Abstract
Purpose
To examine predictors of health-related quality of life (HRQoL) in Hispanic and non-Hispanic White (NHW) breast cancer (BC) survivors.
Design
Cross-sectional study using survey data.
Participants
Women diagnosed with BC at ages 21-79 years, between 2012-2014, recruited from the New Jersey State Cancer Registry.
Methods
HRQoL was assessed using the Functional Assessment Cancer Therapy (FACT-G) instrument. Descriptive statistics compared Hispanics and NHWs, and multivariate regression analyses identified predictors of HRQoL.
Results
HRQoL was significantly higher scores among NHW (85.7 ± 18.5) than Hispanics (79.4 ± 20.1) (p < 0.05). In multivariate analyses, comorbidities (β: −13.3, 95%CI: −20.6, −5.92), late-stage diagnosis (β: −5.67, 95%CI: −10.7, −0.62), lower income (β: −13.9, 95%CI: −19.8, −7.97) and younger age at diagnosis were associated with lower HRQoL.
Conclusion
Socio-demographic and clinic characteristics were significant predictors of HRQoL among diverse BC survivors.
Implications for Psychosocial Oncology
Supportive psychosocial care interventions tailored to the needs of young, low-income BC survivors with comorbidities are needed.
Introduction
Although there are more than 3.8 million women with breast cancer in the United States,Citation1 post-treatment survivors are at risk for recurrence, secondary cancers, and late effects of cancer and its treatment.Citation2 These increased risks, including physical and psychosocial sequelae, have the potential to negatively affect survivors’ HRQoL,Citation2 that is, their perceived physical, functional, social and emotional well-being over time.Citation3 Breast cancer survivors are more likely to experience impaired physical functioningCitation4 and diminished emotional and social well-being due to cancer and its treatment when compared to the general population.Citation5,Citation6 Furthermore, after completing cancer treatment, breast cancer survivors experience increased distress and fear about post-treatment life, loss of support from family and friends and struggle with cancer-related physical and psychological issues.Citation7,Citation8
Factors known to contribute to HRQoL among breast cancer survivors include age at diagnosis, presence of multiple comorbidities, cancer stage, cancer treatment, marital and socio-economic status and social support and coping strategies.Citation9–13 However, most HRQoL studies include non-population based samples and have focused on primarily non-Hispanic White (NHW) breast cancer survivors in large urban settings.Citation9,Citation11,Citation14–16 New Jersey is one of the most racially/ethnically diverse states in the United StatesCitation17; and Hispanics are the largest racial/ethnic minority group, comprising 20.9% of the population.Citation18 Most Hispanics in New Jersey are of Puerto Rican, South American, Dominican Republic, Mexican and Central American descent.Citation19 Counties such as Hudson, Middlesex, Essex, Passaic, Union and Camden have municipalities where Hispanics are the majority (>50%) population.Citation20 Moreover, 77.7% of Hispanics in New Jersey speak at least Spanish and 58.1% are bilingual.Citation21
Socio-economic status and racial/ethnic disparities in breast cancer morbidity persist in New Jersey. For instance, women residing in high poverty areas are diagnosed at more advanced stages of breast cancer compared to women in the wealthiest areas, and Black women have significantly lower breast cancer survival rates than NHWs.Citation22 Although breast cancer’s specific mortality rates in New Jersey are similar for Hispanic and NHW survivors,Citation22 HRQoL of Hispanic women might be lower, as higher proportions of women in this population live in poverty compared to the women of other racial/ethnic groups.Citation17
Given that, well-documented racial/ethnic disparities in cancer diagnoses, treatment and outcomes, concern for potential disparities in HRQoL during survivorship are warranted.Citation12,Citation23–26 Thus far, research on HRQoL comparing experiences of Hispanic versus NHW breast cancer survivors have yielded mixed results.Citation24,Citation26–28 Studies investigating problem-specific issues indicate that Hispanic breast cancer survivors have additional concerns and challenges compared to NHWs, such as, job disruptions and financial hardship due to cancer and its treatment, and higher concerns about physical pain and survival than their NHW peers,Citation24,Citation26,Citation27 which could negatively impact their HRQoL.Citation24 In addition, a study focused primarily on married, employed and highly educated survivors conducted in New Jersey and New York found that Hispanic breast cancer survivors experience lower HRQoL than NHWs.Citation29 A recent systematic review that compared HRQoL of Hispanic breast cancer survivors to HRQoL of women of other racial/ethnic groups reported mixed findings, but concluded that, overall, Hispanic women demonstrated lower HRQoL than non-Hispanics.Citation28 However, other studies conducted among racially/ethnically diverse breast cancer survivors have indicated that race/ethnicity is not a significant predictor of HRQoL.Citation30,Citation31 Thus, these mixed reports could be due to variations in study designs, sampling methods and overall limited inclusion of racial/ethnic minorities in breast cancer survivorship research.Citation12,Citation15,Citation23,Citation28,Citation31
The purpose of this cross-sectional study is to identify and compare socio-demographic and clinical predictors of HRQoL among Hispanic and NHW breast cancer survivors (one to four years post-diagnosis) in New Jersey. We hypothesize that, Hispanic breast cancer survivors will report lower HRQoL than NHW breast cancer survivors. This research extends the cancer survivorship literature by exploring and comparing HRQoL predictors among Hispanic and NHW breast cancer survivors in New Jersey.
