Abstract
There has been considerable discussions on why some types of haemoglobin-based blood substitutes increase vasoactivity whereas a very few others do not. In this study, we prepare four different types of PolyHb each containing different percentage of tetrameric hemoglobin using glutaraldehyde crosslinking and characterized to ensure that they all have the same oxygen affinity. Thus the preparations are prepared from the same chemical method and have the same oxygen affinity. We infused these in the form of 1/6 volume toploading into anesthetized rats to simulate the use of blood substitutes in surgery. Mean arterial pressure (MAP) increased immediately after injection of PolyHb containing 38% or 78% of tetrameric hemoglobin. However, there was no significant increase in blood pressure with the injection of PolyHb containing 16% or 0.4% tetrameric hemoglobin. In electrocardiogram (ECG) study, we observe that high percentage (78%) of tetrameric hemoglobin causes marked changes in ECG immediately after infusion. Injection of PolyHb containing 16% or 38% of tetrameric hemoglobin resulted in minimal elevation of the ST segment. Infusion of PolyHb containing 0.4% of tetrameric hemoglobin did not result in any changes.
INTRODUCTION
There have been considerable discussions on the reasons why infusion of most hemoglobin-based blood substitutes result in increased vasoactivity whereas a few others do not [Citation[1-16]]. One proposal is that the tetrameric hemoglobin component transverses the intercellular junction of the endothelial cell lining and binds nitric oxide from the interstitial space needed for normal vasodilatation [Citation[3-5], Citation[9], Citation[10], Citation[14]]. Another proposal is that it is the rapid release of oxygen that causes the increase in vasoactivity [Citation[16]]. It is likely that both proposals could play some role. Support for the proposal of tetrameric hemoglobin stems from the observation that the infusion of tetrameric hemoglobin blood substitutes prepared by intramolecular crosslinking or by recombinant method increase vasoactivity [Citation[1], Citation[7], Citation[8], Citation[15]]. When recombinant tetrameric hemoglobin is prepared to have low affinity for nitric oxide, they no longer increase vasoactivity when infused [Citation[7]]. Also, polyhemoglobin or conjugated hemoglobin preparation that contain higher percentage of tetrameric hemoglobin also increase vasoactivity when infused in larger amounts [Citation[6]]. There is no reported observation of increased vasoactivity with the infusion of large volumes of polyhemoglobin containing <1% of tetrameric hemoglobin [Citation[9], Citation[10]]. In addition to oxygen affinity and percentage of tetrameric hemoglobin, another proposal is that differences in vasoactivity could be related to the differences in the method of preparation and the chemistry involved [Citation[1]]. In the present study we prepare four different types of PolyHb, each containing different percentage of tetrameric hemoglobin using glutaraldehyde crosslinking and characterized to ensure that they all have the same oxygen affinity. Thus the samples are prepared from the same chemical method and have the same oxygen affinity. In this way, we try as much as possible to eliminate two variables, variation in hemoglobin modification and oxygen affinity, and test only the effects of percentage of tetrameric hemoglobin. We infused these in the form of 1/6 volume toploading into anesthetized rats and look at the effects of on vasoactivity. With the report of possible cardiac effects of some hemoglobin–based blood substitutes, we also follow the electrocardiogram. It is well known that there are differences in vasoactivity responses between anesthetized and unanesthetized animals. We choose to use anesthetized rats rather than unanesthetized rats partly because of animal ethics committee requirements and partly to simulate the use of polyhemoglobin in surgery. We also used the experimental results obtained to calculate the total amount of tetrameric hemoglobin that would result in an increase in blood pressure and ECG changes. We then use this to analyze the amount of tetrameric hemoglobin present in different preparations of PolyHb in 1/6, 1/3, 2/3 and 3/3 blood replacement. It should be emphasized that the polyHb being studied here are prepared in this laboratory for basic research and should not be considered to be comparable to any of the polyhemoglobin prepared by the industries.
