Abstract
Introduction
East-European data on cancer in patients undergoing hemodialysis (HD) are scarce. This study aimed to assess the pattern of cancer and related mortality in patients with end-stage kidney disease (ESKD) undergoing HD.
Methods
Retrospectively analyzing data from 7 HD centers, this study examined 1377 incident HD patients divided into three groups: no-cancers (NoC), cancers that occurred prior to HD initiation (CPI) and de novo cancer developed after HD initiation (DNC). Mortality risk and survival trends within groups were analyzed using Cox regression and Kaplan-Meier methods.
Results
In the cohort, 89.46% of the patients had no cancer (NoC group), 3.63% had cancer before (CPI group), and 6.89% had cancer after HD initiation (DNC group). The mean time from HD initiation to DNC diagnosis was 1 [2.75] years. Older age was associated with a higher risk of developing DNC (p < 0.001). Chronic tubulointerstitial nephritis (CTIN) is more prevalent in cancer patients. The most common cancer sites among DNC patients were the digestive (29.47%) and urinary tracts (18.95%), while those in CPI subjects were hematologic (22%) and digestive (20%). Cancer was an independent predictor of mortality risk (HR = 6.9, 95% [CI]:4.5–10.6, p < 0.001).
Conclusions
East-European ESKD patients undergoing HD have a high incidence of de novo cancers whose primary cancer sites are the digestive and urinary tracts. Almost half of the HD patients with CPI have hematologic and digestive tract cancers. Age and CTIN were associated with cancer risk. Cancer is an independent risk factor for all-cause mortality in patients undergoing hemodialysis (HD).
1. Introduction
Increased cancer incidence and related mortality have been reported in hemodialysis (HD) treated end-stage kidney disease (ESKD) patients [Citation1–4]. There is evidence showing that dialysis patients are two-to-three folds more likely to develop cancer as compared to the general population [Citation1–4].
There is a complex interaction between chronic kidney disease (CKD) and cancer through various pathways. Some types of cancers may induce direct kidney damage, leading to CKD. Many cancer therapies are nephrotoxic and can lead to chronic kidney damage. However, CKD may be a significant risk factor for the development of cancer [Citation1,Citation3,Citation5]. Immune system impairment by uremic toxins, DNA repair deficiencies, and accumulation of carcinogenic compounds due to reduced renal clearance, decreased antioxidant protection, chronic infections, and inflammation have been suggested as potential cancer-promoting factors [Citation3,Citation5,Citation6]. In addition, increasing levels of complement activation and inflammatory factors due to HD therapy, even with more biocompatible membranes, might induce cytokine release, which predisposes patients to the occurrence of cancer [Citation3,Citation5,Citation7].
Epidemiological data from ESKD registries have revealed an increased risk of cancer in dialysis patients in Asia [Citation8,Citation9], Australia/New Zealand [Citation1], the United States [Citation10], Northern [Citation11], Central [Citation12], and Southern European countries [Citation2], with marked area/region and site/type differences. On the other hand, fewer data have been reported from Eastern European countries, such as Romania, where the prevalence of ESKD treated with renal replacement therapy is 1048 per million of the general population [Citation13].
Although previous data reported an increased incidence of cancer in dialysis patients and in pre-dialytic CKD [Citation14], there is uncertainty about the differences between the pattern of cancers that occur after dialysis initiation and cancers related to pre-dialytic CKD. In addition, there are no data concerning the differences between the outcomes of dialysis patients in whom cancer developed after starting dialysis versus patients with cancer before dialysis initiation.
The aims of this study were twofold: to estimate the incidence and pattern of cancers in HD patients, both in whom cancers developed before the start of HD, and in whom cancers occurred after HD initiation, and to evaluate the mortality rate and risk factors of mortality in HD patients with cancer.
2. Materials and methods
2.1 Study population
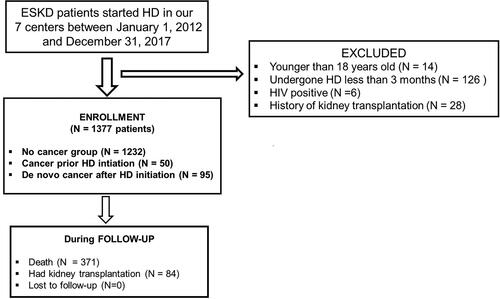
This multicenter retrospective observational study included 1377 ESKD patients treated in seven HD centers in all main geographical areas of Romania, from 1 January 2012 to 31 December 2017. The inclusion criteria were ESKD with at least 3 months of HD treatment and >18 years of age. Patients were excluded if they had a history of kidney transplantation, human immunodeficiency virus (HIV) infection, or had undergone HD therapy for less than 3 months. The end of the study was 31 December 2017, representing either the time of transplantation or death.
