?Mathematical formulae have been encoded as MathML and are displayed in this HTML version using MathJax in order to improve their display. Uncheck the box to turn MathJax off. This feature requires Javascript. Click on a formula to zoom.
?Mathematical formulae have been encoded as MathML and are displayed in this HTML version using MathJax in order to improve their display. Uncheck the box to turn MathJax off. This feature requires Javascript. Click on a formula to zoom.ABSTRACT
In this study, a molecularly imprinted polymer (MIP) was synthesized using trichlorfon as a template molecule. The MIP exhibited a high adsorption capacity toward trichlorfon. Using bovine serum albumin as a carrier, trichlorfon hapten was indirectly labeled with luminol, which competed with free trichlorfon for the combination with a biomimetic antibody of MIP. Based on this direct competitive format, a high throughput flow-injection chemiluminescent biomimetic immunoassay method was developed for fast detection of trace trichlorfon. The influencing factors were investigated and optimized. Under the optimal condition, a lower limit of detection (IC15) of 0.0024 mg/L was obtained using this method. This method was also used to detect the trichlorfon spiked in carrot and cabbage samples, and the results were also verified using the method of gas chromatography.
Introduction
Organophosphorus pesticides’ residues in agricultural products have attracted widespread attention because of their toxicity and potential carcinogenicity (Kwong, Citation2002; Sultatos, Citation1994; Sarabia, Maurer, & Bustos-Obregón, Citation2009). Trichlorfon, an important organophosphorus pesticide, can overstimulate the nervous system and cause serious conditions including nausea, dizziness, confusion, and respiratory paralysis and death at very high exposure levels (Sun et al., Citation2008). Thus, it is important to develop a simple and accurate method to detect trichlorfon residue for food safety and public health.
There are many methods for detecting organophosphorus pesticides, such as gas chromatography (GC) (Xin, Qiao, Xu, & Zhou, Citation2013; Liu, Yang, & Kang, Citation2008), high-performance liquid chromatography (He, Luo, Jiang, & Qu, Citation2010; Li, Zhang, Kong, Qiao, & Xu, Citation2017), gas or liquid chromatography coupled with tandem mass spectrometry (Li, Hu, Huo, & Xu, Citation2006; Chung & Chan, Citation2010; Wang, Zhong, Mao, & Wan, Citation2015), and capillary electrophoresis (Chen & Fung, Citation2010). Most of these described methods offer low detection limits and good sensitivities but need expensive instruments and long analysis times. Chemiluminescence (CL) is a simple analysis method that can be performed quickly and easily (Zhang et al., Citation2015; Biparva, Abedirad, & Kazemi, Citation2015). Unfortunately, the sensitivity of the CL method is relatively low, which causes its direct application to the analysis of food to be limited.
Immunoassays have become a popular tool for detecting pesticide residues based on the specificity of an antigen–antibody reaction, which has many advantages such as high sensitivity and specificity (Song et al., Citation2011; Liu et al., Citation2012). In recent years, CL immunoassays, which combine the short analysis time of chemiluminescent systems with the high sensitivity of immunoassays, have become an attractive analysis technique for pesticides’ residues (Schlaeppi, Kessler, & Foery, Citation1994; Zhang, Citation2015). However, the instability of biological antibodies at a high pH and temperature affects the performance of traditional immunoassays. Furthermore, the acquisition of biological antibodies is complicated (Skerritt, Guihot, Asha, Bea, & Ngk, Citation2003; Wang et al., Citation2010; Yang et al., Citation2015). To overcome the above drawbacks of traditional immunoassay methods, molecularly imprinted polymers (MIPs) have been used as biomimetic antibodies in immunoassays owing to their good stability, easy preparation, and specific adsorption to target molecules (Xu, Gao, Zhang, Chen, & Qiao, Citation2011; Wang et al., Citation2009; Li, Lu, Qiao, & Xu, Citation2017).