Methods
Participants and procedure
Data were obtained from the Improving Patient Access to Quality Cancer Treatment (IMPACT) study, a cross-sectional mailed survey conducted to evaluate breast, cervical, prostate and colorectal cancer survivors’ access to quality care, treatment and health outcomes. The study recruited participants through the New Jersey State Cancer Registry (NJSCR), a statewide population-based cancer registry, between September 2015 and August 2016. Details of the study have been reported previously.Citation32 Stratified random sampling by cancer site and year of diagnosis were used to identify potential study participants for this study. Eligibility criteria for breast cancer survivors were: (1) diagnosis between 2012 and 2014; (2) a primary, non-metastatic breast cancer with no previous history of cancer; (3) cancer stages I, II, III at diagnosis; (4) aged 21–79 years at diagnosis; (5) alive at time of contact and residing in New Jersey at diagnosis; and (6) able to read and write English. Participants were ineligible if they were enrolled in another NJSCR study, requested to be excluded from research studies, or were unable to participate due to a physician’s assessment of mental health issues or other reasons. Eligible participants (n = 2366) received a mailed self-administered cancer-specific survey (∼75 items). Thirty percent (n = 706) of eligible participants (breast, colorectal, prostate and cervical cancer survivors) completed and returned the survey and received a $15 gift card. The current analysis included only female breast cancer survivors who returned a completed survey and self-identified as Hispanic or NHW (n = 259).
Theoretical framework
The Contextual Model of HRQoL, a theoretical framework that includes individual-level (i.e., general health status, cancer-related medical conditions, health literacy and psychological well-being) and systemic-level factors (i.e., socio-ecological, cultural, demographic and health care system), was used to identify and explain predictors of HRQoL among racially/ethnically diverse breast cancer survivors.Citation33 The model is informed by the traditional HRQoL model, bio-psychosocial model, literature reviews from qualitative and quantitative studies with cancer survivors, the cancer and survivorship literature and the multicultural and psychological literature.Citation33 The model can be used to evaluate an individual survivor’s risk for low HRQoL and identify racial/ethnic group disparities in HRQoL outcomes.
Measures
Outcome variables
The FACT-G (version 4) instrument assessed total HRQoL.Citation34 The instrument includes four subscales that evaluate physical well-being (PWB), social/family well-being (SWB), functional well-being (FWB) and emotional well-being (EWB). Subscale scores can be reported individually and/or summed to derive a total score with higher scores indicating better HRQoL. The standard FACT-G total score ranges from 0 to 108 and each subscale range is between 0 and 28 for SWB, PWB, and FWB and between 0 and 24 for EWB. According to FACT-G scoring guidelines, missing data in each subscale can be handled by multiplying the sum of the item scores by the total number of items, then dividing by the number of items answered.Citation35 High reliability (Cronbach’s alpha = 0.90) has been reported for the instrument.Citation36
Independent variables
Socio-demographic, general health status and tumor-related characteristics were included as independent variables based on the Contextual Model of HRQoL framework.
Socio-demographic characteristics were obtained from a self-administered survey and included marital status, health insurance at diagnosis, household income and education. Marital status was categorized as married (married or living as married) and unmarried (divorced, widowed, separated and single). Health insurance at diagnosis was categorized as insured (all insurance types) and uninsured (no insurance). Annual household income was coded as < $50,000 and ≥ $50,000. Education was coded as > high school and ≤ high school.
General health status variables included comorbidities and body mass index (BMI). Comorbidities were obtained using the Charlson comorbidity index (CCI) and recoded as none, one, and two or more. BMI was calculated by dividing weight in pounds by height in inches squared and multiplying by 703.Citation37 Participants were classified into three groups based on weight status: normal weight (BMI: 24.9 kg/m2 and less), overweight (BMI: 25.0 kg/m2 to 29.9 kg/m2) and obese (BMI: 30 kg/m2 or higher).Citation37 Tumor-related characteristics (cancer stage at diagnosis, age at diagnosis, time since diagnosis) were obtained from NJSCR. Cancer stage at diagnosis was categorized as early (stage I) and late (stages II–III). Age at diagnosis was a continuous variable. Time since diagnosis was a proxy variable created by subtracting the difference (in years) between date of survey completion and date of cancer diagnosis.
Statistical analysis
Descriptive statistics were used to compare NHW and Hispanic breast cancer survivors’ individual and systemic-level characteristics and HRQoL measures. The Shapiro–Wilk test of normality revealed non-normality in data distributions. Hence, differences between the two groups were evaluated using the non-parametric Wilcoxon rank-sum test for continuous variables and Fisher’s exact test for categorical variables.
