 ?Mathematical formulae have been encoded as MathML and are displayed in this HTML version using MathJax in order to improve their display. Uncheck the box to turn MathJax off. This feature requires Javascript. Click on a formula to zoom.
?Mathematical formulae have been encoded as MathML and are displayed in this HTML version using MathJax in order to improve their display. Uncheck the box to turn MathJax off. This feature requires Javascript. Click on a formula to zoom.Abstract
The objectives of this study were to investigate the occurrence of eyespot in commercial spring cereal crops in four climatic areas in Tunisia and characterize the dominant species of Oculimacula responsible for the disease. A total of 294 wheat, barley and oat fields were surveyed for eyespot incidence and severity during four cropping seasons from 2010 to 2014. Eyespot was identified in 63.5% of the fields with an average incidence of 23.1% infected stems. The number of infected fields as well as the incidence and severity of disease increased significantly during the 5 years of the study. A significant difference in the occurrence of eyespot among the climatic regions was noted with the wetter areas having greater disease incidence. The highest incidence of eyespot was recorded in durum wheat and bread wheat fields, whereas barley was significantly less infected. The effect of previous crop on eyespot incidence was not significant. All 70 isolates collected in this study were identified as O. yallundae and among them, 39 were identified as MAT1-2 and 31 as MAT1-1. This study highlights the influence of climatic conditions on the distribution of eyespot in the cereal growing areas of Tunisia as well as the increasing occurrence of the disease. The predominance of O. yallundae is an important consideration in the choice of integrated management strategies for eyespot. The presence of both mating types of O. yallundae in similar proportions suggests that sexual reproduction may be occurring.
Résumé
Les objectifs de cette étude ont été d’évaluer l’incidence et la distribution du piétin verse dans les cultures céréalières de printemps localisées dans quatre zones climatiques de la Tunisie et de caractériser les espèces dominantes d’Oculimacula responsables de la maladie. Un total de 294 champs de blé, d’orge et d’avoine a été évalué pour l’incidence et la svéréité du piétin verse au cours de quatre campagnes céréalières de 2010 à 2014. La maladie a été identifiée dans 63,5% des champs avec une incidence moyenne de 23,1%. Le nombre de champs infesté ainsi que l’incidence et la sévérité de la maladie ont augmenté d’une manière significative au cours des 5 années. Par ailleurs, cette étude a révélé des incidences significativement plus importantes dans les régions humides. De plus, les incidences les plus élevées ont été enregistrées dans les champs de blé dur et de blé tendre, tandis que les cultures d’orge ont été significativement moins infestées. L’effet du précédent cultural sur l’incidence du piétin verse n’a pas été significatif; cependant, l’incidence et la sévérité ont été plus importantes lorsque le précédent cultural était une céréale. L’identification moléculaire en utilisant des amorces spécifiques d’espèces a montré que tous les 70 isolats collectés appartiennent à l’espèce O. yallundae. La PCR multiplex des types sexués a révélé que 39 isolats ont été identifiés comme MAT1-2 et 31 comme MAT1-1. Cette étude a mis en évidence l’influence des conditions climatiques sur la distribution du piétin verse dans les zones céréalières de Tunisie d’une part et l’incidence croissante de la maladie d’autre part. La prédominance de l’espèce O. yallundae est à prendre en considération dans le choix des stratégies de lute contre le piétin verse. La présence des deux types sexués de l’espèce O. yallundae à des fréquences similaires suggère que la reproduction sexuée peut avoir lieu dans les systèmes de culture de la Tunisie.
Introduction
Eyespot, caused by Oculimacula yallundae (Wallwork and Spooner) Crous and W. Gams [formerly Tapesia yallundae, anamorph Helgardia (formerly Pseudocercosporella) herpotrichoides] and O. acuformis (Boerema, R. Pieters and Hamer) Crous and W. Gams [formerly Tapesia acuformis, anamorph Helgardia (Pseudocercosporella) acuformis], is an important fungal disease of the stem base of cereals grown in temperate regions such as Northwest Europe, Northwest USA, and New Zealand (Lucas et al. Citation2000; Crous et al. Citation2003). Eyespot forms lesions on the leaf sheaths and culms near the soil level, and their elliptical shape gives rise to the name of the disease, which is also known as strawbreaker foot rot. The species causing this disease are soilborne and infection takes place at the lower stems of plants, resulting in stem breakage, lodging and yield reduction. In the Pacific Northwest of the USA, up to 50% yield reduction has been documented in severely affected fields (Murray Citation2010). These pathogens have a wide host range including cereals and grass species (Lucas et al. Citation2000). Wheat, barley, rye, oats, and other related grasses can be affected, with wheat being the most susceptible (Murray et al. Citation1994; Chapman et al. Citation2008). The primary source of eyespot infection is the conidia, which form on infested plant residues from previous crops in cool moist weather and spread via rain-splashed droplets. The sexual stages of both species have been reported in many regions around the world including Australia (Wallwork Citation1987), Europe (King Citation1990; Bateman et al. Citation1995; Dyer et al. Citation2001), South Africa (Robbertse et al. Citation1994) and the USA (Douhan et al. Citation2002; Vera and Murray Citation2016). Studies have demonstrated a bipolar mating system governed by the MAT1 locus with two alleles MAT1-1 and MAT1-2 (Moreau and Maraite Citation1996). The frequency of both mating types influences the formation of apothecia and their potential role in the epidemiology of the disease.