Luminol is usually used as a luminescent label. However, after it is directly tagged with an antigen or antibody, the luminous intensity is reduced. To improve the luminous efficiency, studies have been devoted to the use of bovine serum albumin (BSA) as a carrier protein to conjugate more luminol (Qi & Zhang, Citation2004). In this study, trichlorfon hapten was indirectly labeled with luminol by using BSA as a carrier, which then competed with free trichlorfon for combination with the MIP. Based on this process, a novel flow-injection chemiluminescent biomimetic immunoassay (FICL-BI) method was developed. The influencing factors of this method were investigated in detail, and the accuracy and applicability of this developed method were evaluated.
Materials and methods
Chemicals and materials
Ethyleneglycol dimethacrylate (EGDMA) was supplied by Sigma-Aldrich Co., Ltd. (St Louis, MO, USA). Dimethylformamide (DMF) and N-hydroxysuccinimide (NHS) were supplied by Sigma-Aldrich (USA). 1-Ethyl-3-(3-dimethyllaminopropyl) carbodiimide hydrochloride (EDC-HCl) was purchased from Shanghai Macklin Biochemical Co., Ltd. (Shanghai, China). The BSA was supplied by Beijing Solarbio Science and Technology Co., Ltd. Methanol, acetic acid, ethyl acetate, sodium hydrogen phosphate (Na2HPO4), sodium dihydrogen phosphate (NaH2PO4), and hydrogen peroxide (H2O2) were obtained from Kaitong Chemical Reagent Co., Ltd. (Tianjin, China). Trichlorfon, monocrotophos, and acephate (99%) were purchased from the Institute for the Control of Agrochemicals, Ministry of Agriculture (Beijing, China). Methacrylic acid (MAA) and 2,2-azobisisobutyronitrile were obtained from Tianjin Chemical Reagent Factory (Tianjin, China), and MAA was vacuum distilled and AIBN was recrystallized before use. Luminol was supplied by Meryer Chemical Technology Co., Ltd. (Shanghai, China). All the other reagents were of analytical grade. Double-distilled water (DDW) obtained from an Aike ultrapure water instrument (Tangshi Kangning Technology, Chengdu, China) was used in all the experiments.
Carrot and cabbage samples were purchased randomly from a supermarket in Tai’an in January 2017.
Apparatus
To measure the adsorption capacity of the MIP, a UV-5500 ultraviolet–visible (UV–vis) spectrophotometer (Shanghai Yuanxi Analysis Instrument Co., Ltd., Shanghai, China) was used. The CL measurements were performed using an IFFM-E flow-injection CL analyzer (Xi’an Remex Analysis Instrument Co., Ltd., Xian, China). A polytetrafluoroethylene tube (0.8 mm i.d. × 5 m) was used to connect all components in the flow-injection system.
Preparation of the MIP
The MIP was prepared by bulk polymerization. Briefly, 0.257 g (l.0 mmol) of trichlorfon and 0.172 g (2.0 mmol) of MAA were dissolved in 3.0 mL of chloroform. After stirring at room temperature for 30 min, 0.793 g (4.0 mmol) of EGDMA and 20 mg of 2,2-azobisisobutyronitrile were added. After the mixture was ultrasonicated for 15 min, the solution was purged with nitrogen, hermetically sealed, and placed in a water bath at 60°C for 24 h. When the polymerization was finished, the synthetic polymers were crushed and sieved through a 200-mesh sieve. The polymer particles were soxhlet extracted with 120 mL of methanol and 30 mL of glacial acetic acid for 24 h, and then washed with methanol for 12 h. Finally, the polymer was dried in a vacuum oven at 60°C for 12 h.
For comparison, a nonimprinted polymer (NIP) was prepared in the same way but without using the template molecule.
Characterization of MIP
To evaluate the adsorption ability of the MIP, an equilibrium adsorption experiment was carried out. Briefly, 20 mg of MIP and 10 mL of a trichlorfon standard solution (100–500 mg/L) were added into a 50 mL volumetric flask. The mixture was shaken for 4 h at room temperature and then centrifuged at 4320×g for 20 min. Afterwards, the supernatant was analyzed by UV spectrometry at 195 nm, and the adsorption capacity (Q) was calculated according to the following equation:
In this equation, C0 and C1 represent the concentrations of trichlorfon in solution before and after absorption, respectively, V is the volume of trichlorfon solution, and M is the mass of MIP.