2.2 Clinical and laboratory data collection
At inclusion, patient data were retrieved from their medical records (personal data, medical history, etiology of CKD, comorbid conditions (coronary artery disease (CAD), stroke, and peripheral vascular disease (PVD)). Laboratory work-up at inclusion included hemoglobin, ferritin, high-sensitivity C-reactive protein (hsCRP), albumin, Ca, PO4, iPTH (intact parathyroid hormone (iPTH), 25-OH vitamin D, hepatitis B virus (HBV), and hepatitis C virus (HCV). The patients were treated with high flux, high surface filters, and ready-to-use dialysis fluid (B. Braun acidic bicarbonate hemodialysis concentrate). Renal anemia and CKD-mineral bone disorder were treated according to The Kidney Disease: Improving Global Outcomes Guidelines [Citation15,Citation16].
The date of cancer diagnosis, site of origin, and histology of the cancer were collected from the patients’ oncology records. All cancers were reported by cancer site, and cell types were in agreement with the International Classification of Disease for Oncology, third edition, first revision (ICD-O-3.1) [Citation17]. The study cohort was divided into three groups according to the presence of cancer: patients on HD therapy without cancer (NoC), patients with cancer diagnosis prior to HD initiation (CPI), and patients with “de novo” cancer diagnosis after dialysis initiation (DNC).
2.3 Statistical analysis
Data were collected and analyzed using the SPSS v.17 software suite (SPSS Inc. Chicago, IL, USA) and are presented as mean ± standard deviation for continuous variables with Gaussian distribution, median (interquartile range) for continuous variables without Gaussian distribution, or percentage for categorical variables. The lower and upper limits of the 95% confidence intervals (CI) used to estimate the prevalence was determined according to Wilson’s procedure for parameters with a Poisson distribution. Survival was evaluated using the Hazard Ratio (HR) method and presented using Kaplan–Meier diagrams. Depending on the distribution of the values, differences between groups were analyzed with the Student t-test (means, Gaussian populations) and Mann-Whitney U test (medians, non-Gaussian populations), for comparison of 2 groups, respectively the ANOVA-test (means, Gaussian populations) and Kruskal–Wallis test (medians non-Gaussian populations), for comparison of 3 groups. We used the Chi-square test or Fisher’s exact test (sample size <5) to assess the significance between proportions, and log-rank test to evaluate the differences between survival curves. To evaluate the involvement of additional factors in relation to death risk, Cox proportional hazards models were built. Continuous variable distributions were tested for normality using the Shapiro–Wilk test and for equality of variances using Levene’s test. The threshold for statistical significance was set at p < 0.05.
3. Results
3.1. Characteristics of patients
A total of 1377 HD patients were included in the study cohort (). The patients were then followed up for a median time of 3.75 (5.57) years after their enrollment, and no patient was lost to follow-up. The baseline characteristics of the cohort are presented in .
Table 1. Comparison between ESKD patients without cancer versus patients with cancer before HD initiation versus patients with de novo cancer after HD initiation.
Cancer diagnosis prior to HD initiation was present in 50 (3.63%) patients (CPI group), and “de novo” cancers diagnosis after HD initiation (DNC group) was detected in 95 (6.89%) patients. A total of 89.46% (1232 patients) of those who enrolled did not present or develop cancers (NoC group) during the follow-up period. The patients in the DNC group were the oldest (median age 67 (14) years), being significantly older than those in the NoC group, with a median age of 61 (17) years), (p < 0.001). The prevalence of cancer in our cohort was not gender-dependent ().
In the CPI group, the mean time from cancer diagnosis to HD initiation was 3.97 ± 4.96 years and in the DNC group, the median time from initiation of HD to de novo cancer diagnosis was 1 (2.75) years. The mean age at the time of cancer diagnosis was significantly lower for the CPI group as compared to the DNC group (58.63 ± 10.23 years vs. 68.32 ± 10.73 years, p < 0.001) ().
