Pathology , 21 : 33 – 43 .
- Cavanagh , D , Ellis , MM and Cook , JKA . (1997) . Relationship between sequence variation in the S1 spike protein of infectious bronchitis virus and the extent of cross-protection in vivo . Avian Pathology , 26 : 63 – 74 .
- Cavanagh , D , Mawditt , K , Britton , P and Naylor , CJ . (1999) . Longitudinal field studies of infectious bronchitis virus and avian pneumovirus in broilers using type-specific polymerase chain reactions . Avian Pathology , 28 : 593 – 605 .
- Cavanagh , D , Mawditt , K , Sharma , M , Drury , SE , Ainsworth , HL , Britton , P and Gough , RE . (2001) . Detection of a coronavirus from turkey poults in Europe genetically related to infectious bronchitis virus of chickens . Avian Pathology , 30 : 355 – 368 .
- Cavanagh , D , Mawditt , K , Welchman , D , de , B , Britton , P and Gough , RE . (2002) . Coronaviruses from pheasants (Phasianus colchicus) are genetically closely related to coronaviruses of domestic fowl (infectious bronchitis virus) and turkeys . Avian Pathology , 31 : 81 – 93 .
- Cook , JKA . (1984) . The classification of new serotypes of infectious bronchitis virus isolated from poultry flocks in Britain between 1981 and 1983 . Avian Pathology , 13 : 733 – 741 .
- Cook , JKA and Huggins , MB . (1986) . Newly isolated serotypes of infectious bronchitis virus: their role in disease . Avian Pathology , 15 : 129 – 138 .
- Cook , JKA , Orbell , SJ , Woods , MA and Huggins , MB . (1996) . A survey of the presence of a new infectious bronchitis virus designated 4/91 (793B) . Veterinary Record , 138 : 178 – 180 .
- Cook , JKA , Huggins , MB , Orbell , SJ , Mawditt , K and Cavanagh , D . (2001a) . Infectious bronchitis virus vaccine interferes with the replication of avian pneumovirus vaccine in domestic fowl . Avian Pathology , 30 : 233 – 242 .
- Cook , JKA , Chesher , J , Baxendale , W , Greenwood , N , Huggins , MB and Orbell , SJ . (2001b) . Protection of chickens against renal damage caused by a nephropathogenic infectious bronchitis virus . Avian Pathology , 30 : 423 – 426 .
- Enjuanes L Brian D Cavanagh D Holmes K Lai MMC Laude H Masters P Rottier P Siddell S Spaan WJM Taguchi F Talbot P (2000) Coronaviridae In M.H.V. Van Regenmortel, C.M. Fauquet, D.H.L. Bishop, E.B. Carstens, M.K. Estes, S.M. Lemon, J. Maniloff, M.A. Mayo, D.J. McGeoch, C.R. Pringle & R.B. Wickner (Eds.) Virus Taxonomy, Seventh Report of the International Committee on Taxonomy of Viruses (pp. 835–849) New York Academic Press
- Farsang , A , Ros , C , Renström , LHM , Baule , C , Soós , T and Belák , S . (2002) . Molecular epizootiology of infectious bronchitis virus in Sweden indicating the involvement of a vaccine strain . Avian Pathology , 31 : 229 – 236 .
- Felsentein J (1993) PHYLIP (Phylogeny Inference Package) version 3.5c Seattle WA Department of Genetics, University of Washington
- Gough , RE , Randall , CJ , Dagless , M , Alexander , DJ , Cox , WJ and Pearson , D . (1992) . A ‘new’ strain of infectious bronchitis virus infecting domestic fowl in Great Britain . The Veterinary Record , 130 : 493
- Guy , JS . (2000) . Turkey coronavirus is more closely related to avian infectious bronchitis virus than to mammalian coronaviruses . Avian Pathology , 29 : 206 – 212 .
- Hodgson , T , Casai , R , Dove , B , Britton , P and Cavanagh , D . (2004) . Recombinant infectious bronchitis coronavirus Beaudette with the spike protein gene of the pathogenic M41 strain remains attenuated but induces protective immunity . Journal of Virology , 78 : 13804 – 13811 .
- Ito , T , Suzuki , Y , Takada , A , Kawamoto , A , Otsuki , K , Masuda , H , Yamada , M , Suzuki , T , Kida , H and Kawaoka , Y . (1997) . Differences in sialic acid–galactose linkages in the chicken amnion and allantois influence human influenza virus receptor specificity and variant selection . Journal of Virology , 71 : 3357 – 3362 .
- Kant , A , Koch , G , van Roozelaar , DJ , Kusters , JG , Poelwijk , FAJ and van der Zeijst , BAM . (1992) . Location of antigenic sites defined by neutralizing monclonal antibodies on the S1 avian infectious bronchitis virus glycopolypeptide . Journal of General Virology , 73 : 591 – 596 .
- Katz , JM and Webster , RG . (1992) . Amino acid sequence identity between the HA1 of influenza A (H3N2) viruses grown in mammalian and primary chick kidney cells . Journal of General Virology , 73 : 1159 – 1165 .
- Koch , G , Hartog , L , Kant , A and van Roozelaar , DJ . (1990) . Antigenic domains on the peplomer protein of avian infectious bronchitis virus: correlation with biological functions . Journal of General Virology , 71 : 1929 – 1935 .
- Lee , CW and Jackwood , MW . (2001) . Origin and evolution of Georgia 98 (GA98), a new serotype of avian infectious bronchitis virus . Virus Research , 80 : 33 – 39 .
- Li , H and Yang , H-C . (2001) . Sequence analysis of nephropathogenic infectious bronchitis virus strains of the Massachusetts genotype in Beijing . Avian Pathology , 30 : 535 – 541 .
- Matthijs , MRG , van Eck , JHH , Landman , WJM and Stegeman , JA . (2003) . Ability of Massachusetts-type infectious bronchitis virus to increase colibacillosis susceptibility in commercial broilers: a comparison between vaccine and virulent field virus . Avian Pathology , 32 : 473 – 481 .
- Meulemans , G , Boschmans , M , Decaesstecker , M , van den Berg , TP , Denis , P and Cavanagh , D . (2001) . Epidemiology of infectious bronchitis virus in Belgian broilers: a retrospective study, 1986 to 1995 . Avian Pathology , 30 : 411 – 421 .
- Parsons , D , Ellis , MM , Cavanagh , D and Cook , JKA . (1992) . Characterisation of an avian infectious bronchitis virus isolated from IB-vaccinated broiler breeder flocks . The Veterinary Record , 131 : 408 – 411 .
- Picault JP Drouin P Lamande J Allee C Toux JY Le Coq H Guittet M Bennejean G (1995) L'epizootie recente de bronchite infectieuse aviaire en France: importance, evolution et etiologie Proceedings of the 1eres Journée de la Recherche Avicole, Angers 28–29, March 177 179
- Schultze , B , Cavanagh , D and Herrler , G . (1992) . Neuraminidase treatment of avian infectious bronchitis coronavirus reveals a hemagglutinating activity that is dependent on sialic acid-containing receptors on erythrocytes . Virology , 189 : 792 – 794 .
- Thompson , JD , Gibson , TJ , Plewniak , F , Jeanmougin , F and Higgins , DG . (1994) . The clustal_X windows interface: flexible strategies for multiple sequence alignment aided by quality analysis tools . Nucleic Acids Research , 25 : 4876 – 4882 .
- Welchman , D , de , B , Bradbury , JM , Cavanagh , D and Aebischer , NJ . (2002) . Infectious agents associated with respiratory disease in pheasants . Veterinary Record , 150 : 658 – 664 .