Synthesis of trichlorfon hapten
The trichlorfon hapten was synthesized as follows: 5.664 g of trichlorfon (22 mmol) and 2.002 g of succinic anhydride (20 mmol) were placed in a small beaker, and then 5 mL of anhydrous pyridine was added. After stirring for 10 min, the mixture was placed under the dark condition for 18 h. Afterwards, the reaction mixture was purged with nitrogen to remove pyridine. Then, 50 mL of DDW was added and the pH of the mixture was adjusted to 8–9 with a saturated Na2CO3 solution. After extraction with ethyl acetate (3 × 30 mL), the organic phase was evaporated to dryness under reduced pressure.
Synthesis of trichlorfon hapten–BSA–luminol CL marker
The trichlorfon hapten–BSA conjugate was synthesized based on a previous method (Manclus, Primo, & Montoya, Citation1996; Qian et al., Citation2009). 7 mg of NHS was added to 25 mg of synthetic trichlorfon hapten dissolved in 1 mL of DMF. After stirring for 1 h, 11.7 mg of EDC-HCl was added. The solution was stirred for 2 h, and then 12 mL of borax buffer containing 60 mg of BSA was added dropwise. The mixture was reacted under the dark condition for another 6 h with stirring. When the reaction was completed, the mixture was dialyzed with the PBS (0.01 M, pH 7.4), and the dialysate was replaced every 12 h until the hapten could not be detected.
The trichlorfon–BSA conjugate was tagged with luminol according to the reported method (Qi & Zhang, Citation2004). Briefly, 20 mg of luminol was added in 2 mL of 2.4 mol/L cold HCl, and the mixture was stirred in an ice bath (0–4°C) until it was dissolved. Then, 20 mg of NaNO2 was added and the mixed solution was placed under dark condition for 15 min. After the pH value of the reaction mixture was adjusted to 8.5 with cold borax buffer (pH 9.0), 12 mL of trichlorfon–BSA conjugate was added, and the mixture was then reacted for 24 h at 0–4°C. Finally, the compound was dialyzed with PBS (0.01 M, pH 7.4), and the dialysate was replaced every 12 h until luminol could not be detected using a UV–vis spectrophotometer.
The FICL-BI procedure
The FICL-BI procedure was carried out as follows: 2 mL of trichlorfon–BSA–luminol CL marker (diluted 5 times with PBS) and 2 mL of trichlorfon standard solution were mixed, and then 2 mg of the MIP (biomimetic antibody) was added. The mixture was shaken for 75 min at room temperature and then centrifuged at 5000 rpm for 20 min. Finally, the supernatant was injected into the flow-injection CL analyzer for analysis according to the FICL process.
The FICL process was performed using an IFFM-E flow-injection and CL analyzer. Na2CO3-NaHCO3 buffer solution was delivered by a primary pump at 60 r/min through the channel and mixed with the CL marker (or supernatant of the competition reaction) and H2O2 solutions that were delivered by the secondary pump at 45 r/min through a six-way injection valve. The running times for the primary and secondary pumps were 15 and 8 s, respectively. The CL signal produced in the flow cell was monitored with a photomultiplier and recorded by a computer equipped with a data acquisition interface. Finally, the inhibition rate was calculated.
Selectivity test
Cross-reactivity studies can be used to evaluate the selectivity properties of the proposed method. Trichlorfon and its structural analogues (monocrotophos and acephate) were detected using the FICL-BI procedure under the optimal conditions, and their standard curves were established.
In addition, to further study the specific adsorption of MIP, the standard curves of the FICL-BI method at trichlorfon concentrations from 1.0 to 5000 μg/L were established using MIP and NIP as biomimetic antibodies, respectively.