Ordinary least square (OLS) multivariate regression was used to assess the effect of individual and systemic-level predictors on HRQoL outcomes in Hispanic and NHW breast cancer survivors. Five separate OLS multivariate regression analyses were conducted to predict total HRQoL (FACT-G) and the four domains of HRQoL (i.e. PWB, FWB, SWB, EWB). All five final regression models included predictors based on theoretical and contextual importance. In addition, for each OLS regression model, cases with missing data were omitted (n = 56 or 22%) and robust standard error was used to reduce heterogeneity of error terms. All statistical tests were conducted at a two-sided significance level of 0.05 using R software (version 3.5.0).
Results
Sample characteristics
displays socio-demographic and clinic characteristics of breast cancer survivors (N = 259) overall and by racial/ethnic group. There were significant differences by race/ethnicity in insurance at diagnosis and annual household income. Almost 87% of NHW breast cancer survivors had insurance at diagnosis compared to 65.4% of Hispanics (p < 0.001). Similarly, a higher proportion of NHW (53.6%) had an annual household income of ≥ $50,000 compared to 20.4% of Hispanics (p < 0.001). In addition, 56% of survivors were married and 66% had more than a high school education. More than half (53.7%) of Hispanic women were within one to two years post-diagnosis, whereas only 29.7% of NHW women were within one to two years post-diagnosis (p < 0.001). Over half of survivors (51%) had at least one comorbidity and 72% were overweight or obese. Most survivors (70%) had early-stage (stage I) breast cancer. Average age at diagnosis was 55.2 ± 11.8 years and NHW survivors were older than Hispanic survivors at diagnosis (56.8 vs. 48.9 years, respectively; p < 0.001).
Table 1. Socio-demographic and clinical characteristics of Hispanic and non-Hispanic White breast cancer survivors (N = 259).
shows that the total HRQoL (FACT-G) score was significantly higher among NHW (85.7 ± 18.5) compared to Hispanic survivors (79.4 ± 20.1) (p < 0.05). NHW survivors also reported higher social well-being (22.3 ± 6.5) compared to Hispanic survivors (21.8 ± 5.4) (p < 0.01). Similarly, emotional well-being was higher among NHW survivors (19.3 ± 4.6) compared to Hispanic survivors (17.3 ± 5.4) (p < 0.01). There were no significant differences in physical and functional well-being by race/ethnicity ().
Figure 1. Health-related Quality of Life (HRQoL) mean scores by race/ethnicity. FACT-G includes total well-being scores. PWB, physical well-being; FWB, functional well-being; SWB, social well-being; and EWB, emotional well-being. * p < 0.05.
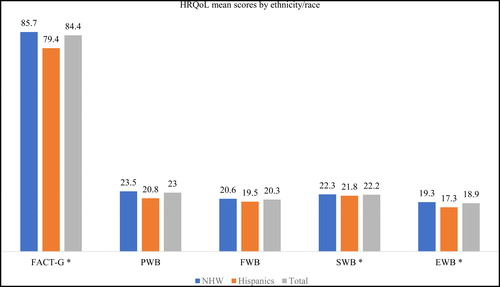
Table 2. Multivariate regression results for total HRQL (FACT-G) and four domains of HRQL (N = 203).
Multivariable regression analyses
Total HRQoL (FACT-G)
In the adjusted multivariable model for total HRQoL, increase in age at diagnosis, early stage cancer, shorter time since diagnosis, higher household income and having no comorbidities were associated with increased total quality of life . Total HRQoL improved with every one-year increase in age at diagnosis (β = 0.42, 95% CI: 0.19, 0.66); survivors within two to three years post-diagnosis had a higher total HRQoL (β = 6.74, 95% CI: 0.11, 13.4) compared to those three to four years post-diagnosis. Late-stage cancer (β = −5.67, 95% CI: −10.7, −0.62) compared to early-stage cancer, annual household income < $50,000 (β = −13.9, 95% CI: −19.8, −7.97) compared to ≥ $50,000 and two or more comorbidities (β = −13.3, 95% CI: −20.6, −5.92) compared to none were significantly associated with lower total HRQoL. Education, marital status, health insurance at diagnosis, race/ethnicity and BMI were not significant predictors of total HRQoL.
Physical well-being
In the adjusted multivariable model for physical well-being, increase in age at diagnosis, higher household income, being uninsured at diagnosis and having no comorbidities were associated with higher physical well-being . Physical well-being improved with every one-year increase in age at diagnosis (β = 0.11, 95% CI: 0.04, 0.18); being uninsured at diagnosis (β = 1.95, 95% CI: 0.01, 3.89) compared to being insured was also associated with higher physical well-being. Lower physical well-being was significantly predicted by annual household income <$50,000 (β = −3.76, 95% CI: −5.49, −2.04) compared to ≥ $50,000 and having two or more comorbidities (β = −2.92, 95% CI: −5.28, −0.56) compared to none. No other socio-demographic or clinical characteristics significantly predicted physical well-being.