MATERIALS AND METHODS
Materials
Glutaraldehyde (25%), L-lysine (monohydrochloride, SigmaUltra > 99%), L-tyrosine (98% TLC), Sephacryl-300 HR, were purchased from Sigma-Aldrich (Ontario, Canada). Purified bovine hemoglobin was purchased from Biopure Biopharmaceuticals (Cambridge, MA, USA). All other reagents were of analytical grade.
Preparation of PolyHb Containing Different Percentage of Tetrameric Hemoglobin
(1) PolyHb Contain 78% of Low Molecular Weight Molecules (<100 KDa)
Reaction mixtures were prepared containing hemoglobin (9–11 g/dl). Prior to the start of crosslinking, 1.3 M lysine was added at a molar ratio of 5:1 lysine/hemoglobin. Crosslinking reaction was started with the addition of glutaraldehyde (5%) at molar ratio of 5:1 glutaraldehyde/hemoglobin. Glutaraldehyde was added in four equal aliquots over a period of 15 minutes. After 3 hours at 4°C under aerobic conditions with constant stirring, reaction was stopped with 2.0 M lysine at a molar ratio of 200:1 lysine/hemoglobin. Solutions were dialyzed against lactated ringer's solution overnight and passed through sterile 0.45 µM filter. Aliquots (4 ml) of the crosslinked preparation were concentrated using 100,000 MWCO Amicon Ultra-4 centrifugal filter devices (Millipore Corporation, Ontario, Canada). Samples were centrifuged at 4000 rpm for 60 minutes at 4°C. Then, retentate was collected. Hemoglobin concentration was determined by cyanomethemoglobin at 540 nm with Perkin-Elmer Lambda 4B UV/VIS Spectrophotometer.
(2) PolyHb Contain 38% of Low Molecular Weight Molecules (<100 KDa)
Reaction mixtures were prepared containing hemoglobin (9–11 g/dl). Prior to the start of crosslinking, 1.3 M lysine was added at a molar ratio of 5:1 lysine/hemoglobin. Crosslinking reaction was started with the addition of glutaraldehyde (5%) at molar ratio of 10:1 glutaraldehyde/hemoglobin. Glutaraldehyde was added in four equal aliquots over a period of 15 minutes. After 24 hours at 4 °C under aerobic conditions with constant stirring, reaction was stopped with 2.0 M lysine at a molar ratio of 200:1 lysine/hemoglobin. Solutions were dialyzed against lactated ringer's solution overnight and passed through sterile 0.45 µM filter. Aliquots (4 ml) of the crosslinked preparation were concentrated using 100,000 MWCO Amicon Ultra-4 centrifugal filter devices (Millipore Corporation, Ontario, Canada). Samples were centrifuged at 4000 rpm for 60 minutes at 4°C. Then, retentate was collected. Hemoglobin concentration was determined by cyanomethemoglobin at 540 nm with Perkin-Elmer Lambda 4B UV/VIS Spectrophotometer.
(3) PolyHb Contain 16% of Low Molecular Weight Molecules (<100 KDa)
Reaction mixtures were prepared containing hemoglobin (9–11 g/dl). Prior to the start of crosslinking, 1.3 M lysine was added at a molar ratio of 6:1 lysine/hemoglobin. Crosslinking reaction was started with the addition of glutaraldehyde (5%) at molar ratio of 16:1 glutaraldehyde/hemoglobin. Glutaraldehyde was added in four equal aliquots over a period of 15 minutes. After 24 hours at 4°C under aerobic conditions with constant stirring, reaction was stopped with 2.0 M lysine at a molar ratio of 200:1 lysine/hemoglobin. Solutions were dialyzed against lactated ringer's solution overnight and passed through sterile 0.45 µM filter. Aliquots (4 ml) of the crosslinked preparation were concentrated using 100,000 MWCO Amicon Ultra-4 centrifugal filter devices (Millipore Corporation, Ontario, Canada). Samples were centrifuged at 4000 rpm for 60 minutes at 4°C. Then, retentate was collected. Hemoglobin concentration was determined by cyanomethemoglobin at 540 nm with Perkin-Elmer Lambda 4B UV/VIS Spectrophotometer.