In cancer patients, the etiology of ESKD was dominated by chronic tubulointerstitial nephritis (CTIN), with a significantly higher prevalence in the CPI group (54%) than in the DNC (32.63%) and NoC (17.12%) groups (all P significant) (). The prevalence of DM as the etiology of ESKD was significantly lower in the cancer group than in the NoC group (p < 0.05) ().
Cardiovascular disease (CAD, stroke, and PVD) as a comorbid condition in ESKD, as expected, was very high (), but CAD was significantly more prevalent in both cancer groups than in the NoC group (p < 0.001). The prevalence of hepatitis B and/or C virus infection did not differ significantly among the three groups.
ESKD patients with cancer (both CPI and DNC groups) had significantly lower average hemoglobin, albumin, PO4, iPTH, 25-OH vitamin D levels, and had significantly higher levels of hs-CRP. These patients were treated with a lower dose of HD therapy (lower effective Kt/V (eKt/V)) and a higher dose of erythropoiesis-stimulating agents per week (ESA)/week (p < 0.001) than the NoC group ().
3.2. Cancer sites
In the CPI group, the most common cancer sites were hematological cancers (22%–11 patients), digestive tract (20%), female genital tract (18%), and cancers of the urinary tract (16%). In the DNC group, the most common cancer sites were the digestive tract (29.47%), urinary tract (18.95%), and hematological cancers (15.79%), followed by cancers of the respiratory tract (11.58%) (). Regarding the gender distribution of cancer sites, in the CPI group, the most frequent cancer sites in men were the urinary tract (30.76%), followed by the digestive tract (26.92%), and in women, the female genital tract (37.5%), followed by hematological cancers (29.16%). In the DNC group, the most frequent cancer sites were in the males, the digestive tract (28.71%), followed by the urinary tract (22.58%), while in the women were the female genital tract (24.24%) and hematological cancers (24.24%) ().
Table 2. Primary site of cancer in the CPI and DNC groups.
3.3. Cancer and associated-risk of mortality
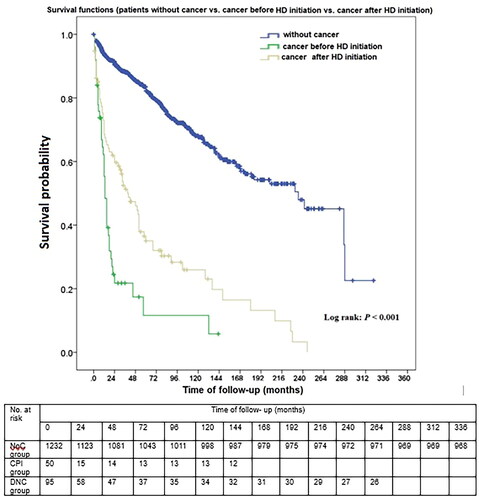
During a median follow-up of 3.75 (5.75) years, 371 (26.94%) patients died. All-cause mortality rate was significantly higher in patients with cancer. Thus, the DNC group had an increased death rate compared with the NoC group (72.63% vs. 21.42%; p < 0.001). The CPI group also had a higher probability of death than the NoC group (76% vs. 21.42%; p < 0.001). This significant difference in survival was also represented by the Kaplan–Meier survival curve (log-rank; p < 0.001) (). The mortality rate did not substantially differ between the two cancer groups, CPI and DNC, respectively (76% vs. 72.63%; p = 0.696), but the average survival time from cancer diagnosis to death was significantly lower in the DNC group than in the CPI (0.73 ± 1.06 years vs. 3.88 ± 3.81 years, p < 0.001) ().
Figure 2. Survival curves of HD patients with cancer after HD initiation, with cancer before HD initiation and without cancer.
In the regression analysis, the presence of cancer was associated with all-cause mortality in the univariate model (HR:3.68; 95% CI:2.90–4.68; p < 0.001) (). To assess the independent association between cancer and the risk of death in our cohort, we performed backward conditional multivariate regression analysis. Our models included age, HD dose (eKt/V), cancer presence, comorbidities (CAD, DM, and stroke), and laboratory variables. The stepwise algorithm in the Cox model accepted the following predictors: age, cancer, CAD, 25-OH vitamin D, hemoglobin, hsCRP, and eKt/V. The resulting model revealed a significant increase in the risk of death associated with age (HR = 1.03, p = 0.042), cancer (HR = 6.89, p < 0.001), CAD (HR =3.6, p = 0.007), and hsCRP (HR =1.006, p < 0.001). In contrast, higher levels of hemoglobin, 25-OH vitamin D, and eKt/V decreased the risk of mortality ().