Functional well-being
In the adjusted multivariable model for functional well-being, increase in age at diagnosis, early stage cancer, shorter time since diagnosis, higher household income and having no comorbidities were associated with higher functional well-being. Functional well-being improved with every one-year increase in age at diagnosis (β = 0.11, 95% CI: 0.03, 0.19); survivors within two to three years post-diagnosis had higher functional well-being (β = 2.57, 95% CI: 0.30, 4.85) than those three to four years post-diagnosis. Lower functional well-being was significantly predicted by late-stage cancer (β = −2.46, 95% CI: −4.32, −0.60) compared to early-stage cancer, annual household income < $50,000 (β = −4.99, 95% CI: −7.02, −2.95) compared to ≥ $50,000 and having two or more comorbidities (β = −3.77, 95% CI: −6.22, −1.31) compared to none. Education, marital status, health insurance at diagnosis, race/ethnicity and BMI were not significant predictors of functional well-being.
Social well-being
In the adjusted multivariable model for social well-being, increase in age at diagnosis, shorter time since diagnosis, higher household income, being married, and having no comorbidities were associated with higher social well-being. Social well-being improved with every one-year increase in age at diagnosis (β = 0.10, 95% CI: 0.02, 0.18); survivors within one to two years post-diagnosis had higher social well-being (β = 2.49, 95% CI: 0.24, 4.73) than those three to four years post-diagnosis. Lower social well-being was significantly predicted by annual household income < $50,000 (β = −3.03, 95% CI: −5.13, −0.93) compared to ≥ $50,000 being unmarried (β = −2.76, 95% CI: −4.73, −0.79) compared to being married, and two or more comorbidities (β = −3.23, 95% CI: −5.56, −0.91) compared to none. Cancer stage, education, health insurance at diagnosis, race/ethnicity, and BMI were not significant predictors of social well-being.
Emotional well-being
In the adjusted multivariable model for emotional well-being, increase in age at diagnosis, higher household income and having no comorbidities were associated with higher emotional well-being. Emotional well-being improved with every one-year increase in age at diagnosis (β = 0.10, 95% CI: 0.04, 0.17). Annual household income < $50,000 (β = −2.12, 95% CI: −3.72, −0.53) compared to ≥ $50,000 and two or more comorbidities (β = −3.34, 95% CI: −5.19, −1.49) compared to none were significant predictors of lower emotional well-being. No other socio-demographic and clinical characteristics were significant predictors of emotional well-being.
Discussion
This study examined socio-demographic and clinical predictors of HRQoL in Hispanic and NHW breast cancer survivors in New Jersey. Results indicated lower total HRQoL among Hispanic and NHW breast cancer survivors compared to other studies that have examined similar racial/ethnic groups.Citation10,Citation23 Although Hispanics (vs NHWs) had lower social and emotional well-being scores in the bivariate analysis, there were no significant differences in total HRQoL by race/ethnicity in the adjusted analyses. However, we observed lower reported social and emotional well-being scores in our sample of Hispanic women than those in other studies of Hispanic survivors.Citation10,Citation23 Results could be due to geographic-specific socio-economic barriers experienced by Hispanic survivors in this study and larger contextual issues. For instance, among Hispanics in New Jersey, 6.5% do not speak any English and 13.1% do not speak English well,Citation21 which could further limit their ability to communicate with non-language concordant providers. Further, the Hispanic population in the US is more likely to experience discrimination in healthcare settings compared to their NHW counterparts and less likely to seek health care due to anticipated discrimination,Citation38 which may elevate their stress levels and decrease emotional well-being.
In this study, younger age at diagnosis, lower annual household income and having two or more comorbidities were predictors of lower HRQoL across all four domains (i.e. physical, functional, social and emotional well-being). Overall, total HRQoL improved with every one-year increase in age at diagnosis. The negative impact of breast cancer and its treatment on the well-being of younger breast cancer survivors has been reported in other research studies.Citation12,Citation30,Citation39–42 This could be due to higher psychological distress or physical and sexual problems experienced by younger women with breast cancer.Citation30,Citation41,Citation42 Young breast cancer survivors tend to have greater life disruptions after cancer, such as, changes in child-rearing activities, concerns about career and work, and uncertainty about cancer recurrence.Citation9,Citation30,Citation41 Furthermore, younger women may receive more aggressive treatments than older women, compromising their physical and functional well-being.Citation42 They might also experience adverse reproductive and sexual health effects of cancer treatments, such as, entering early menopause, having fertility concerns and body image issues.Citation41,Citation42 Hence, this study confirms and extends the research findings that younger breast cancer survivors experience more physical, functional, social, and emotional concerns, and report lower HRQoL than older women with breast cancer.