(4) PolyHb Containing 0.4% of Low Molecular Weight Molecules (<100 KDa)
Preliminary study was also carried out using PolyHb containing 0.4% low molecular weight molecules (<100 KDa). Method of preparation will be described elsewhere [Citation[17]].
Molecular Weight Distribution
Samples from the above cross-linked PolyHb solutions were analyzed by size-exclusion gel filtration liquid chromatography. A Sephacryl-300 HR column (1.6 cm × 70 cm, Vtotal = 105 ml), was prepared and equilibrated with 0.1 M Tris · HCl (pH 7.5). Samples (3.5 mg Hb/250 µl) were passed through the column at a flow rate of 15 ml/hour. The eluting fractions were collected every 15 minutes for a period of 8 hours. The elution profiles were monitored at 280 nm.
Determination of Oxygen Affinity of PolyHb
The oxygen absorption curves for PolyHb were determined by the TCS Hemoxanalyser (TCS Medical Products Co., USA). Samples (4 ml) containing 0.2 g/dl of PolyHb at 37°C 0.1 M PBS at pH 7.4 were used to obtain hemoglobin oxygen dissociation curves.
The Effect of Intravenous Injection of PolyHb on Animal
Male Wistar rats (180–270 g) were purchased from Charles River (St-Constant, Quebec, Canada). They were kept in a controlled 12-hr light/dark environment with food and water ad libitum. All animal experiments were performed according to the regulations of McGill University on Animal Care. In this study, three animal groups (3 rats in each group) were tested: (1) intravenous injection with PolyHb containing 78% tetrameric hemoglobin; (2) intravenous injection with PolyHb containing 38% tetrameric hemoglobin; (3) intravenous injection with PolyHb containing 16% tetrameric hemoglobin; (4) preliminary study of intravenous injection with PolyHb containing 0.4% tetrameric hemoglobin was also carried out. The rats were first anaesthetized with intraperitoneal injection of sodium pentobarbital (65 mg/kg, Somnotol, Decton Dickinson, NJ, USA). Polyethylene cannulae were inserted and secured distal to the superficial epigastric branches in the femoral arteries (PE-10, Clay Adams) and veins (PE-50, Clay Adams). Both the venous and arterial catheters were filled with heparin saline (50 IU/ml) to avoid blood clotting. Samples were injected through femoral vein. The measurement of blood pressure was through femoral artery, and it was connected to the BIOPAC system (BIOPAC Systems, Inc., Goleta, CA, USA). When cannulations were done, thirty minutes control blood pressure and electrocardiogram (ECG) were recorded, and then the sample was injected into the vein catheter using a peristaltic pump (Minipuls 2, Gilson Medical Electronics, Middleton, WI, USA) with injection flow rate at 0.5 ml/min. The total sample injection amount was 1/6 of the whole blood volume in rat. After the injection, blood pressure was continuously monitored for another two hours. During the time of monitoring, another dose of sodium pentobarbital was given if the rat woke up. When all monitoring procedures were done, catheters were removed, the vessels were ligated, and local skin was sutured.
RESULTS
Molecular Weight Distribution
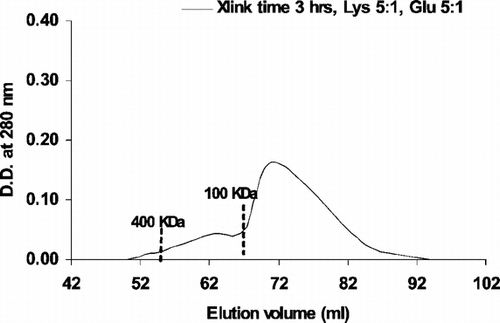
(1) PolyHb Contain 78% of Low Molecular Weight Molecules (<100 KDa)
To determine the percentage of small molecular weight in PolyHb, sample was analyzed by gel filtration chromatography using a Sephacryl-300 HR column. shows that with crosslinking time of 3 hours, initial lysine (Lys) ratio at 5:1 lysine:hemoglobin and glutaraldehyde (Glu) ratio at 5:1 glutaraldehyde:hemoglobin, the percentage of molecular weight.