During this last decade, with the increase in monoculture, farmers in Tunisia reported severe crop lodging of wheat especially in the humid areas. Initial observations in 2011 from 19 crops confirmed that eyespot was widespreadss in those areas (Gargouri et al. Citation2014). The overall objective of this study is to understand better the epidemiology of eyespot under Tunisian farming systems. Thus, the specific objectives were to conduct systematic surveys for eyespot in the four main cereal-growing regions of Tunisia, evaluate the incidence of the disease and the areas at risk, and characterize the associated pathogens by sequencing the internal transcribed spacer region (ITS) and determination of mating types using multiplex PCR.
Material and methods
Survey and sample collection
Durum wheat, bread wheat, barley and oat crops were surveyed at the early dough stage in commercial fields from 2010 to 2014 across four different climatic zones of Tunisia: the humid (annual precipitation above 800 mm), sub-humid (annual precipitation between 600 and 800 mm), upper semi-arid (annual precipitation between 500 and 600 mm) and lower semi-arid (annual precipitation between 400 and 500 mm) (Bortoli et al. Citation1969). Crops were chosen arbitrarily at approximately 10 km intervals along the main roads. GPS (latitude and longitude) coordinates were recorded for each crop and the previous crops were noted where possible (). The monthly rainfall (mm) and temperature (°C) data for 2009/10, 2010/11, 2012/13 and 2013/14 from representative locations in each zone were obtained from the National Institute of Meteorology. Fifty plants were collected randomly along a W-shaped transect in each crop, for examination in the laboratory. The plant bases were washed in the laboratory and examined visually for symptoms of eyespot. Disease incidence (percentage of diseased stems) was recorded. The severity of the disease was estimated based on the extent of browning using a discoloration rating scale of 0 to 5 on the first 30 cm of the main tiller, where: 0 = no symptoms, 1 = eyespot lesions with <25% of the stem discoloured, 2 = eyespot lesions with 25%—50% of the stem discoloured 3 = 50%—75%, 4 = eyespot lesions with 75%—100% of the stem discoloured, and 5 = eyespot lesions with discolouration >30 cm.
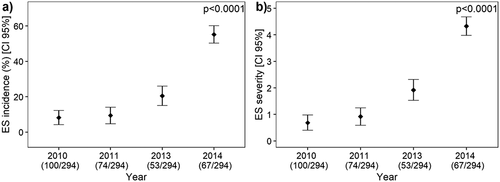
Fig. 1 Mean incidence (a) and severity (b) of eyespot (ES) in Tunisian cereal fields surveyed in 2010, 2011, 2013 and 2014.
A severity score was then calculated for each field as follows:
Where: ni is the number of plants in each severity rating from 0 to 5
Pathogen isolation
The pathogen was isolated from diseased stems collected in 2013 and 2014. Stems with eyespot symptoms were surface-disinfected with 70% ethanol for 1 min, rinsed twice in sterile water, and dried on sterile filter paper. Stem segments of approximately 0.5 cm length were cut aseptically from each of the lesions and plated on ¼-strength PDA (Potato Dextrose Agar) (Difco Laboratories, Detroit, MI). Plates were incubated at 18°C under continuous near-UV light for 1 to 2 weeks until hyphae grew from the lesion (Murray Citation1992). Hyphae of putative Oculimacula spp. with compact mycelium were subcultured onto PDA for purification. A second step of purification was carried out by diluting hyphal fragments in sterile water and then spreading 1 mL of the suspension on the surface of PDA. Single colonies were transferred aseptically to PDA in 10 mL tubes for storage at 4°C. For morphological identification, the isolates were incubated at room temperature on PDA for at least 2 weeks. Colony characteristics were considered for identification (Murray Citation1996).
DNA extraction
Seventy isolates were grown on PDA for 3 weeks at room temperature after which the surface mycelium was removed and freeze-dried. Total genomic DNA was extracted from the mycelial powder following Möller et al. (Citation1992), re-suspended in Tris-EDTA buffer (10 mM Tris, 0.1 mM EDTA, pH 8.0) and stored at −20°C.