Table 3. Cox proportional hazards regression analysis for risk factors of all-cause mortality.
Discussion
This is the first study examining the spectrum of cancer and its association with mortality in a large Eastern European cohort of 1377 ESKD patients undergoing HD, representing 12.4% of the patients from The Romanian Renal Registry [Citation18]. Cancer was diagnosed before HD initiation in 3.63% of the patients, and 6.89% of the patients developed cancer after HD initiation. Patients with “de novo” cancer after HD initiation were significantly older than those without cancer. Chronic tubulointerstitial nephritis was the main cause of ESKD in both CPI and DNC groups. In the CPI group, the most common cancer sites were hematological cancer, followed by the digestive tract, while in the DNC group, the most common cancer sites were the digestive tract and urinary tract. In patients with cancer, the all-cause mortality rate was more than three-fold higher than that in the NoC group, with cancer being an independent predictor of death. Furthermore, the DNC group had a significantly shorter overall survival time, measured from the time of cancer diagnosis to death, than the CPI group.
It is well known that there are various features of ESKD patients which contrast and are worse among Eastern European than Western Europe countries because of different geographic, cultural, educational, socio-economic and infrastructure characteristics [Citation19, Citation20]. Though there are some reports about the interrelation between ESKD and cancer among Western European hemodialysed patients [Citation2, Citation12, Citation21], data from East-European countries are lacking. To the best of our knowledge, this is the first study which investigated cancer prevalence, a pattern of cancer site distribution and cancer-associated mortality risk among a large Eastern-European hemodialysed cohort of patients.
In our large-sample and multicenter study, the prevalence of cancer was significantly higher as compared to the general population from Romania, both for cancers before HD initiation (3.63% vs.1.35% p < 0.001) and for cancer after HD initiation (6.89% vs. 1.35%, p < 0.001) [Citation22].
In ESKD patients undergoing HD, an increased risk of de novo cancers was reported in the late 1990s [Citation21]. The incidence and prevalence rates of de novo cancers in ESKD patients vary widely from 2.3% [Citation23] to 13.9% [Citation2], depending on the geographic area, follow-up time, dialysis treatment duration, and gender of the included patients [Citation2, Citation8, Citation10, Citation21, Citation24]. In a Korean cohort of HD patients, during a follow-up period of approximately 13 years, the incidence of “de novo” cancers in stable HD patients was 7.82% (the average time from initiation to cancer diagnosis was 5 ± 3 years). The most frequent cancer sites in men are colorectal cancer, followed by liver, stomach, lung, and kidney cancers, and in women, colorectal cancers, followed by breast, thyroid gland, and stomach cancers [Citation9]. In Taiwan, during the 9 years of follow-up, 5.8% of the patients developed “de novo” cancers, and the most frequent cancer sites in men were the liver, followed by the urinary bladder and kidney; in women, bladder, kidney, and breast cancers were more prevalent [Citation24]. In the United States, the cumulative incidence of all cancer types for 5 years was 9.48%, and the most frequent cancer sites were the kidney and renal pelvis with an increasing incidence, and colorectal cancers, lung, and pancreas showed a decreasing trend [Citation10]. In a North-Eastern Italian study, HD patients presented a 1.3-fold higher risk than the general population, with an elevated risk for kidney, skin, and oral cavity cancers [Citation2]. In our Eastern European HD patients, the cumulative incidence of de novo cancer was 6.89%, and the median time from HD initiation to cancer diagnosis was 1 (2.75) years. A previous study reported that the average time from HD initiation to cancer diagnosis varied between 2.5 and 3.4 years [Citation8, Citation21, Citation24]. Concerning the sites of cancer distribution according to gender, in our DNC cohort, the most frequent sites of cancer in men were the digestive and urinary tract, and in the female genital tract and hematological cancers.
The increased risk of cancer in patients with predialytic CKD is still debated. Some population-based studies suggest that predialytic CKD patients are at a higher risk of developing cancer than the general population [Citation25, Citation26]. It seems that in the elderly, for every 10 mL/min/1.73 m2 decline in eGFR, the risk of cancer increases by 29% [Citation25]. However, the Atherosclerosis Risk in Communities (ARIC) cohort in the USA [Citation27] and the 2016 meta-analysis by Wong et al. failed to demonstrate an increased risk of cancer in predialytic CKD patients [Citation28].