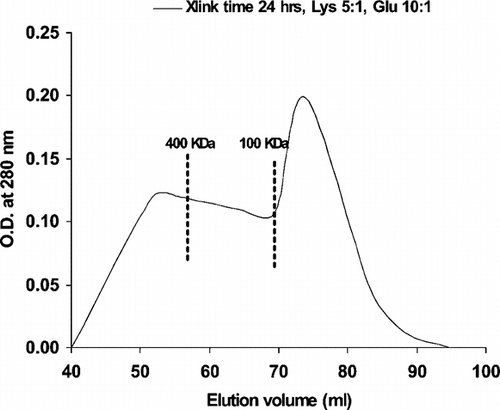
(2) PolyHb Contain 38% of Low Molecular Weight Molecules (<100 KDa)
In this study, we keep initial lysine ratio at 5:1 lysine:hemoglobin, but increase glutaraldehyde ratio to 10:1 glutaraldehyde:hemoglobin and increase crosslinking time to 24 hours. Based on our elution profile of PolyHb in , the percentage of small molecules is 38%.
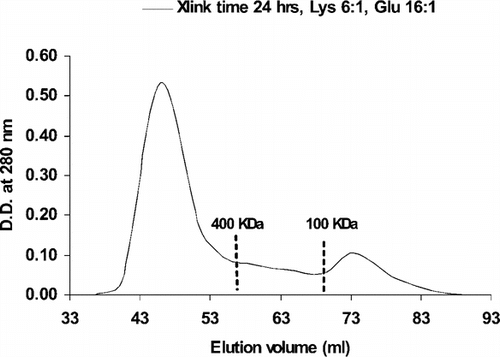
(3) PolyHb Contain 16% of Low Molecular Weight Molecules (<100 KDa)
We use initial lysine ratio at 6:1 lysine:hemoglobin, glutaraldehyde ratio of 16:1 and crosslinking time of 24 hours. From our results in , the percentage of small molecular weight molecules is 16%.
(4) PolyHb Containing 0.4% of Low Molecular Weight Molecules (<100 KDa). Details will be reported elsewhere [Citation[17]]
Oxygen Affinity of PolyHb Preparations
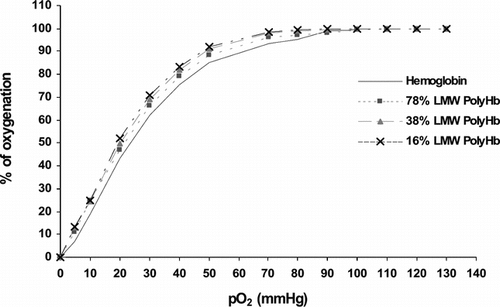
We then studied the oxygen characteristics of PolyHb, which contain different percentage of low molecular weight (<100 KDa). The results in show that there is no significant difference between the different preparations of PolyHb and the original ultrapure hemoglobin used for these PolyHb.
Figure 4 Oxygen dissociation curve of hemoglobin in the free form and PolyHb form. Hemoglobin: free form; 78% LMW PolyHb: contains 78% of low molecular weight molecules (<100 KDa); 38% LMW PolyHb: contains 38% of low molecular weight molecules (<100 KDa); 16% LMW PolyHb: contains 16% of low molecular weight molecules (<100 KDa).