Species identification
Two sets of primers were used for the identification of Oculimacula spp.; the primer pair Ty16/Ty16R that amplifies a 1.05 Kb fragment specific for O. yallundae and the primer pair Ta05F/Ta05R that amplifies a 0.33 Kb fragment specific for O. acuformis. These primers were generated from RAPD amplification products specific to isolates of either species (Nicholson et al. Citation1997). DNA was amplified following Nicholson et al. (Citation1997).
ITS amplification and sequencing
The primer pairs ITS1/ITS4 (White et al. Citation1990) were used to amplify the internal transcribed spacer region (ITS) of the small subunit rDNA of four isolates (TN410, TN411, TN412 and TN413). DNA was initially denatured at 94°C for 2 min, followed by 35 cycles of 94°C for 30 s, 55°C for 30 s, 72°C for 30 s, and final extension at 72°C for 10 min. PCR products were purified using a PCR purification kit (Qiagen, Valencia, CA) following the manufacturer’s protocol, then submitted to ELIM Biopharm (Hayward, CA) for sequencing. The resulting DNA sequences were used as the query in a BLAST analysis of Genbank to search for highly similar sequences (http://blast.ncbi.nlm.nih.gov/Blast.cgi).
Mating type identification
Three primers were used in multiplex PCR reactions: MT5315 (AGGAGGGCTACTGGAGGTG) common to both mating types and MT317 and MT3311 (GTTACAGCGATGACTCCAGCG and GTAATTGGACCCACGAGACAAG) specific for MAT 1–1 and MAT 1–2 DNA, respectively (Dyer et al. Citation2001). Thermocycling conditions were 94°C for 4 min; 40 cycles of 94°C for 1 min, 55°C for 1 min, 72°C for 1 min 15 s; and 72°C for 5 min. The PCR products were resolved on a 1.2% agarose gel containing SYBR Green and examined using an ultraviolet light.
Statistical analysis
Eyespot data (incidence and severity) were subjected to analysis of variance (ANOVA) using the generalized linear model (GLM) (normal distribution: identity function), followed by Least-Squares Means (LSMEANS) using JMP®11.0 (SAS Institute Inc., Cary, NC).
Results
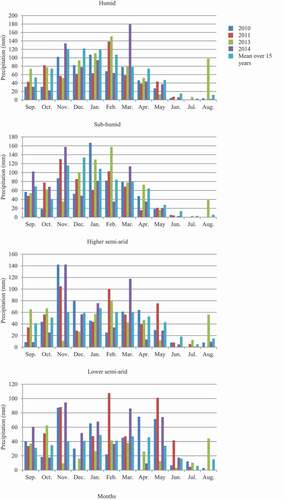
Two hundred ninety-four wheat, barley and oat fields located in the four climatic regions of Tunisia were sampled for eyespot incidence from 2010 to 2014. Over the 4-year period, eyespot was observed in 63.5% of the fields and the incidence of the disease averaged 23.1% and reached 100% in some fields in 2014. The number of fields with eyespot symptoms increased over the years from30% in 2010 to 97% in 2014 (). There was a significant increase in the incidence and severity of the disease recorded in the fields across the sampling years, with the highest incidences and severity in 2014 (, ). The humid and sub-humid regions were the most affected by eyespot with 84.2% and 69% of the crops infested, respectively, whereas the lower semi-arid was the least affected with only three fields out of the 12 surveyed showing eyespot symptoms and the incidence per field did not exceed 4% (). In contrast, all 20 fields surveyed in 2012 from the humid and sub-humid zones were affected with high incidence. The monthly rainfall and total precipitation of four stations located in the four climatic regions is represented in and , respectively. Total precipitation in wetter regions varied between 581 and 808 mm, whereas it varied between 327 and 552 in the semi-arid areas. In the cropping season 2013–2014, monthly rainfall for March recorded in the four climatic stations was higher than in the three other cropping seasons and exceeded the 15-year mean.
Table 1. Incidence of eyespot (ES) in the 294 crops surveyed during 2010, 2011, 2013 and 2014
Table 2. Percent incidence and severity of eyespot (ES) in the four climatic regions surveyed across 4 years
Table 3. Annual precipitation (mm) recorded during 2009/10, 2010/11, 2012/13 and 2013/14 cropping seasons in the humid, sub-humid, higher semi-arid and lower semi-arid regions and the mean over 15 years
Fig. 2 Monthly rainfall (mm) recorded during the 2009/10, 2010/11, 2012/13, and 2013/14 cropping seasons at meteorological stations located in the humid, sub-humid, higher semi-arid and lower semi-arid regions of Tunisia, and the mean over 15 years (Source: National Institute of Meteorology of Tunis).