Having an annual household income of < $50,000 (versus ≥$50,000) was a predictor of lower physical, functional, social and emotional well-being. Multiple studies have found that breast cancer survivors of low socio-economic status might experience psychosocial distress including clinical depression and anxiety, financial hardship and lower total HRQoL.Citation23,Citation43–45 Possible explanations for the negative effects of low socio-economic status on HRQoL outcomes could be due to lack of access to material and social resources such as healthcare services, dietary supplements, transportation and recreational centers.Citation13 Low-income women often lack the sense of financial security, personal control and reduced stress level that characterize women of high socio-economic status.Citation13
Consistent with other studies, having two or more comorbidities (versus no comorbidities) was also associated with lower physical, functional, social and emotional well-being.Citation23,Citation39,Citation46 Compared to the general population, breast cancer survivors are at higher risk of developing chronic conditions such as obesity, hypertension, diabetes and low bone mass due to the late effects of cancer treatment.Citation46–48 Breast cancer survivors with multiple comorbidities also are more likely to experience prolonged hospitalizations and in-patient death than breast cancer survivors without comorbidities.Citation46 Studies suggest that, survivors with comorbidities require additional guidance on post-treatment to understand the broader implications of cancer and its treatment on their overall health.Citation49 Thus, the high burden of multiple comorbidities may be associated with higher health care needs, increased cost of care and disability, and decreased physical functioning,Citation50 impacting breast cancer survivors’ HRQoL.
Stage at diagnosis was also predictive of total HRQoL and functional well-being: women diagnosed with late-stage breast cancer reported lower functional well-being than women with early-stage breast cancer. Decreased functional well-being is likely due to the routine use of multi-modal treatment approaches for late-stage breast cancer which are associated with more adverse treatment effects.Citation30,Citation51 The negative impact of these therapies interacts with ongoing comorbidities and increases the burden on survivors to manage both health related impacts of their cancer treatment and ongoing chronic disease management.
Furthermore, in this study, women two to three years post-diagnosis reported better total HRQoL (FACT-G) than women three to four years post-diagnosis. This finding is inconsistent with reports of other research that find positive associations between years since diagnosis and total well-being in diverse breast cancer survivors.Citation12,Citation25 However, results may be due to higher distress and uncertainty among longer term (three to four years post diagnosis) breast cancer survivors compared to women at earlier phases in the survivorship trajectory (two to three years post diagnosis).Citation52,Citation53 Research shows that, healthcare providers give less cancer care information over time and thus longer-term breast cancer survivors become concerned about their experienced or perceived loss of oncology team in the follow up phase of care.Citation53 Furthermore, due to the fragmented healthcare delivery system, assuring coordinated, multidisciplinary care for cancer treatment among long-term cancer survivors can be challenging and might affect continuity of follow-up care.Citation54 Also, long-term breast cancer survivors experience less social support as their networks may not understand ongoing psychosocial and physical problems and become less inclined to provide needed support over time.Citation55
Limitations
There are some limitations to consider when interpreting results of this study. First, the cross-sectional study design prevented us from examining HRQoL over time. Second, conducting the study in New Jersey might limit the study’s generalizability to other populations. However, breast cancer survivors were recruited from a statewide, population-based registry and New Jersey is a densely populated state with diverse racial/ethnic, geographic and socio-economic groups.Citation56 Third, the response rate in the original survey was 30%, which could contain non-response bias; however, the response rate was similar to other studies that used population-based cancer registries. For instance, the American Cancer Society’s cancer survivorship studies that recruited survivors from 25 population-based cancer registries in 22 states had the median response rate of 34.9% (min. 15.9% and max. 52.1%).Citation57 Self-reported data among respondents also may contain response bias. Fourth, the small sample size of Hispanic women may have resulted in insufficient power to detect significant differences in HRQol by race/ethnicity. In addition, the survey was available in English only, thus excluded Spanish-speaking breast cancer survivors. However, in New Jersey, most Hispanics (79.7%) speak English at least ‘well’ and only 6.5% do not speak English at all.Citation21 Furthermore, coding some independent variables such as household income, education attainment and insurance at diagnosis as dichotomous variables may lead to loss of important differences in Hispanic and NHWs; and other unmeasured variables such as employment, current age, treatment type, social support, resiliency, spirituality, depression and patient-provider communication that might influence HRQoL were not available.
Despite these limitations, there are several strengths of this research. Predictors of HRQoL were explored and compared in a racially/ethnically diverse sample of breast cancer survivors in New Jersey. The well-being of breast cancer survivors has been largely studied in states such as California and New York and in large urban settings such as Washington, DC and Miami, FL.Citation15,Citation28 This study also used a multidimensional, validated measure to assess HRQoL and obtained cancer-related data from the NJSCR.