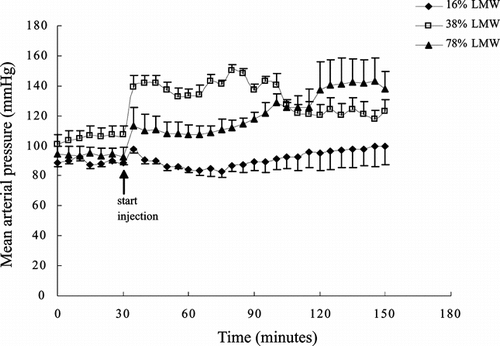
The Effect of Intravenous Infusion on Mean Arterial Pressure (MAP)
In the following study, we investigated the effects of these PolyHb preparations in rats. The mean arterial pressure (MAP) for the 30 minutes before infusion served as the baseline. Our results showed that infusion of 1/6 the total blood volume of 16% low molecular weight PolyHb did not increase the mean arterial pressure (). On the other hand, infusion of PolyHb containing 38% and 78% of tetrameric hemoglobin markedly increased blood pressure immediately after injections. At 100 minutes, the blood pressure in these two groups started to decrease. Preliminary study showed that similar infusion of PolyHb containing 0.4% of tetrameric hemoglobin did not result in any changes in MAP.
Figure 5 Mean arterial pressure (MAP) with injections of PolyHb samples containing 16%, 38% and 78% of low molecular weight (LMW) components respectively. Injections were started at 30 minutes. All values are represented as mean ±SEM. Preliminary study using PolyHb with 0.4% tetrameric haemoglobin did not show any changes in MAP.
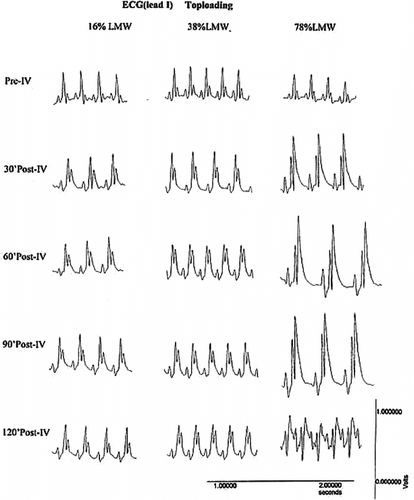
The Effect of Intravenous Infusion on Electrocardiogram
showed the effects of infusions of different preparations of PolyHb on ECG. There were minor ST elevations in the two groups with infusions of PolyHb containing 16% or 38% tetrameric hemoglobin. However, in the group with infusion of 78% tetrameric hemoglobin, the QRS was markedly changed when compared to the first two groups. For this group the marked change was maintained for at least 3 hours. Preliminary study of 1/6 toploading using PolyHb containing 0.4% tetrameric hemoglobin shows no significant changes in ECG.
Figure 6 Changes in electrocardiogram (ECG) after injection of 1/6 toploading with PolyHb containing 16%, 38% and 78% of tetramers at 30, 60, 90, and 120 minutes. Pre-IV: before intravenous injection; Post-IV: after intravenous injection. Preliminary study shows that similar toploading using PolyHb with 0.4% tetramers did not result in any changes.