Sample preparation
To verify the accuracy of the established method, blank carrot and cabbage samples spiked with trichlorfon were analyzed. Briefly, 2.0 g of samples were weighed into a small beaker and then spiked with 1.0 mL of trichlorfon standard solution (5.0 or 50.0 mg/L). After incubation for 4 h, the spiked samples were ultrasonicated with 3 × 10 mL of PBS for 30 min. The extracts were collected into a 50 mL volumetric flask and then diluted with PBS to 50 mL. After filtration through a 0.45-μm membrane, the extract was analyzed by the established FICL-BI method and the recovery rates were calculated.
Gas chromatography analysis
To identify the accuracy of the FICL-BI method, blank carrot and cabbage samples spiked with trichlorfon were also determined by GC according to the method described in GB/T 5009.20-2003 (China). Briefly, 50.0 g of carrot and cabbage samples were separately weighed into a 300-mL beaker and mashed. Then, 50 mL of DDW and 100 mL of acetone were added. The mixture was ultrasonicated for 20 min, and then 100 mL of the filtrate was transferred to a 500-mL separatory funnel. After 10 g of sodium chloride was added, the separatory funnel was shaken vigorously for 3 min. The aqueous phase was separated again by adding 2 × 50 mL of dichloromethane. All of the organic phases were collected. After being condensed by a rotary evaporator, the resulting extraction was diluted to 10 mL using chromatography grade dichloromethane. Finally, the extractions were filtered using a 0.22-μm filter membrane, and 1.0 μL of the filtrate was injected into the GC for analysis.
Statistical analysis
The ANOVA (SAS Institute, Cary, NC, USA) test was performed to evaluate the difference between the results obtained by the FICL-BI and GC methods.
Results and discussion
The equilibrium binding experiments of MIP
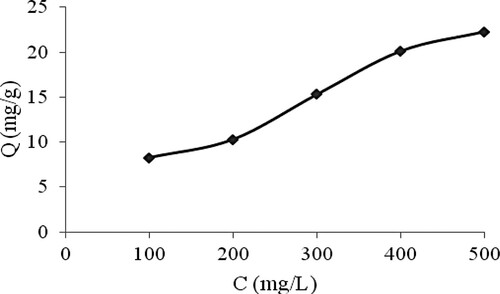
In this study, the adsorption capacities of the MIP toward trichlorfon at concentrations between 100 and 500 mg/L were investigated. As shown in , the adsorption capacities of MIP increased with increasing concentration of trichlorfon. An adsorption capacity of 20.13 mg/g was obtained when the trichlorfon concentration was 400 mg/L. Therefore, the prepared MIP had high adsorption performance, which has potential to be used as a biomimetic antibody in the immune reaction. This might result from the imprinting effect. During the polymerization step, the mixed templates were incorporated with the functional monomer. Subsequent removal of the template molecules resulted in imprinted cavities having structure, size, and spatial arrangement complementary to trichlorfon.
The stability of the CL marker
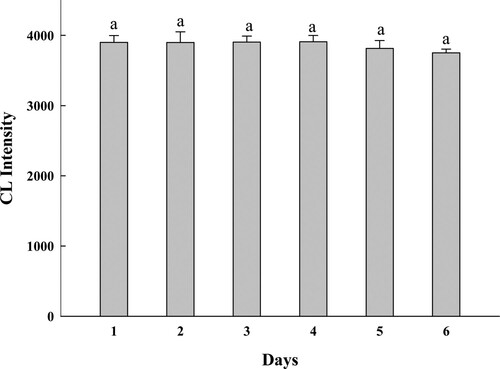
To study the stability of trichlorfon hapten–BSA–luminol marker, the CL intensity of the marker that was diluted five times with PBS was detected for six consecutive days. The result is shown in . The ANOVA (SAS Institute, Cary, NC, USA) test was performed to evaluate the difference between the CL intensities of the markers obtained on different days. There was no significant difference in the CL intensities (P > 0.05), which indicated that the synthetic trichlorfon hapten–BSA–luminol marker had good stability and could be used for subsequent experiments.
Optimization of the operating conditions of the FICL-BI
To improve the performance of the FICL-BI method, the operating conditions, such as the H2O2 concentration, preparation solution, and pH of the PBS solution, were optimized.