Implications for psychosocial oncology
The results of the study highlight the need for additional research into how health care professionals might help breast cancer survivors prevent, reduce and manage burdens associated with other health comorbidities. The management and care of cancer survivors with multiple comorbidities can be challenging and might result in fragmented care due to a lack of shared care and coordination between healthcare professionals.Citation54 Also, it is essential to coordinate care beyond immediate cancer needs and integrate other aspects of medical care, including preexisting comorbidities.Citation50 Some helpful approaches to coordination of care for cancer survivors with multiple comorbidities include increasing collaborations with primary care services, effectively using electronic health records to facilitate care coordination and utilization of community-based cancer care.Citation50,Citation58 Additionally, there is a need to develop more tailored behavioral and counseling interventions targeting young and low-income breast cancer survivors. Existing interventions to improve HRQoL among younger survivors include group psychotherapies to reduce anxiety and fear of recurrence,Citation59 couples-based psychosocial interventions,Citation60 web-based social network and peer-counseling interventions.Citation30 For low-income breast cancer survivors, navigation assistance programs in safety-net healthcare systems such as telephone-delivered reminders and written informational materials as well as collaborative care management programs to improve major depressive symptoms can be delivered to improve survivors’ well-being.Citation61 Lastly, psychosocial oncology interventions should be inclusive of racial/ethnic minorities, including Hispanic breast cancer survivors with limited English proficiency and provide linguistically accessible, culturally appropriate community-based and web-based social and peer support groups to improve HRQoL of racial/ethnic minorities.Citation12,Citation30
Compliance with ethical standards
All procedures performed in this study involving human participants were in accordance with the ethical standards of the Rutgers Biomedical and Health Sciences Institutional Review Board.
Ethical approval
This secondary research obtained de-identified data from Rutgers University. The study protocol was reviewed and approved by the Institutional Review Boards at Oregon State University and Rutgers University. The study adheres to the tenets of the Declaration of Helsinki.
Acknowledgements
We thank David Rotter and Grace Lu-Yao for their early contributions to this study.
Conflict of interest
The authors declare that they have no conflicts of interest.
Availability of data and material
The data sets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Code availability
The codes for analyses are available from the corresponding author on reasonable request. All statistical tests were conducted at a 2-sided significance level of 0.05 using R software (version 3.5.0).
Additional information
Funding
References
- American Cancer Society, Inc. Breast Cancer Facts & Figures 2019-2020. 2019. https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/breast-cancer-facts-and-figures/breast-cancer-facts-and-figures-2019-2020.pdf.
- Bellizzi KM, Rowland JH, Jeffery DD, McNeelT. Health behaviors of cancer survivors: examining opportunities for cancer control intervention. J Clin Oncol. 2005;23(34):8884–8893. doi:https://doi.org/10.1200/JCO.2005.02.2343
- Centers for Disease Control and Prevention. Health-Related Quality of Life (HRQOL). 2018. https://www.cdc.gov/hrqol/index.htm.
- LeMastersT, Madhavan S, Sambamoorthi U, Kurian S. A population-based study comparing HRQoL among breast, prostate, and colorectal cancer survivors to propensity score matched controls, by cancer type, and gender. Psycho-oncology. 2013;22(10):2270–2282.
- Yabroff KR, Lawrence WF, Clauser S, Davis WW, Brown ML. Burden of illness in cancer survivors: findings from a population-based national sample. J Natl Cancer Inst. 2004;96(17):1322–1330.
- DiSipioT, Hayes S, Newman B, Janda M. Health-related quality of life 18 months after breast cancer: comparison with the general population of Queensland, Australia. Support Care Cancer. 2008;16(10):1141–1150.
- Costanzo E, Lutgendorf S, Mattes M, et al. Adjusting to life after treatment: distress and quality of life following treatment for breast cancer. Br J Cancer. 2007;97(12):1625–1631. doi:https://doi.org/10.1038/sj.bjc.6604091
- Lethborg CE, Kissane D, Burns WI, Snyder R. Cast adrift: the experience of completing treatment among women with early stage breast cancer. J Psychosoc Oncol. 2000;18(4):73–90.
- Avis NE, Crawford S, Manuel J. Quality of life among younger women with breast cancer. JCO. 2005;23(15):3322–3330.
- Graves KD, Jensen RE, Canar J, et al. Through the lens of culture: quality of life among Latina breast cancer survivors. Breast Cancer Res Treat. 2012;136(2):603–613.
- Paek M-S, Ip EH, Levine B, Avis NE. Longitudinal reciprocal relationships between quality of life and coping strategies among women with breast cancer. Ann Behav Med. 2016;50(5):775–783. doi:https://doi.org/10.1007/s12160-016-9803-y
- Ashing-Giwa KT, Tejero JS, Kim J, Padilla GV, Hellemann G. Examining predictive models of HRQOL in a population-based, multiethnic sample of women with breast carcinoma. Qual Life Res. 2007;16(3):413–428.
- Short PF, Mallonee EL. Income disparities in the quality of life of cancer survivors. Med Care. 2006;44(1):16–23.
- Meyerowitz BE, Richardson J, Hudson S, Leedham B. Ethnicity and cancer outcomes: Behavioral and psychosocial considerations. Psychol Bull. 1998;123(1):47–70. doi:https://doi.org/10.1037/0033-2909.123.1.47
- Luckett T, Goldstein D, Butow PN, et al. Psychological morbidity and quality of life of ethnic minority patients with cancer: a systematic review and meta-analysis. Lancet Oncol. 2011;12(13):1240–1248.