Analysis of the Theoretical Effects of Increasing Volumes of Infusion
The above observation is only for the infusion of PolyHb in volumes equal to 1/6 the total blood volume. With increase in the volume of PolyHb infused, the total amount of tetrameric hemoglobin will also increase. We therefore calculated the amount of tetrameric Hb that would be present in blood volume replacement of 1/6, 1/3, 1/2 and 3/3 using PolyHb containing respectively 1%, 2%, 4%, 8%, 16%, 38% and 78% tetramers (). When given as 1/6 toploading, the total amounts of tetramers present are 0.093 g/Kg in PolyHb with 16% tetramer, 0.222 g/Kg in PolyHb with 38% tetramer and 0.455 g/Kg in PolyHb with 78% tetramer. Since our results show that 1/6 toploading with 38% tetramers cause significant increase in blood pressure but 16% did not, we can say that total tetramers of 0.222 g/Kg or more would cause a significant increase in blood pressure, whereas total tetramers of 0.093/Kg or less would not cause a significant increase. With increase in blood volume replacement, e.g. 1/3, 1/2 or 3/3, the total amount of tetramers present in the larger volume of polyHb will increase. Therefore, from this we calculate the theoretical point where the total tetrameric hemoglobin will be equal to the amount that causes significant increases in the mean blood pressure for 1/6, 1/3, 1/2 and 3/3 volume replacement using PolyHb containing 1%, 2%, 4%, 8%, 16%, 38% and 78% tetramers. As shown in and , for PolyHb containing 1% or 2% tetrameric hemoglobin, the total tetrameric hemoglobin levels for up to 3/3 volume replacement do not reach the amount that would result in significant increases in MAP. For PolyHb with 4% tetramers 1/6, 1/3 and 1/2 would not, but 3/3 volume replacement would have total tetrameric hemoglobin higher than the 0.093 g/Kg that would not result in a significant increase in MAP but lower than that of 0.222 g that would result in a significant increase in MAP. Thus it is likely that for PolyHb containing 4% tetramers variable results could be obtained. PolyHb with 8% tetramers contain a total amount of tetrameric hemoglobin equivalent to the amount that could cause significant increases in mean arterial blood pressure when the volume replacement is 3/3 of total blood volume. Theoretically, 1/6 and 1/3 volume replacement would not and 1/2 replacement may have variable results.
Table 1 Analysis of the total amounts of tetrameric hemoglobin
Figure 7 The amount of tetrameric Hb (g/Kg body weight) in PolyHb with 1%, 2%, 4%, 8%, 16%, 38%, and 78% of tetrameric Hb for 1/6, 1/3, 1/2, and 3/3 blood volume replacement. The top dash line represents the tetramer amount (0.455 g/Kg body weight) where severe changes occur in ECG. The middle line shows the tetramer level where blood pressure started to markedly increase (0.222 g/Kg body weight), and this is also the highest concentration that do not cause marked changes in ECG, except for a slight elevation of the ST segment. The bottom horizontal line shows the level of tetrameric Hb (0.093 g/Kg body weight) that did not cause an increase in arterial blood pressure.
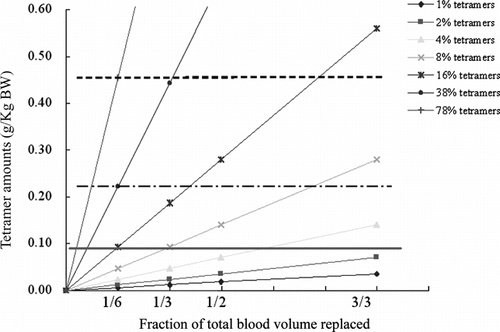
The above analysis corresponds to the actual observations by a number of investigators using different types of blood substitutes. Thus, PolyHb with less than 1% of tetramers does not result in vasoactivity, even when used in large volume replacement [Citation[9], Citation[10]]. For PolyHb containing <4% tetramers, some vasoactivity may be found when given in large amounts, but this is variable. For those preparations that contain 100% tetrameric hemoglobin, there is vasoactivity when smaller volumes are infused [Citation[7], Citation[8], Citation[15]].