There are some serum proteins in markers. High concentration of H2O2 has influence on the protein. Furthermore, H2O2 as an oxidant in the luminescence system also has an important effect on the CL intensity of the trichlorfon hapten–BSA–luminol marker. Using NaHCO3-Na2CO3 (pH 9.5) as a buffer solution, the H2O2 concentrations in the range of 0.004–1.2 mol/L were tested. As shown in (a), the CL intensity increased with increasing H2O2 concentration, and reached a maximum when the H2O2 concentration was 0.4 mol/L. As the H2O2 concentration continued to increase, the intensity gradually decreased. Therefore, 0.4 mol/L H2O2 was chosen for subsequent experiments.
Figure 3. (a) The influence of H2O2 concentrations on CL intensity of the marker, (b) The effect of different preparation solutions on the inhibition of trichlorfon, and (c) The influence of PBS in different pH on the inhibition of trichlorfon.
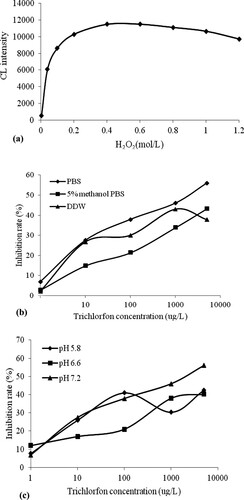
The preparation solution of the standard solution and samples affected the recognition ability of the MIP, which directly affected the competition reaction of the method. In this study, different preparation solutions were investigated. The results in (b) indicate that when PBS was used as the preparation solution, a higher inhibition rate and better competition effect were obtained compared with 5% methanol/PBS and DDW. Therefore, PBS was chosen for the optimal preparation solution.
The pH of the PBS solution was also an important factor for the biomimetic immunoassay. PBS solutions with pH values of 5.8, 6.6, and 7.2 were investigated. (c) shows that a better inhibition rate was obtained when the pH of PBS was 7.2. Thus, a PBS solution with a pH of 7.2 was selected as the preparation solution.
Selectivity of the FICL-BI method
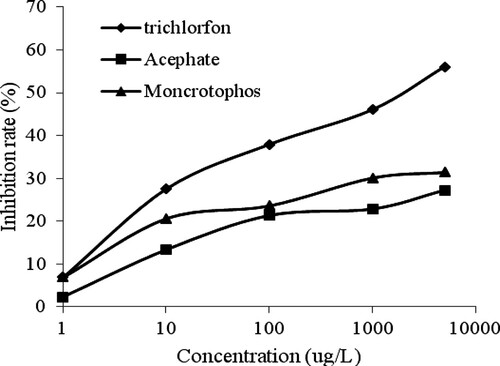
To evaluate the selectivity of the FICL-BI method, monocrotophos and acephate, two structurally analogous compounds of trichlorfon, were chosen for the cross-reactivity experiments. The results in show that the inhibition rate toward trichlorfon was higher than that toward monocrotophos and acephate at the same concentrations.
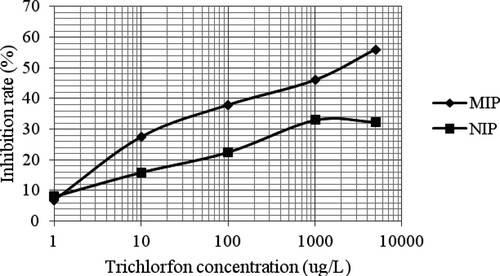
The standard curves with MIP and NIP as biomimetic antibodies are displayed in . A higher inhibition rate was obtained at the same concentrations when MIP was used as a biomimetic antibody compared with NIP. The inhibition rate was 56% for MIP at a trichlorfon concentration of 5000 μg/L, whereas an inhibition rate of 29.4% was obtained for NIP under the same conditions. These results showed that MIP had a higher selectivity for trichlorfon and could be used as a biomimetic antibody in the FICL-BI method.