- Härtl K, Engel J, Herschbach P, Reinecker H, Sommer H, Friese K. Personality traits and psychosocial stress: quality of life over 2 years following breast cancer diagnosis and psychological impact factors. Psychooncology. 2010;19(2):160–169. doi:https://doi.org/10.1002/pon.1536
- New Jersey Commission on Rationalizing Health Care Resources: New Jersey. Chapter 2 Final Report; 2008.
- U.S. Census Bureau. Quick Facts: New Jersey. 2019. https://www.census.gov/quickfacts/fact/table/NJ/RHI725219#RHI725219.
- U.S. Census Bureau. 2013-2017 American Community Survey 5-Year Estimates. 2017. https://archive.vn/20200213011237/ https://factfinder.census.gov/bkmk/table/1.0/en/ACS/17_5YR/B03001/0400000US34#selection-1127.7-1127.81.
- Wu S-Y. Growing New Jersey minority population reaches majority in some municipalities.2011. http://lwldolstatenjus/labor/lpa/pub/lmv/LMV_7.pdf.
- U.S. Census Bureau. Language spoken at home by ability to speak English. 2020. https://data.census.gov/cedsci/table?q=New%20Jersey%20Populations%20and%20People%20hispanic&tid=ACSDT1Y2018.B16006&hidePreview=false.
- Niu X, Pawlish KS, Roche LM. Cancer survival disparities by race/ethnicity and socioeconomic status in New Jersey. J Health Care Poor Underserv. 2010;21(1):144–160.
- Ashing-Giwa KT, Lim J-w. Examining the impact of socioeconomic status and socio-ecologic stress on physical and mental health quality of life among breast cancer survivors. Oncol Nurs Forum. 2009;36(1):79–88. doi:https://doi.org/10.1188/09.ONF.79-88
- Ashing‐Giwa KT, Padilla G, Tejero J, et al. Understanding the breast cancer experience of women: a qualitative study of African American, Asian American, Latina and Caucasian cancer survivors. Psychooncology. 2004;13(6):408–428. doi:https://doi.org/10.1002/pon.750
- Bowen DJ, Alfano CM, McGregorBA, et al. Possible socioeconomic and ethnic disparities in quality of life in a cohort of breast cancer survivors. Breast Cancer Res Treat. 2007;106(1):85–95.
- Giedzinska AS, Meyerowitz BE, Ganz PA, Rowland JH. Health-related quality of life in a multiethnic sample of breast cancer survivors. Ann Behav Med. 2004;28(1):39–51. doi:https://doi.org/10.1207/s15324796abm2801_6
- Spencer SM, Lehman JM, Wynings C, et al. Concerns about breast cancer and relations to psychosocial well-being in a multiethnic sample of early-stage patients. Health Psychol. 1999;18(2):159–168.
- Yanez B, Thompson EH, Stanton AL. Quality of life among Latina breast cancer patients: a systematic review of the literature. J Cancer Surviv. 2011;5(2):191–207. doi:https://doi.org/10.1007/s11764-011-0171-0
- Sammarco A, Konecny LM. Quality of life, social support, and uncertainty among Latina breast cancer survivors. Oncol Nurs Forum. 2008;35(5):844–849. doi:https://doi.org/10.1188/08.ONF.844-849.
- Janz NK, Mujahid MS, Hawley ST, et al. Racial/ethnic differences in quality of life after diagnosis of breast cancer. J Cancer Surviv. 2009;3(4):212–222.
- Levine EG, Yoo GJ, Aviv C. Predictors of quality of life among ethnically diverse breast cancer survivors. Appl Res Qual Life. 2017;12(1):1–16.
- Doose M, McGee-Avila J, Stroup AM, et al. Shared care during breast and colorectal cancer treatment: is it associated with patient-reported care quality? J Healthcare Qual. 2019;41(5):281–296.
- Ashing-Giwa KT. The contextual model of HRQoL: a paradigm for expanding the HRQoL framework. Qual Life Res. 2005;14(2):297–307. doi:https://doi.org/10.1007/s11136-004-0729-7
- Cella DF, Tulsky DS, Gray G, et al. The Functional Assessment of Cancer Therapy scale: development and validation of the general measure. J Clin Oncol. 1993;11(3):570–579. doi:https://doi.org/10.1200/JCO.1993.11.3.570
- FACIT.org. FACT-G scoring & interpretation materials. 2010. http://www.facit.org/FACITOrg/Questionnaires.
- Brady MJ, Cella DF, Mo F, et al. Reliability and validity of the functional assessment of cancer therapy-breast quality-of-life instrument. J Clin Oncol. 1997;15(3):974–986. doi:https://doi.org/10.1200/JCO.1997.15.3.974
- Centers for Disease Control and Prevention. About adult BMI. 2017. https://www.cdc.gov/healthyweight/assessing/bmi/adult_bmi/index.html.
- Findling MG, Bleich SN, Casey LS, et al. Discrimination in the United States: Experiences of Latinos. Health Serv Res. 2019;54(S2):1409–1418.
- Pakiz B, Ganz PA, Sedjo RL, et al. Correlates of quality of life in overweight or obese breast cancer survivors at enrollment into a weight loss trial. Psycho‐Oncology. 2016;25(2):142–149.