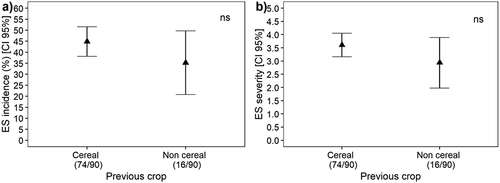
Bread wheat and durum wheat were most affected by eyespot, with 72% of bread wheat and 62% of durum wheat fields showing characteristic symptoms and disease incidence averaging 26% and 23%, respectively, whereas 30% of barley and 23% of oat fields had diseased plants, with a mean incidence of 6.1% and 2.1%, respectively (). This study did not find any effect of the previous crop (cereal vs. non-cereal) on the incidence and severity of eyespot ().
Fig. 3 Incidence (a) and severity (b) of eyespot (ES) in durum and bread wheat, oat and barley fields sampled in Tunisia in 2010, 2011, 2013 and 2014.
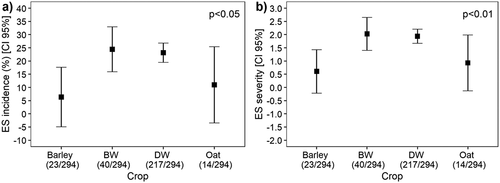
Fig. 4 Incidence (a) and severity (b) of eyespot in cereal fields grown after a cereal crop or a non-cereal crop across the sampling years 2010, 2011, 2013, 2014.
Pathogen identification
On artificial media, Oculimacula spp. grew relatively slowly and were often overgrown by Fusarium spp., mainly F. culmorum, and consistently by Phaeosphaeria pontiformis, an endophyte in some adult plants of the family Poaceae. The ITS sequences of three isolates of P. pontiformis were deposited in GenBank under the accession numbers MW188648.1, MW188649.1 and MW188650.1. All 70 isolates of Oculimacula spp. purified in this work were identified as O. yallundae based on cultural characteristic on PDA (smoky grey colonies with smooth edges). These observations were confirmed by PCR analysis where only primers specific for O. yallundae generated a fragment of expected size (1.05 Kb) in all isolates tested. Sequencing of the ITS amplicons and NCBI blast showed strain TN410 had 100% homology to O. yallundae (accession number JF412009.1). The sequence was deposited in Genbank and given the accession number MW148484.1.
Mating type identification
Mating types were identified in O. yallundae following amplification of an 812 bp MAT1-1-specific product or a 418 bp MAT1-2-specific product. Among the 70 isolates analysed, 39 were MAT1-2 and 31 were MAT1-1. In some fields, both mating types were present.
Discussion
There is a paucity of information available on the distribution of soil and residue-borne diseases and their causal agents on cereals in Tunisia. This study from 2010 to 2014 is the first systematic survey of the incidence of eyespot on cereals across four climatic regions in Tunisia. During this period, eyespot was detected in over 60% of the fields surveyed, and disease incidence within fields varied from 0% to 100% with an average of 23.1%. A wide distribution of eyespot in cereal crops has been reported previously in many regions such as the northwest of Europe (Polley and Thomas Citation1991) and the Pacific Northwest of the USA (Murray Citation1996) where the disease can reduce yield by up to 50% in winter wheat-growing areas.
Eyespot incidence increased from the semi-arid regions to the more humid areas. These increases are mostly due to the weather conditions. Indeed, the humid and sub-humid zones are characterized by annual rainfall of more than 600 mm, whereas in the semi-arid areas the rainfall is less than 500 mm. The number of fields affected with eyespot as well as the incidence and severity of the disease increased significantly over this 5-year period. Epidemics of eyespot are dependent upon the amount of primary inoculum and environmental conditions, particularly temperature and moisture (Rowe and Powelson Citation1973; Lucas et al. Citation2000). Thus, rainfall and the number of rainy days are the key factors influencing the incidence of eyespot. Matusinsky et al. (Citation2009) reported that the most important factor affecting eyespot infection in wheat is the number of days with rainfall above 3 mm from October to April, a period that covers most of cereal cycle in both countries. Similarly, Ramanauskiene and Gaurilcikiene (Citation2012) found that the number of rainy days with rainfall above 1 mm during the summer period influenced the severity of eyespot and revealed a moderately statistically significant linear correlation. In a recent study, de Mol et al. (Citation2018) found that an increasing number of days with high precipitation during the six weeks after sowing led to greater eyespot incidence. The precipitation in March of 2014 may have contributed to the increase in the incidence and severity of eyespot recorded in this study. It is not possible from this study, however, to draw conclusions on the factors that have led to the increase in incidence and severity reported here. Other factors such as increasing monoculture that favours the build-up of inoculum of Oculimacula spp., or the development of resistance in Oculimacula spp. populations to some fungicides could be involved.