Nevertheless, 3.6% of our ESKD patients were diagnosed with cancer before HD initiation. The most frequent cancer sites in these patients were hematological cancers and the digestive tract. The hematological cancers are associated with a higher risk of ESKD [Citation29, Citation30]. Multiple myeloma or lymphoma may directly cause kidney failure, and the renal involvement reported in these patients varies between 7% and 34% [Citation29]. Digestive tract cancers are more frequently reported in patients with CKD, particularly in subjects younger than 40 years [Citation31]. A complex relationship exists between cancer and CKD. Hematological cancers may directly cause kidney injury, while anticancer treatments (chemotherapy and surgery) may also alter kidney function, leading to ESKD [Citation5, Citation14]. In our study, patients with CPI (even those treated with HD) had a significantly longer survival time from cancer diagnosis to death than DNC subjects. This could be related to the fact that ESKD patients with de novo cancers have limited oncological interventions (i.e., limited chemotherapy options, altered pharmacokinetics and pharmacodynamics of anticancer agents, higher toxicity risk of drugs, and sometimes contraindications for a surgical approach). [Citation5, Citation32, Citation33]. These factors may influence the need for screening strategies [Citation3].
Age has been reported to be a risk factor for cancer in the general population and among patients with HD-treated ESKD. In our study, DNC patients were significantly older than non-cancer patients. A higher risk of cancer in older patients has also been reported in other studies, including ESKD patients [Citation9, Citation10, Citation24]. These findings have been attributed to excess inflammation induced by ESKD and aging [Citation7, Citation34]. In contrast, some studies reported a high risk for de novo cancers in younger dialysis patients [Citation2, Citation4].
Although DM is considered a risk factor for cancer in the general population, our study failed to support this statement in patients having DM as the primary cause of ESKD. In our cohort, patients with DM had a lower risk for de novo cancers; similar results were reported in another HD cohort [Citation9, Citation10, Citation24]. The phenomenon of reverse epidemiology could explain this finding because, in ESKD patients, several conventional risk factors could have different predictive patterns owing to survival bias and time variances between competing risk factors [Citation35]. Another reason could be that DM patients have a relatively shorter life expectancy and less time to cancer occurrence [Citation10, Citation24].
In our cohort, CTIN as a cause of ESKD was significantly more frequent in patients with cancer. Some toxins involved in CTIN etiology, (aristolochic acid and analgesics), are associated with an increased risk of urothelial cancers [Citation16], and several studies have emphasized the strong association of analgesic nephropathy [Citation36], aristolochic acid nephropathy (AAN) [Citation37], and Balkan-Endemic nephropathy (BEN) [Citation38] with the occurrence of urinary tract cancer. The etiology of CTIN was not registered in our patient’s data; therefore, we could not further elaborate on this issue.
In our study, the most common sites of cancer in patients with DNC were the digestive and urinary tract. A high risk of digestive tract cancer could probably be related to the higher prevalence of hepatitis C viral infection in individuals with ESKD, which is involved in the development and progression of liver cancer, but the specific mechanism for this increased risk is still debated [Citation39]. Furthermore, the high risk for kidney and urinary tract cancers is possibly related to the etiology of CKD chronic kidney disease (urological anomalies or the development of acquired cystic kidney disease) [Citation2, Citation4, Citation5, Citation40].
In the last two decades, it has become obvious that cancers alter the prognosis of CKD patients both before and after HD initiation [Citation1, Citation23, Citation41]. Cancer mortality is 2-to 3-fold higher in ESKD patients treated with HD than in the general population [Citation1, Citation23, Citation41]. In our cohort, cancer was an independent risk factor for all-cause mortality, both in CPI group and in DNC patients. Mortality rates were significantly higher in all cancer patients and did not differ between the two groups of patients (before and after starting HD); however, the length of survival from the time of cancer diagnosis was significantly shorter in the DNC group than in the CPI group.