Analytical parameters of the FICL-BI method
The analytical parameters of the developed FICL-BI method for the analysis of trichlorfon were evaluated. The sensitivity (IC50) of the established FICL-BI method for trichlorfon was 2 mg/L, and the limit of detection (IC15) was 0.0024 mg/L. According to the “Japanese Positive List System”, the maximum allowed concentration of trichlorfon residue in vegetables is 10 μg/L. Therefore, the FICL-BI analysis method can meet the requirements for the detection of trace trichlorfon.
Accuracy evaluation of the FICL-BI method
To verify the accuracy of the established FICL-BI method, recovery experiments were carried out by spiking trichlorfon at two different levels (2.5 and 25 μg/g) in blank carrot and cabbage samples (). Triple measurements were conducted at each concentration. Satisfactory recoveries between 74.47% and 94.48% were obtained.
Table 1. Analytical result of FICL-BI and GC methods (n = 3).
Comparative analysis of the spiked samples was performed by the GC method, and the recoveries of 81.19% to 104.75% were obtained. There was no significant difference between the results obtained by the above two methods (P > 0.05). Based on above results, we can conclude that the developed FICL-BI method had high accuracy and can be used as a tool to detect trichlorfon in vegetable samples.
Advantages of the developed FICL-BI method
The established FICL-BI method has many advantages, even though it has lower sensitivity than that of GC (Liu et al., Citation2008) and high-performance liquid chromatography (Li et al., Citation2017). First, using MIP as a biomimetic antibody can overcome the instability of biological antibodies. Moreover, the instruments are inexpensive and reagent consumption is low. Finally, using BSA as a carrier protein could result in the conjugation of more luminol, which would subsequently improve the luminous efficiency and stability of the experimental results. More importantly, the biomimetic antibody can be reused more than 10 times without the loss of sensitivity, which would reduce the analysis cost.
Conclusions
In this study, an FICL-BI method for the fast detection of trichlorfon was developed based on MIP as a biomimetic antibody. The presented method was applied for the detection of trichlorfon in vegetables with good recoveries. Thus, this study provides a simple tool for the analysis of trichlorfon pesticide in vegetable samples.
Disclosure statement
No potential conflict of interest was reported by the authors.
Additional information
Funding
Notes on contributors
Shoumin Wang
Shoumin Wang obtained her master’s degree from Shandong Agricultural University, Taian, China. Her research interests are chemiluminescence and immunoassay.
Mingdi Jiang
Mingdi Jiang obtained her bachelor’s degree from Shandong Agricultural University, Taian, China, in 2016. Her research interests are food immunoassay detection methods.
Zeliang Ju
Zeliang Ju was born in 1973, and is now an associate research fellow in Agricultural Bureau of Linqu, Shandong Province. His research interest is food analysis.
Xuguang Qiao
Xuguang Qiao was born in 1965, and is now a Professor in the College of Food Science and Engineering, Shandong Agricultural University. His research interests focus on vegetable processing technology, and food quality control.
Zhixiang Xu
Zhixiang Xu was born in 1973, and is now a Professor in the College of Food Science and Engineering, Shandong Agricultural University. His research interests focus on tailored (bio) molecular recognition interfaces, molecularly imprinted materials, and biomimetic immunosorbent assay.