In South Australia, eyespot has been causing increasing problems, the change being attributed mainly to changes in farming systems (Evans and Wallwork Citation2019). An increase in eyespot incidence has also been observed in central and northern Italy (Covarelli and Santori Citation2000; Innocenti et al. Citation2000; Covarelli and Nicholson Citation2002), with 25%-30% grain yield reductions occurring in cases of severe disease (Innocenti et al. Citation2013). The incidence of eyespot increased significantly in Europe following the development of resistance to methyl benzimidazole (MBC) fungicides (Brown et al. Citation1984; King and Griffin Citation1985).
Our results indicated that bread wheat and durum were significantly more affected than barley. Eyespot is associated with several cereal crops including wheat, barley, rye and oats, with wheat being the most susceptible (Murray et al. Citation1994; Chapman et al. Citation2008). More recently, Evans and Wallwork (Citation2015) found that in the Eyre Peninsula of South Australia, barley varieties usually have a lower incidence of eyespot than bread wheat varieties.
In this survey, it was not possible to identify an effect of the previous crop on the incidence and severity of eyespot. It is difficult in such surveys to demonstrate the role of previous crop to reduce disease incidence and severity as many other factors may influence the outcomes.
The species O. yallundae was the only causal agent of eyespot identified in this study. Both O. yallundae and O. acuformis are associated with eyespot disease in most cereal growing areas in the world, except South Africa, where only O. yallundae was identified (Lucas et al. Citation2000). Recently in Lithuania, O. acuformis was found to be the dominant species on Triticale (Ramanauskien and Gaurilcikiene Citation2016). In many regions of Europe, O. yallundae has been replaced by O. acuformis as the dominant species in winter wheat crops (Lucas et al. Citation2000; Glazek Citation2009). A similar increase has also been reported in the Pacific Northwest of the USA (Douhan et al. Citation2002). The apparent shift in eyespot populations from O. yallundae to O. acuformis in many parts of Europe has been attributed to differential resistance to fungicides and host preference (Birchmore et al. Citation1992; Bateman Citation2002; Bierman et al. Citation2002; Blein et al. Citation2009). An alternative explanation for the increase in frequency of O. acuformis in the Pacific Northwest may be its ability to better colonize winter wheat cultivars with eyespot resistance than O. yallundae. It has also been suggested that the production of more winter barley in Europe may have selected for O. acuformis, which is equally pathogenic on wheat, barley and rye, whereas O. yallundae is less virulent on barley and rye than on wheat (Hollins et al. Citation1985).
More recently, however, there is evidence that the prevalence of O. acuformis over O. yallundae has declined. Parnell et al. (Citation2008) suggested that this might be due to the reduction in the use of prochloraz and an increase in the use of the anilinopyrimidine fungicide cyprodinil, which is selective for O. yallundae. Such population shifts illustrate the necessity of regular surveys so that data are available to inform the most appropriate integrated management strategies for the disease.
More intensive sampling is needed to determine better the relative importance of each species in the Tunisian farming system. Several differences have been reported between the pathogens such as host pathogenicity (Hollins et al. Citation1985), infection strategy (Daniels et al. Citation1991), colonization mode (Blein et al. Citation2009), the effectiveness of plant resistance genes(Burt et al. Citation2010) and fungicide sensitivity (Bierman et al. Citation2002; Parnell et al. Citation2008). Thus, accurate identification of the species associated with eyespot and their relative prevalence is crucial for designing improved management strategies including fungicides, genetic resistance and cultural practices.
This survey revealed the presence of both mating types of O. yallundae in Tunisia and sometimes in the same field, with a slightly greater percentage of MAT1-2. Both mating types have been also observed in the same field and in other regions including Europe, New Zealand, and the USA (Dyer et al. Citation1996, Citation2001; Douhan et al. Citation2002).These findings support the potential occurrence of sexual reproduction in Tunisian fields. However, reproductive inferences drawn from mating types should be made with caution. Some sexual species have a strong mating type skew with a dominance of one mating type (Yan et al. Citation2002). The presence of both MAT1-1 and MAT1-2 isolates has been reported in field populations of supposedly asexual fungi for which no sexual stage has yet been described (Arie et al. Citation2000; Tóth et al. Citation2004). In addition, low levels of fertility may also be observed in some fungal species despite the occurrence of both mating types (Aoki and O’Donnell Citation1999; Bentley et al. Citation2008). While the presence of mating types with equal frequencies is a strong evidence of sexual reproduction, this should be coupled with population genetics studies that demonstrate recombination is actually occurring (Nieuwenhuis and James Citation2016).The ability of O. yallundae and O. acuformis to reproduce sexually also will depend on factors other than the presence of both mating types within a population, such as the presence of suitable substrates and conducive environmental conditions. The frequency of sexual reproduction under Tunisian conditions needs further study, including evaluation of mating type ratio in a larger population, investigation of genetic diversity and the population structure of O. yallundae, and evaluation of the occurrence, distribution and persistence of apothecia in commercial fields. Such information is particularly important to improve understanding of the biology and the potential adaptability of O. yallundae and eyespot epidemiology in Tunisian farming systems, serving as the basis for developing integrated disease management strategies.