All of the above data and discussions raise an important issue for cancer screening in HD patients. The cumulative incidence, prevalence of cancer risk factors, and cancer sites seem to differ depending on geographic areas in ESKD patients on HD maintenance. Screening tests and methods also have limited specificity and sensitivity in patients with ESKD treated with HD [Citation42]. However, HD patients have a short life expectancy; hence, survival benefits after diagnosis and treatment of cancer are small. Aggressive therapeutic interventions may reduce their survival [Citation43]. Currently, there are no guidelines for cancer screening and therapy in patients with ESKD treated with HD, although an individualized or HD center-level approach should be encouraged. Therefore, the geographic (HD center) epidemiology of cancer in HD patients is important. Nevertheless, screening for cancer before a kidney transplant and in patients undergoing immunosuppressive therapy is mandatory.
Our study had several limitations. First, we were not able to obtain data on cancer treatment, status, or stage at the time of inclusion in the study. Second, our cohort included ESKD patients from different regions of Romania, and we could not compare the cancer incidence in HD patients with the data of the general population because summary statistics for the general population were not available for these regions. Third, there were no data on cancer-related risk factors. Finally, we assessed the risk of mortality by performing a multivariate Cox regression analysis; however, given the drawbacks of observational studies, we cannot rule out the possibility of residual confounding factors.
Our study had some strengths. First, this is the first Eastern European study based on a nationally representative ESKD cohort with good generalizability. Second, rigorous control of data accuracy was performed to ensure the consistency of the results. Finally, multiple potential confounders were included in this analysis.
Conclusions
In our Eastern-European cohort of ESKD patients treated with HD, the incidence of de novo cancers was high, and the main cancer sites were the digestive tract and the urinary tract. Older age and CTIN as a cause of ESKD were associated with cancer risk in these patients. In patients with cancer before the start of HD, the most frequent cancer types are hematological and digestive cancers. Cancer is a strong and independent risk factor for all-cause mortality in patients treated with HD. Furthermore, a significantly lower survival rate is a characteristic of de novo cancers in patients undergoing HD. The results of our study highlight the imperative need for the development of a local/areal screening system for cancer in patients with ESKD undergoing HD.
Author contributions
FG, MC, AA and AS designed the study and wrote the manuscript. OS, VI, LP, FB, OM, ASS, MG, FP, and RT: collected and entered data; BT - performed the statistical analysis; FG, MC, BT, and AS contributed to data acquisition and interpretation; FG, MC, LP, FP, RT, and AS critically reviewed and revised the article. All the authors have read and approved the final manuscript.
Ethical statement
The study was approved by the BBraun Avitum Ltd. Romania Ethical Committee (Board of Human Studies), and all patients provided written informed consent before enrollment. This study was conducted in accordance with the requirements of the Declaration of Helsinki.
Acknowledgments
The authors are grateful to Marius Parv for his assistance in editing and translating this manuscript.
Disclosure statement
No potential conflict of interest was reported by the author(s).
Data availability statement
The original contributions of this study are included in the article/supplementary material. Further inquiries can be directed to the corresponding authors.
Correction Statement
This article has been corrected with minor changes. These changes do not impact the academic content of the article
Additional information
Funding
References
- Rosales BM, De La Mata N, Vajdic CM, et al. Cancer mortality in people receiving dialysis for kidney failure: an Australian and New Zealand cohort study, 1980–2013. Am J Kidney Dis. 2022;80(4):1–9. doi: 10.1053/j.ajkd.2022.03.010.
- Taborelli M, Toffolutti F, Del Zotto S, et al. Increased cancer risk in patients undergoing dialysis: a population-based cohort study in North-Eastern Italy. BMC Nephrol. 2019;20(1):107. doi: 10.1186/s12882-019-1283-4.
- Cheung CY, Tang SCW. Oncology in nephrology comes of age: a focus on chronic dialysis patients. Nephrology. 2019;24(4):380–386. doi: 10.1111/nep.13525.
- Xie X, Li F, Xie L, et al. Meta-analysis of cancer risk among end stage renal disease undergoing maintenance dialysis. Open Life Sci. 2023; 18(1):20220553. doi: 10.1515/biol-2022-0553.
- Hu M, Wang Q, Liu B, et al. Chronic kidney disease and cancer: inter-Relationships and mechanisms. Front Cell Dev Biol. 2022;10:868715. doi: 10.3389/fcell.2022.868715.
- Stewart JH, Vajdic CM, van Leeuwen MT, et al. The pattern of excess cancer in dialysis and transplantation. Nephrol Dial Transplant. 2009;24(10):3225–3231. doi: 10.1093/ndt/gfp331.