References
- Biparva, P., Abedirad, S. M., & Kazemi, S. Y. (2015). Silver nanoparticles enhanced a novel TCPO-H2O2–safranin O chemiluminescence system for determination of 6-mercaptopurine. Spectrochimica Acta Part A Molecular and Biomolecular Spectroscopy, 145, 454–460. doi: https://doi.org/10.1016/j.saa.2015.03.019
- Chen, Q., & Fung, Y. (2010). Capillary electrophoresis with immobilized quantum dot fluorescence detection for rapid determination of organophosphorus pesticides in vegetables. Electrophoresis, 31(18), 3107–3114. doi: https://doi.org/10.1002/elps.201000260
- Chung, S. W., & Chan, B. T. (2010). Validation and use of a fast sample preparation method and liquid chromatography–tandem mass spectrometry in analysis of ultra-trace levels of 98 organophosphorus pesticide and carbamate residues in a total diet study involving diversified food types. Journal of Chromatography A, 1217(29), 4815–4824. doi: https://doi.org/10.1016/j.chroma.2010.05.043
- He, L., Luo, X., Jiang, X., & Qu, L. (2010). A new 1, 3-dibutylimidazolium hexafluorophosphate ionic liquid-based dispersive liquid–liquid microextraction to determine organophosphorus pesticides in water and fruit samples by high-performance liquid chromatography. Journal of Chromatography A, 1217(31), 5013–5020. doi: https://doi.org/10.1016/j.chroma.2010.05.057
- Kwong, T. C. (2002). Organophosphate pesticides: Biochemistry and clinical toxicology. Therapeutic Drug Monitoring, 24(1), 144–149. doi: https://doi.org/10.1097/00007691-200202000-00022
- Li, X., Hu, Y., Huo, T., & Xu, C. (2006). Comparison of the determination of chloramphenicol residues in aquaculture tissues by time-resolved fluoroimmunoassay and with liquid chromatography and tandem mass spectrometry. Food and Agricultural Immunology, 17(3–4), 191–199. doi: https://doi.org/10.1080/09540100601090349
- Li, J., Lu, J., Qiao, X., & Xu, Z. (2017). A study on biomimetic immunoassay-capillary electrophoresis method based on molecularly imprinted polymer for determination of trace trichlorfon residue in vegetables. Food Chemistry, 221, 1285–1290. doi: https://doi.org/10.1016/j.foodchem.2016.11.028
- Liu, B., Ge, Y., Zhang, Y., Song, Y., Lv, Y., & Wang, X. (2012). Production of the class-specific antibody and development of direct competitive ELISA for multi-residue detection of organophosphorus pesticides. Food and Agricultural Immunology, 23(2), 157–168. doi: https://doi.org/10.1080/09540105.2011.608120
- Liu, Y., Yang, C. Z., & Kang, Q. H. (2008). Determination of trichlorfon residues in foodstuffs of animal origin by pyrolysis gas chromatography. Chinese Journal of Analysis Laboratory, 27(2), 119.
- Li, D., Zhang, X., Kong, F., Qiao, X., & Xu, Z. (2017). Molecularly imprinted solid-phase extraction coupled with high-performance liquid chromatography for the determination of trace trichlorfon and monocrotophos residues in fruits. Food Analytical Methods, 10(5), 1284–1292. doi: https://doi.org/10.1007/s12161-016-0687-z
- Manclus, J. J., Primo, J., & Montoya, A. (1996). Development of enzyme-linked immunosorbent assays for the insecticide chlorpyrifos. 1. Monoclonal Antibody Production and Immunoassay Design . Journal of Agricultural and Food Chemistry, 44(12), 4052–4062. doi: https://doi.org/10.1021/jf960144q
- Qian, G. L., Wang, L. M., Wu, Y. R., Zhang, Q., Sun, Q., & Liu, Y. (2009). A monoclonal antibody-based sensitive enzyme-linked immunosorbent assay (ELISA) for the analysis of the organophosphorous pesticides chlorpyrifos-methyl in real samples. Food Chemistry, 117(2), 364–370. doi: https://doi.org/10.1016/j.foodchem.2009.03.097
- Qi, H., & Zhang, C. (2004). Homogeneous electrogenerated chemiluminescence immunoassay for the determination of digoxin. Analytica Chimica Acta, 501(1), 31–35. doi: https://doi.org/10.1016/j.aca.2003.09.009
- Sarabia, L., Maurer, I., & Bustos-Obregón, E. (2009). Melatonin prevents damage elicited by the organophosphorous pesticide diazinon on mouse sperm DNA. Ecotoxicology and Environmental Safety, 72, 663–668. doi: https://doi.org/10.1016/j.ecoenv.2008.04.023