Acknowledgements
The first author is grateful to the Fulbright Foundation for the award of a Research fellowship (July 2015–October 2015) at Washington State University.
Disclosure statement
No potential conflict of interest was reported by the author(s).
Additional information
Funding
References
- Aoki T, O’Donnell K. 1999. Morphological characterization of Gibberella coronicola sp. nov., obtained through mating experiments of Fusarium pseudograminearum. Mycoscience. 40(6):443–453. doi:https://doi.org/10.1007/BF02461021.
- Arie T, Kaneko I, Yoshida T, Noguchi M, Nomura Y, Yamaguchi I. 2000. Mating-type genes from asexual phytopathogenic ascomycetes Fusarium oxysporum and Alternaria alternata. Mol Plant Microbe Interact. 13(12):1330–1339. doi:https://doi.org/10.1094/MPMI.2000.13.12.1330.
- Bateman GL. 2002. Long-term performance of fungicides applied to control eyespot in winter wheat. Ann Appl Biol. 141(1):29–33. doi:https://doi.org/10.1111/j.1744-7348.2002.tb00192.x.
- Bateman GL, Dyer PS, Manzhula L. 1995. Development of apothecia ofTapesia yallundae in contrasting populations selected by fungicides. Eur J Plant Pathol. 101(6):695–699. doi:https://doi.org/10.1007/BF01874874.
- Bentley AR, Summerell BA, Burgess LW. 2008. Sexual compatibility in Fusarium pseudograminearum (Gibberella coronicola). Mycol Res. 112(9):1101–1106. doi:https://doi.org/10.1016/j.mycres.2008.04.016.
- Bierman SM, Fitt BDL, Van Den Bosch F, Bateman GL, Jenkyn JF, Welham SJ. 2002. Changes in populations of the eye spot fungi Tapesia yallundae and T. acuformis under different fungicide regimes in successive crops of winter wheat, 1984-2000. Plant Pathol. 51(2):191–201. doi:https://doi.org/10.1046/j.1365-3059.2002.00673.x.
- Birchmore RJ, Ashman PI, Stanley S, and Russell PE. 1992. Effects of crop history on sensitivity to prochloraz of Pseudocercosporella herpotrichoides isolates from cereals in Western Europe. Brighton Crop Protection Conference – Pests and Diseases (Brighton, England). p. 183–189.
- Blein M, Levrel A, Lemoine J, Gautier V, Chevalier M, Barloy D. 2009. Oculimacula yallundae life style revisited: relationships between the timing of eyespot symptom appearance, the development of the pathogen and the responses of infected partially resistant wheat plants. Plant Pathol. 58(1):1–11. doi:https://doi.org/10.1111/j.1365-3059.2008.01940.x.
- Bortoli L, Gounot M, Jacquinet JC. 1969. Climatologie et bioclimatologie de la Tunisie septentrionale. Annales de l’INRA, Tunisie. 42:1–236.
- Brown MC, Taylor GS, Epton HAS. 1984. Carbendazim resistance in the eyespot pathogen Pseudocercosporella herpotrichoides. Plant Pathol. 33(1):101–111. doi:https://doi.org/10.1111/j.1365-3059.1984.tb00593.x.
- Burt C, Hollins TW, Powell N, Nicholson P. 2010. Differential seedling resistance to the eyespot pathogens, Oculimacula yallundae and Oculimacula acuformis, conferred by Pch2 in wheat and among accessions of Triticum monococcum. Plant Pathol. 59(5):819–828. doi:https://doi.org/10.1111/j.1365-3059.2010.02307.x.
- Chapman N, Burt C, Dong H, Nicholson P. 2008. The development of PCR-based markers for the selection of eyespot resistance genes Pch1 and Pch2. Theor Appl Genet. 117(3):425–433. doi:https://doi.org/10.1007/s00122-008-0786-3.
- Covarelli L, Nicholson P. 2002. Identificazione dei patogeni responsabili del mal del piede del frumento mediante PCR. Atti Giornate Fitopatologiche. 2:449‒454.
- Covarelli L, Santori A. 2000. Influenza dell’epoca di semina e di trattamenti fungicid isul mal del piede e sullaseptoriosi del frumentotenero in condizioni di momosuccession e prolungata. Atti Giornate Fitopatologiche. 2:253‒258.