DISCUSSION
There are three theories proposed for the observation that infusion of some hemoglobin-based blood substitutes result in increased vasoactivity whereas a few others do not result in increased vasoactivity. One theory proposes that tetrameric hemoglobin transverses the intercellular junction of the endothelial cell lining and binds nitric oxide from the interstitial space needed for normal vasodilatation [Citation[3-5], Citation[9], Citation[10], Citation[14]]. Another theory is that most modified hemoglobin in the soluble form, unlike hemoglobin inside red blood cells, release oxygen too rapidly resulting in increased vasoactivity [Citation[16]]. A third proposal is that this can be related to the type of modification used [Citation[1]]. It is likely that all three factors play their roles in different situations. The proposal of tetrameric hemoglobin has much support since infusion of tetrameric hemoglobin blood substitutes prepared by intramolecular crosslinking or by recombinant method increase vasoactivity [Citation[1], Citation[7], Citation[8], Citation[15]]. When recombinant tetrameric hemoglobin is prepared to have low affinity for nitric oxide, they no longer increase vasoactivity when infused [Citation[7]]. Also, polyhemoglobin or conjugated hemoglobin preparation that contain higher percentage of tetrameric hemoglobin also increase vasoactivity when infused in larger amounts [Citation[6]]. Furthermore, there is no reported observation of increased vasoactivity with the infusion of large volumes of polyhemoglobin containing <1% of tetrameric haemoglobin [Citation[9], Citation[10]]. In the present study we attempt to eliminate two variables, variation in hemoglobin modification and oxygen affinity, and test only the effects of tetrameric hemoglobin. We do this by preparing four different types of PolyHb, each containing different percentage of tetrameric hemoglobin. All four samples are prepared using glutaraldehyde crosslinking and characterized to ensure that they all have the same oxygen affinity. Thus the preparations are prepared from the same chemical method and have the same oxygen affinity.
Our results seem to show that unlike PolyHb with low percentage of tetrameric hemoglobin, the presence of high percentage of tetrameric hemoglobin in PolyHb preparations results in increased vasoactivity. With the report of possible cardiac effects of some hemoglobin-based blood substitutes, we also follow the electrocardiogram. What is also very important is the observation that high levels of tetrameric hemoglobin in the polyhemoglobin preparation can result in changes in the electrocardiogram. Low level of tetrameric hemoglobin did not result in changes in the electrocardiogram. Although the percentage of tetrameric hemoglobin in polyHb preparation is important, the total amount of blood volume replaced should also be taken into consideration, since increasing the volume of PolyHb also results in an increase in the total amount of tetrameric hemoglobin. This would apply to both effects on vasoactivity and ECG. It should be emphasized that the above analysis only applies to rats under general anesthesia to simulate the use of PolyHb in surgery. It is well known that the vasoactivity of unanesthetized animals is much more sensitive. However, we are restrained by animal ethics committee from using unanesthetized animals. Finally, it should be mentioned again that the polyHb preparations in this study are prepared in this laboratory for basic research and are not in any way comparable to any of the products prepared by the industries.
The authors gratefully acknowledge the operating grant from the Canadian Institutes of Health Research (TMSC, principle investigator) and the MSSS-FRSQ Research Group (d'equipe) award from the Quebec Ministry of Health's Hemovigillance and Transfusion Medicine Program (TMSC, principle investigator).
REFERENCES
- Alayash, A. (1999). Hemoglobin-based blood substitutes: Oxygen carriers, pressor agents, or oxidants? Nature Biotechnology 17(6): 545–549, [PUBMED], [INFOTRIEVE], [CSA]
- Caron, A., Malfatti, E., Aguejouf, O., Faivre-Fiorina, B., Menu, P. (2001). Vasoconstrictive response of rat mesenteric arterioles following infusion of cross-linked, polymerized, and conjugated hemoglobin solutions. Artif. Cells Blood Substit. Immobil. Biotechnol. 29(1): 19–30, [PUBMED], [INFOTRIEVE], [CROSSREF], [CSA]
- Chang, T.M.S. (1997). Blood Substitutes: Principles, Methods, Products and Clinical Trials, Basel, New York: Karger Landes Systems. (free access at www.medicine.mcgill.ca/artcell/bloodsubvol1.pdf).