- Cobo G, Lindholm B, Stenvinkel P. Chronic inflammation in end-stage renal disease and dialysis. Nephrol Dial Transplant. 2018;33(suppl_3):iii35–iii40. doi: 10.1093/ndt/gfy175.
- Lee MJ, Lee E, Park B, et al. Epidemiological characteristics of cancers in patients with end-stage kidney disease: a Korean nationwide study. Sci Rep. 2021;11(1):3929. doi: 10.1038/s41598-021-83164-6.
- Myung J, Choi JH, Yi JH, et al. Cancer incidence according to the national health information database in Korean patients with end-stage renal disease receiving hemodialysis. Korean J Intern Med. 2020;35(5):1210–1219. doi: 10.3904/kjim.2018.400.
- Butler AM, Olshan AF, Kshirsagar AV, et al. Cancer incidence among US medicare ESRD patients receiving hemodialysis, 1996-2009. Am J Kidney Dis. 2015;65(5):763–772. doi: 10.1053/j.ajkd.2014.12.013.
- Hortlund M, Mühr LSA, Storm H, et al. Cancer risks after solid organ transplantation and after long-term dialysis. Int J Cancer. 2017;140(5):1091–1101. doi: 10.1002/ijc.30531.
- Béchade C, Dejardin O, Bara S, et al. Survival of patients with cancer starting chronic dialysis: data from kidney and cancer registries in lower normandy. Nephrology. 2018;23(12):1125–1130. doi: 10.1111/nep.13091.
- Stel VS, de Jong RW, Kramer A, et al. Supplemented ERA-EDTA registry data evaluated the frequency of dialysis, kidney transplantation, and comprehensive conservative management for patients with kidney failure in Europe. Kidney Int. 2021;100(1):182–195. doi: 10.1016/j.kint.2020.12.010.
- Stengel B. Chronic kidney disease and cancer: a troubling connection. J Nephrol. 2010;23:253–262.
- Kidney Disease: improving Global Outcomes (KDIGO) Anemia Work Group. KDIGO clinical practice guideline for anemia in chronic kidney disease. Kidney Int Suppl. 2012;2:279–335.
- Kidney Disease: improving Global Outcomes (KDIGO) CKD-MBD Update Work Group. KDIGO 2017 clinical practice guideline update for the diagnosis, evaluation, prevention, and treatment of chronic kidney disease-mineral and bone disorder (CKD-MBD). Kidney Int Suppl. 2017;7(1):1–59.
- World Health Organization. International classification of diseases for oncology (ICD-O)., 3rd edition, 1st revision. Geneva: World Health Organization, 2013.
- Available from: https://www.registrulrenal.ro/resurse_statistica/Raport_RRR_2020_covid.pdf.
- Sever MŞ, Jager KJ, Vanholder R, et al. A roadmap for optimizing chronic kidney disease patient care and patient-oriented research in the Eastern European nephrology community. Clin Kidney J. 2020;14(1):23–35. doi: 10.1093/ckj/sfaa218.
- Spasovski G, Rroji M, Vazelov E, et al. Nephrology in the Eastern and Central European region: challenges and opportunities. Kidney Int. 2019;96(2):287–290. doi: 10.1016/j.kint.2019.02.040.
- Maisonneuve P, Agodoa L, Gellert R, et al. Cancer in patients on dialysis for end-stage renal disease: an international collaborative study. Lancet. 1999;354(9173):93–99. doi: 10.1016/s0140-6736(99)06154-1.
- Available from: https://gco.iarc.fr/today/data/factsheets/populations/642-romania-fact-sheets.pdf.
- Lee JE, Han SH, Cho BC, et al. Cancer in patients on chronic dialysis in Korea. J Korean Med Sci. 2009;24 (Suppl 1):S95–S101. doi: 10.3346/jkms.2009.24.S1.S95.
- Chien CC, Han MM, Chiu YH, et al. Epidemiology of cancer in end-stage renal disease dialysis patients: a national cohort study in Taiwan. J Cancer. 2017;8(1):9–18. doi: 10.7150/jca.16550.
- Wong G, Hayen A, Chapman JR, et al. Association of CKD and cancer risk in older people. J Am Soc Nephrol. 2009;20(6):1341–1350. doi: 10.1681/ASN.2008090998.