- Howard-Anderson J, Ganz PA, Bower JE, Stanton AL. Quality of life, fertility concerns, and behavioral health outcomes in younger breast cancer survivors: a systematic review. J Natl Cancer Inst. 2012;104(5):386–405. doi:https://doi.org/10.1093/jnci/djr541
- Ganz PA, Greendale GA, Petersen L, Kahn B, Bower JE. Breast cancer in younger women: reproductive and late health effects of treatment. J Clin Oncol. 2003;21(22):4184–4193. doi:https://doi.org/10.1200/JCO.2003.04.196
- Wenzel LB, Fairclough DL, Brady MJ, Cella D, et al. Age-related differences in the quality of life of breast carcinoma patients after treatment. Cancer. 1999;86(9):1768–1774.
- Kwan ML, John EM, Caan BJ, et al. Obesity and mortality after breast cancer by race/ethnicity: The California Breast Cancer Survivorship Consortium. Am J Epidemiol. 2014;179(1):95–111.
- Maly RC, Liu Y, Liang LJ, Ganz PA. Quality of life over 5 years after a breast cancer diagnosis among low‐income women: effects of race/ethnicity and patient‐physician communication. Cancer. 2015;121(6):916–926.
- Gordon LG, Merollini KM, Lowe A, Chan RJ. A systematic review of financial toxicity among cancer survivors: we can’t pay the co-pay. Patient. 2017;10(3):295–309.
- Fu M, Axelrod D, Guth A, et al. Comorbidities and quality of life among breast cancer survivors: a prospective study. J Pers Med. 2015;5(3):229–242. doi:https://doi.org/10.3390/jpm5030229
- Edgington A, Morgan MA. Looking beyond recurrence: comorbidities in cancer survivors. Clin J Oncol Nurs. 2011;15(1):E3–12.
- White A, Pollack LA, Smith JL, Thompson T, Underwood JM, Fairley T. Racial and ethnic differences in health status and health behavior among breast cancer survivors—Behavioral Risk Factor Surveillance System, 2009. J Cancer Surviv. 2013;7(1):93–103.
- O’Malley DM, Hudson SV, Ohman-Strickland PA, et al. Follow-up care education and information: identifying cancer survivors in need of more guidance. J Canc Educ. 2016;31(1):63–69.
- Sarfati D, Koczwara B, Jackson C. The impact of comorbidity on cancer and its treatment. CA Cancer J Clin. 2016;66(4):337–350. doi:https://doi.org/10.3322/caac.21342
- Lantz PM, Mujahid M, Schwartz K, et al. The influence of race, ethnicity, and individual socioeconomic factors on breast cancer stage at diagnosis. Am J Public Health. 2006;96(12):2173–2178.
- Hodgkinson K, Butow P, Hunt GE, Pendlebury S, Hobbs KM, Wain G. Breast cancersurvivors' supportive care needs 2-10 years after diagnosis. Support Care Cancer. 2007;15(5):515–523. doi:https://doi.org/10.1007/s00520-006-0170-2
- Gray RE, Fitch M, Greenberg M, Hampson A, Doherty M, Labrecque M. The information needs of well, longer-term survivors of breast cancer. Patient Educ Couns. 1998;33(3):245–255.
- Hewitt M, Greenfield S, Stovall E. From Cancer Patient to Cancer Survivor: lost in Transition. Washington, DC: National Academies Press; 2005.
- HolznerB, Kemmler G, Kopp M, et al. Quality of life in breast cancer patients-not enough attention for long-term survivors? Psychosomatics. 2001;42(2):117–123. doi:https://doi.org/10.1176/appi.psy.42.2.117
- State Cancer Profiles. Quick profiles: New Jersey. 2019. https://statecancerprofiles.cancer.gov/quick-profiles/index.php?statename=newjersey#t=0.
- Smith T, Stein KD, Mehta CC, et al. The rationale, design, and implementation of the American Cancer Society's studies of cancer survivors. Cancer. 2007;109(1):1–12. doi:https://doi.org/10.1002/cncr.22387
- Levit LA, Balogh E, Nass SJ, Ganz P. Delivering High-Quality Cancer Care: charting a New Course for a System in Crisis. Washington, DC: National Academies Press; 2013.
- Herschbach P, Berg P, Waadt S, et al. Group psychotherapy of dysfunctional fear of progression in patients with chronic arthritis or cancer. Behav Med. 2012;38(1):1–38.
- Baik OM, Adams KB. Improving the well-being of couples facing cancer: a review of couples-based psychosocial interventions. J Marital Fam Ther. 2011;37(2):250–266. doi:https://doi.org/10.1111/j.1752-0606.2010.00217.x
- Ell K, Vourlekis B, Xie B, et al. Cancer treatment adherence among low-income women with breast or gynecologic cancer: a randomized controlled trial of patient navigation. Cancer: Interdiscipl Int J Am Cancer Soc. 2009;115(19):4606–4615. doi:https://doi.org/10.1002/cncr.24500