- Chang, T.M.S. (2000). Red blood cell substitutes. Best Practice & Research: Clinical Haematology 13(4): 651–668, [CROSSREF], [CSA]
- Chang, T.M.S. (2003). New generations of red blood cell substitutes. J Internal Medicine 253: 527–535, [CROSSREF], [CSA]
- De Angelo, J. (1999). Nitric oxide scavengers in the treatment of shock associated with systemic inflammatory response syndrome. Exp. Opin. Pharmacother. 1: 19–29, [CROSSREF], [CSA]
- Doherty, D.H., Doyle, M.P., Curry, S.R., Vali, R.J., Fattor, T.J., Olson, J.S., Lemon, D.D. (1998). Rate of reaction with nitric oxide determines the hypertensive effect of cell-free hemoglobin. Nature Biotechnology 16: 672–676, [PUBMED], [INFOTRIEVE], [CROSSREF], [CSA]
- Freytag, J.W., Templeton, D. (1997). OptroTM (recombinant human hemoglobin): A therapeutic for the delivery of oxygen and the restoration of blood volume in the treatment of acute blood loss in trauma and surgery, in Red Cell Substitutes; Basic Principles and Clinical Application, A.S. Rudolph, R. Rabinovici, G.Z. Feuerstein, Eds., Marcel Dekker, Inc.: New York, pp. 325–334.
- Gould, S.A., Moore, E.E., Hoyt, D.B., Burch, J.M., Haenel, J.B., Garcia, J., DeWoskin, R., Moss, G.S. (1998). The first randomized trial of human polymerized hemoglobin as a blood substitute in acute trauma and emergent surgery. J. Am. Coll. Surg. 187: 113–120, [PUBMED], [INFOTRIEVE], [CROSSREF], [CSA]
- Gould, S.A., Moore, E.E., Hoyt, D.B., Ness, P.M., Norris, E.J., Carson, J.L., Hides, G.A., Freeman, I.H., DeWoskin, R., Moss, G.S. (2002). The life-sustaining capacity of human polymerized hemoglobin when red cells might be unavailable. J. Am. Coll. Surg. 195(4): 445–452, [PUBMED], [INFOTRIEVE], [CROSSREF], [CSA]
- LaMuraglia, G.M., O'Hara, P.J., Baker, W.H., Naslund, T.C., Norris, E.J., Li, J., Vandermeersch, E. (2000). The reduction of the allogenic transfusion requirement in aortic surgery with a hemoglobin-based solution. J. Vasc. Surg. 31: 299–308, [PUBMED], [INFOTRIEVE], [CROSSREF], [CSA]
- Sprung, J., Kindscher, J.D., Wahr, J.A., Levy, J.H., Monk, T.G., Moritz, M.W., O'Hara, P.J. (2002). The use of bovine hemoglobin glutamer-250 (Hemopure) in surgical patients: Results of a multicenter, randomized, single-blinded trial. Anesth Analg 94: 799–808, [PUBMED], [INFOTRIEVE], [CROSSREF], [CSA]
- Lieberthal, W., Fuhro, R., Alam, H., Rhee, P., Szebeni, J., Hechtman, H.B., Favuzza, J., Veech, R.L., Valeri, C.R. (2002). Comparison of the effects of a 50% exchange-transfusion with albumin, hetastarch, and modified hemoglobin solutions. Shock 17(1): 61–69, [PUBMED], [INFOTRIEVE], [CROSSREF], [CSA]
- Matheson, B., Kwansa, H.E., Bucci, E., Rebel, A., Koehler, R.C. (2002). Vascular response to infusions of a nonextravasating hemoglobin polymer. J. Appl. Physiol. 93: 1479–1486, [PUBMED], [INFOTRIEVE], [CSA]
- Vuylsteke, A., Davidson, H.J., Ho, W.S., Ritchie, A.J., Callingham, B.A., White, R., Hiley, C.R. (2001). Effect of the blood substitute diaspirin crosslinked hemoglobin in rat mesenteric and human radial collateral arteries. J. Cardiovasc. Pharmacol. 37(4): 394–405, [PUBMED], [INFOTRIEVE], [CROSSREF], [CSA]
- Winslow, R. (2003). Current status of blood substitute research: Towards a new paradigm. J. Int. Med. 253: 508–517, [CROSSREF], [CSA]
- Chang, T.M.S. Personal communication.