- Iff S, Craig JC, Turner R, et al. Reduced estimated GFR and cancer mortality. Am J Kidney Dis. 2014;63(1):23–30. doi: 10.1053/j.ajkd.2013.07.008.
- Mok Y, Ballew SH, Sang Y, et al. Albuminuria, kidney function, and cancer risk in the community. Am J Epidemiol. 2020;189(9):942–950. doi: 10.1093/aje/kwaa043.
- Wong G, Staplin N, Emberson J, et al. Chronic kidney disease and the risk of cancer: an individual patient data meta-analysis of 32,057 participants from six prospective studies. BMC Cancer. 2016;16:488. doi: 10.1186/s12885-016-2532-6.
- Morel A, Meuleman MS, Moktefi A, et al. Renal diseases associated with hematologic malignancies and thymoma in the absence of renal monoclonal immunoglobulin deposits. Diagnostics. 2021;11(4):710. doi: 10.3390/diagnostics11040710.
- Varga A, Tilea I, Petra DN, et al. Cardiovascular events throughout the disease course in chronic myeloid leukaemia patients treated with tyrosine kinase Inhibitors – a single-centre retrospective study. JCM. 2020;9(10):3269. doi: 10.3390/jcm9103269.
- Oh HJ, Lee HA, Moon CM, et al. Incidence risk of various types of digestive cancers in patients with pre-dialytic chronic kidney disease: a nationwide population-based cohort study. PLoS One. 2018;13(11):e0207756. doi: 10.1371/journal.pone.0207756.
- Capasso A, Benigni A, Capitanio U, et al. Summary of the international conference on Onco-Nephrology: an emerging field in medicine. Kidney Int. 2019;96(3):555–567. doi: 10.1016/j.kint.2019.04.043.
- Małyszko J, Kozlowski L, Kozłowska K, et al. Cancer and the kidney: dangerous liaisons or price paid for the progress in medicine? Oncotarget. 2017;8(39):66601–66619. doi: 10.18632/oncotarget.18094.
- Zinger A, Cho WC, Ben-Yehuda A. Cancer and aging – the inflammatory connection. Aging Dis. 2017;8(5):611–627. doi: 10.14336/AD.2016.1230.
- Kalantar-Zadeh K, Block G, Humphreys MH, et al. Reverse epidemiology of cardiovascular risk factors in maintenance dialysis patients. Kidney Int. 2003;63(3):793–808. doi: 10.1046/j.1523-1755.2003.00803.x.
- Lornoy W, Becaus S, de Vleeschouwer M, et al. Renal cell carcinoma, a new complication of analgesic nephropathy. Lancet. 1986;1(8492):1271–1272. doi: 10.1016/s0140-6736(86)91407-8.
- Nortier JL, Martinez MC, Schmeiser HH, et al. Urothelial carcinoma associated with the use of a Chinese herb (Aristolochia fangchi). N Engl J Med. 2000;342(23):1686–1692. doi: 10.1056/NEJM200006083422301.
- Stefanovic V, Polenakovic M, Toncheva D. Urothelial carcinoma associated with Balkan endemic nephropathy. A worldwide disease. Pathol Biol. 2011;59(5):286–291. doi: 10.1016/j.patbio.2009.05.002.
- Lin MY, Kuo MC, Hung CC, et al. Association of dialysis with the risks of cancers. PLoS One. 2015;10(4):e0122856. doi: 10.1371/journal.pone.0122856.
- Gomotirceanu AM, Sabău A, Gomotîrceanu F, et al. CEUS utility in the diagnosis of a stage I renal cancer developed on polycystic kidney. A case report. Med Ultrason. 2023:1–3. Online ahead of print. doi: 10.11152/mu-3889.
- Vogelzang JL, van Stralen KJ, Noordzij M, et al. Mortality from infections and malignancies in patients treated with renal replacement therapy: data from the ERA-EDTA registry. Nephrol Dial Transplant. 2015;30(6):1028–1037. doi: 10.1093/ndt/gfv007.
- Coppolino G, Bolignano D, Rivoli L, et al. Tumour markers and kidney function: a systematic review. Biomed Res Int. 2014;2014:647541. doi: 10.1155/2014/647541.
- Rosner MH. Cancer screening in patients undergoing maintenance dialysis: who, what, and when. Am J Kidney Dis. 2020;76(4):558–566. doi: 10.1053/j.ajkd.2019.12.018.