- Crous PW, Groeneward JZ, Gams W. 2003. Eyespot of cereals revisited: ITS phylogeny reveals new species relationships. Eur J Plant Pathol. 109(8):841–850. doi:https://doi.org/10.1023/A:1026111030426.
- Daniels A, Lucas JA, Peberdy JF. 1991. Morphology and ultrastructure of W and R pathotypes of Pseudocercosporella herpotrichoides on wheat seedlings. Mycol Res. 95(4):385–397. doi:https://doi.org/10.1016/S0953-7562(09)80834-4.
- de Mol F, Winter M, Gerowitta B. 2018. Weather determines the occurrence of wheat stem base diseases in biogas cropping systems. Crop Protec. 114:1–11. doi:https://doi.org/10.1016/j.cropro.2018.07.017
- Douhan GW, Murray TD, Dyer PS. 2002. Species and mating type distribution of Tapesia yallundae and T. acuformis and occurrence of apothecia in the US Pacific Northwest. Phytopathology. 92(7):703–709. doi:https://doi.org/10.1094/PHYTO.2002.92.7.703.
- Dyer PS, Bateman GL, Wood HM. 2001. Development of apothecia of the eyespot pathogen Tapesia on cereal crop stubble residue in England. Plant Pathol. 50:356–362. doi:https://doi.org/10.1046/j.1365-3059.2001.00575.x
- Dyer PS, Nicholson P, Lucas JA, Peberdy JF. 1996. Tapesia acuformis as a causal agent of eyespot disease of cereals and evidence for a heterothallic mating system using molecular markers. Mycol Res. 100(10):1219–1226. doi:https://doi.org/10.1016/S0953-7562(96)80184-5.
- Evans M, Wallwork H. 2015. Eyespot – variety tolerance and fungicide efficacy. Eyre Peninsula Farming Syst. 2015(Summary):97–100.
- Evans M, Wallwork H. 2019. Eyespot of winter cereals – an emerging disease. https://communities.grdc.com.au/field-crop-diseases/eyespot-winter-cereals/.
- Gargouri S, Bouatrous A, Debchi R, Fakhfakh M. 2014. Confirmation of Oculimacula yalundae causing eyespot of cereals in Tunisia. New Dis Rep. 29(1):10. doi:https://doi.org/10.5197/j.2044-0588.2014.029.010.
- Glazek M. 2009. Occurrence of eyespot on winter wheat in the central-southern region of Poland. J Plant Prot Res. 49(4):426–433. doi:https://doi.org/10.2478/v10045-009-0068-3.
- Hollins TW, Scott PR, Paine JR. 1985. Morphology, benomyl resistance and pathogenicity to wheat and rye of isolates of Pseudocercosporella herpotrichoides. Plant Pathol. 34(3):369–379. doi:https://doi.org/10.1111/j.1365-3059.1985.tb01375.x.
- Innocenti G, Montanari M, Marenghi A, Toderi G. 2000. Ramulispora herpotrichoides in cereal vernini in diverse situazionicolturali. Atti Giornate Fitopatologiche. 2:241‒246.
- Innocenti G, Roberti R, Alvisi G. 2013. Efficacy of foliage fungicides against eyespot of winter wheat in Northern Italy. Phytopathol Mediterr. 52:442–448. doi:https://doi.org/10.14601/Phytopathol_Mediterr-11069
- King AC. 1990. The first report of Tapesia yallundae as the teleomorph of Pseudocercosporella herpotrichoides var. acuformis and its occurrence in the Federal Republic of Germany. Plant Pathol. 39:44–49. doi:https://doi.org/10.1111/j.1365-3059.1990.tb02474.x
- King JE, Griffin MJ. 1985. Survey of benomyl resistance in Pseudocercosporella herpotrichoides on winter wheat and barley in England and Wales in 1983. Plant Pathol. 34(2):272–283. doi:https://doi.org/10.1111/j.1365-3059.1985.tb01359.x.
- Lucas JA, Dyer PS, Murray TD. 2000. Pathogenicity, host specificity, and population biology of Tapesias pp., causal agents of eyespot disease of cereals. Adv Bot Res. 33:225–258. doi:https://doi.org/10.1016/S0065-2296(00)33044-0
- Matusinsky P, Mikolasova R, Klem K, Spitzer T. 2009. Eyespot infection risks on wheat with respect to climatic conditions and soil management. J Plant Pathol. 91:93–101.
- Möller EM, Bahnweg G, Sandermann H, Geiger HH. 1992. A simple and efficient protocol for isolation of high molecular weight DNA from filamentous fungi, fruit bodies, and infected plant tissues. Nucleic Acids Res. 20(22):6115–6116. doi:https://doi.org/10.1093/nar/20.22.6115.
- Moreau JM, Maraite H. 1996. Evidence for a heterothallic mating system in Tapesia acuformis using benomyl sensitivity and esterase isoenzyme profiles. Mycol Res. 100(10):1227–1236. doi:https://doi.org/10.1016/S0953-7562(96)80185-7.