- Schlaeppi, J. M. A., Kessler, A., & Foery, W. (1994). Development of a magnetic particle-based automated chemiluminescent immunoassay for triasulfuron. Journal of Agricultural and Food Chemistry, 42(9), 1914–1919. doi: https://doi.org/10.1021/jf00045a016
- Skerritt, J. H., Guihot, S. L., Asha, M. B., Bea, R., & Ngk, K. (2003). Sensitive immunoassays for methyl-parathion and parathion and their application to residues in foodstuffs. Food and Agricultural Immunology, 15(15), 1–15. doi: https://doi.org/10.1080/0954010031000138069
- Song, C., Liu, Q., Zhi, A., Yang, J., Zhi, Y., Li, Q., … Zhang, G. (2011). Development of a lateral flow colloidal gold immunoassay strip for the rapid detection of olaquindox residues. Journal of Agricultural and Food Chemistry, 59(17), 9319–9326. doi: https://doi.org/10.1021/jf202213m
- Sultatos, L. G. (1994). Mammalian toxicology of organophosphorus pesticides. Journal of Toxicology and Environmental Health, Part A Current Issues, 43(3), 271–289. doi: https://doi.org/10.1080/15287399409531921
- Sun, F., Betzendahl, I., Van Wemmel, K., Cortvrindt, R., Smitz, J., Pacchierotti, F., & Eichenlaub-Ritter, U. (2008). Trichlorfon-induced polyploidy and nondisjunction in mouse oocytes from preantral follicle culture. Mutation Research/Genetic Toxicology and Environmental Mutagenesis, 651(1–2), 114–124. doi: https://doi.org/10.1016/j.mrgentox.2007.10.008
- Wang, C., Li, X., Liu, Y., Guo, Y., Xie, R., Gui, W., & Zhu, G. (2010). Development of a Mab-based heterologous immunoassay for the broad-selective determination of organophosphorus pesticides. Journal of Agricultural and Food Chemistry, 58(9), 5658–5663. doi: https://doi.org/10.1021/jf904575k
- Wang, S., Xu, Z., Fang, G., Zhang, Y., Liu, B., & Zhu, H. (2009). Development of a biomimetic enzyme-linked immunosorbent assay method for the determination of estrone in environmental water using novel molecularly imprinted films of controlled thickness as artificial antibodies. Journal of Agricultural and Food Chemistry, 57(11), 4528–4534. doi: https://doi.org/10.1021/jf900505k
- Wang, B., Zhong, Y., Mao, X., & Wan, Y. (2015). A simple one-step extraction method for the determination of organophosphorus pesticides in shuanghuanglian and antivirus oral liquids by gas chromatography-tandem mass spectrometry. Analytical Methods, 7(16), 6821–6827. doi: https://doi.org/10.1039/C5AY01519G
- Xin, J., Qiao, X., Xu, Z., & Zhou, J. (2013). Molecularly imprinted polymer as sorbent for solid-phase extraction coupling to gas chromatography for the simultaneous determination of trichlorfon and monocrotophos residues in vegetables. Food Analytical Methods, 6(1), 274–281. doi: https://doi.org/10.1007/s12161-012-9432-4
- Xu, Z. X., Gao, H. J., Zhang, L. M., Chen, X. Q., & Qiao, X. G. (2011). The biomimetic immunoassay based on molecularly imprinted polymer: A comprehensive review of recent progress and future prospects. Journal of Food Science, 76(2), R69–R75. doi: https://doi.org/10.1111/j.1750-3841.2010.02020.x
- Yang, J. Y., Zhang, Y., Wang, H., Xu, Z. L., Eremin, S. A., Shen, Y. D., Wu, Qing, Lei, H.T., & Sun, Y. M. (2015). Development of fluorescence polarisation immunoassay for carbofuran in food and environmental water samples. Food and Agricultural Immunology, 26(3), 340-355. doi: https://doi.org/10.1080/09540105.2014.914890
- Zhang, Y. (2015). Development of chemiluminescent enzyme immunoassay for the determination of aflatoxin M1 in milk products. Food and Agricultural Immunology, 26(2), 204–217. doi: https://doi.org/10.1080/09540105.2014.884056
- Zhang, Y., Yang, J., Lei, H., Wang, H., Xu, Z., Shen, Y., … Sun, Y. (2015). Development of chemiluminescent enzyme immunoassay for the determination of malachite green in seafood. Food and Agricultural Immunology, 26(2), 204–217. doi: https://doi.org/10.1080/09540105.2014.884056