- Murray TD. 1996. Resistance to benzimidazole fungicides in the cereal eyespot pathogen, Pseudocercosporella herpotrichoides, in the Pacific Northwest 1984-1990. Plant Dis. 80(1):19–23. doi:https://doi.org/10.1094/PD-80-0019.
- Murray TD. 2010. Eyespot (strawbreaker foot rot). In: Bockus WW, Bowden RL, Hunger RM, Murray TD, Smiley RW, Morrill W, editors. Compendium of wheat diseases and insects. 3rd ed. St. Paul (MN): APS Press; p. 32–34.
- Murray TD, De La Penă RC, Yildirim A, Jones SS. 1994. A new source of resistance to Pseudocercosporella herpotrichoides, cause of eyespot disease of wheat, located on chromosome 4V of Dasypyrum villosum. Plant Breed. 113(4):281–286. doi:https://doi.org/10.1111/j.1439-0523.1994.tb00737.x.
- Murray TD. 1992. Pseudocercosporella. In: Singleton L, Ruch CM, Mihail JD, editors. Methods for research on soilborne phytopathogenic fungi. St. Paul (MN): APS Press; p. 149–152.
- Nicholson P, Rezanoor HN, Simpson DR, Joyce D. 1997. Differentiation and quantification of the cereal eyespot fungi Tapesia yallundae and Tapesia acuformis using a PCR assay. Plant Pathol. 46(6):842–856. doi:https://doi.org/10.1046/j.1365-3059.1997.d01-74.x.
- Nieuwenhuis BPS, James TY. 2016. The frequency of sex in fungi. Phil Trans R Soc B. 371(1706):20150540. doi:https://doi.org/10.1098/rstb.2015.0540.
- Parnell S, Gilligan CA, Lucas JA, Bock CH, Van Den Bosch F. 2008. Changes in fungicide sensitivity and relative species abundance in Oculimacula yallundae and O. acuformis populations (eyespot disease of cereals) in Western Europe. Plant Pathol. 57(3):509–517. doi:https://doi.org/10.1111/j.1365-3059.2007.01786.x.
- Polley RW, Thomas MR. 1991. Surveys of diseases of winterwheat in England and Wales, 1976-1988. Ann Appl Biol. 119(1):1–20. doi:https://doi.org/10.1111/j.1744-7348.1991.tb04839.x.
- Ramanauskien J, Gaurilcikiene I. 2016. Incidence of eyespot in winter wheat and quantification of the fungi Oculimacula acuformis and O. yallundae in Lithuania. J Plant Dis Prot. 123(2):75–81. doi:https://doi.org/10.1007/s41348-016-0013-4.
- Ramanauskiene J, Gaurilcikiene I. 2012. The peculiarities of winter wheat eyespot management under Lithuanian conditions. Commun Appl Biol Sci. 77:287–295.
- Robbertse B, Crous PW, Holz G. 1994. Tapesia yallundae collected from wheat stubble in South Africa. Mycopathologia. 125(1):23–28. doi:https://doi.org/10.1007/BF01103971.
- Rowe RC, Powelson RL. 1973. Epidemiology of Cercosporella foot rot of wheat: disease spread. Phytopathology. 63(8):984–988. doi:https://doi.org/10.1094/Phyto-63-984.
- Tóth B, Mesterházy Á, Nicholson P. 2004. Mycotoxin production and molecular variability of European and American isolates of Fusarium culmorum. Eur J Plant Pathol. 110(5/6):587–599. doi:https://doi.org/10.1023/B:EJPP.0000032398.74570.ab.
- Vera DI, Murray TD. 2016. Occurrence and survival of apothecia of the eyespot pathogens Oculimacula acuformis and O. yallundae on wheat stubble in the U.S. Pacific Northwest. Plant Dis. 100(5):991–995. doi:https://doi.org/10.1094/PDIS-09-15-1056-RE.
- Wallwork H. 1987. A Tapesia teleomorph for Pseudocercosporella herpotrichoides, the cause of eyespot of wheat. Australas Plant Pathol. 16(4):92–93. doi:https://doi.org/10.1071/APP9870092.
- White TJ, Bruns T, Lee S, Taylor J. 1990. Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In: Innis MA, Gelfand DH, Sninsky JJ, White TJ, editors. PCR protocols, a guide to methods and applications. San Diego (CA): Academic Press; p. 315–322.
- Yan Z, Li X, Xu J. 2002. Geographic distribution of mating type alleles of Cryptococcus neoformans in four areas of the United States. J Clin Microbiol. 40(3):965–972. doi:https://doi.org/10.1128/JCM.40.3.965-972.2002.
