ABSTRACT
Objective: To estimate the health and economic impact of the reduction in mortality and cardiovascular hospitalizations, associated with correct diagnosis of cardiac transthyretin amyloidosis (ATTR-CM), from the Spanish National Health System (NHS) perspective.
Methods: A costs and effects analysis were performed (probabilistic Markov model) with time horizons between 1 and 15 years, comparing the correct diagnosis of ATTR-CM versus the non-diagnosis. Transition probabilities were obtained from the ATTR-ACT study (placebo arm) and from the literature. Costs and healthcare resources were obtained from Spanish sources (€ 2019) and from a panel of Spanish clinical experts.
Results: After 1, 5, 10 and 15 years, the diagnosis of ATTR-CM would generate a gain of 0.031 (95%CI 0.025; 0.038); 0.387 (95%CI 0.329; 0.435); 0.754 (95%CI 0.678; 0.781) and 0.944 (95%CI 0.905; 0.983) life years per patient, respectively, with savings of € 212 (95%CI € −632; 633), € 2,289 (95%CI € 2,250; 2,517), € 2,859 (95%CI € 2,584; 3,149) and € 2,906 (95%CI € 2,669; 3,450) per patient, respectively, versus the non-diagnosis.
Conclusions: Just by correctly diagnosing ATTR-CM, years of life would be gained, cardiovascular hospitalizations would be avoided, and savings would be generated for the NHS, compared to the non-diagnosis of the disease.
1. Introduction
Transthyretin amyloidosis (ATTR) is a set of diseases characterized by the deposit of transthyretin in several locations of the body [Citation1]. Transthyretin amyloid cardiomyopathy (ATTR-CM) is caused by the accumulation of transthyretin amyloid fibers in the myocardium causing cardiomyopathy, heart failure (HF), and ultimately, death [Citation1]. ATTR-CM is a degenerative disease whose prevalence is currently unknown and greatly underdiagnosed. Its symptoms may be confused with other forms of HF [Citation2]. This fact together with the lack, until now, of an available treatment has led to a situation in which the patient ends up receiving only symptomatic treatment for HF. Nevertheless, in patients with ATTR-CM, this treatment should be adjusted since some of the drugs that are used are not appropriate for controlling the symptoms of the disease, and may even worsen the patient’s clinical condition [Citation2]. ATTR-CM has a large impact on mortality, with median survival in affected patients that ranges between 24 and 66 months [Citation3]. similar to what can be observed, for example, in some cancer patients [Citation4,Citation5].
In February 2020, the European Commission approved the indication of tafamidis in ‘the treatment of hereditary or wild-type transthyretin amyloidosis in adult patients with cardiomyopathy (ATTR-CM)’ [Citation6]. To date, it is the only drug approved for the treatment of ATTR-CM.
Currently there are no economic studies that evaluate the consequences that the possible worsening due to the incorrect symptomatic treatment may be causing in these patients. For this reason, our objective is to analyze whether an incorrect diagnosis in patients with ATTR-CM that results in symptomatic HF treatment with no adjustments may be causing a loss of health outcomes, as well as an increase in the associated direct healthcare costs [Citation2]. Specifically, the study objective was to estimate the health and economic impact of the reduction in mortality and hospitalization rate due to cardiovascular causes, resulting exclusively from the correct diagnosis of ATTR-CM, thus without taking into account the costs associated with starting a specific therapy.
2. Design and methods
2.1. Markov model
A probabilistic economic Markov model was created using a second order Monte Carlo simulation with several time horizons (1, 5, 10, and 15 years). The model was developed using the TreeAge Pro Healthcare 2020 programme (R1.0) [Citation7].
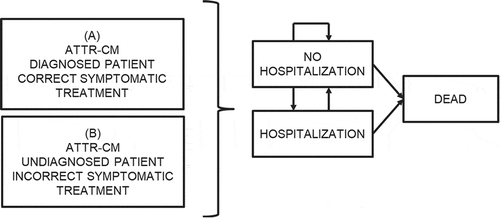
The model structure (Markov states and patient transitions between those states) is presented in . The simulation starts with the patients in a hypothetical cohort with ATTR-CM who are not hospitalized for cardiovascular reasons. These patients may or may not be correctly diagnosed (the consequences of this fact are those analyzed in this study). Subsequently, in 1-year periods (the duration of the model cycles) a portion of these patients may continue without being hospitalized (continue in the ‘no hospitalisation’ state), they may require hospitalization for cardiovascular causes (they move into the ‘hospitalisation’ state), or they may die of any cause (they move into the ‘death’ state). The hospitalized patients may recover, returning to the ‘no hospitalisation’ state or they may die.
Figure 1. Markov model. Analysis: A vs B
2.2. Model variables
The assumptions of the model were made based on data extracted from the literature. These assumptions were validated by a panel of Spanish experts, 3 clinicians and a hospital pharmacist, with experience with ATTR-CM. The model variables were the probabilities of transition between the Markov states and the annual costs per patient in each Markov state.
2.3. Transition probabilities
The diagnosis of ATTR-CM assumes that the patient would receive a correctly adjusted symptomatic treatment (i.e. ‘correct symptomatic treatment’), with the resulting reduction in mortality and hospitalization due to cardiovascular causes. To represent this population, the results observed in the placebo arm of the ATTR-ACT study [Citation8] were used; this was the pivotal clinical trial of tafamidis in ATTR-CM that included patients with both the wild-type (ATTRwt) and hereditary (ATTRv) form of ATTR-CM. The model considered the results obtained in the placebo arm, representing those patients who, being correctly diagnosed, received a correct symptomatic treatment adjusted to their disease. In the undiagnosed patients, who would receive an ‘incorrect symptomatic treatment’, a mortality and hospitalization rate similar to that of patients with uncontrolled HF was assumed [Citation9–11]. The mortality rate of hospitalized patients was obtained from the study by Carrasco-Sánchez et al. [Citation12], conducted in our setting. The hospitalization rates for cardiovascular causes and mortality used in the model are presented in . The transition probabilities (Pt) between the Markov states are calculated based on the annual rates using the formula: Pt = 1-e−rt, where r is the observed rate and t the time in which it would occur (1 year in the case of our model, the duration of one cycle, i.e. the period in which the transitions between the states occur) [Citation13].
Table 1. Markov model probabilities
2.4. Costs of Markov states
summarizes the annual costs per patient of the different states (diagnosis [only in year 1], hospitalization for cardiovascular causes, ‘correct’ and ‘incorrect’ symptomatic treatment). The cost of diagnosis (€ 928.18 ± 529.71), which is only computed at the start of the simulation, and of hospitalization for cardiovascular causes, assuming that it would correspond to diagnosis-related group (DRG) 127, for HF (€ 4,611.34 ± 1,037.49), were obtained from the average of the public costs of the 17 Autonomous Communities of Spain (year 2019). The use of resources to diagnose ATTR-CM was estimated by the panel of clinical experts ().
Table 2. Economic model costs (€ 2019)
It was assumed, according to the opinion of the expert panel, that the cost of symptomatic treatment would be different with or without the ATTR-CM diagnosis. That way, according to the panel, it was estimated that the annual cost of ‘correct’ symptomatic treatment of patients with diagnosed ATTR-CM would be € 225.60 ± 23.02 (). To perform this estimate, the experts assessed the use in clinical practice of the different pharmacological treatments for patients with HF (diuretics, beta-blockers, oral anticoagulants, ACE inhibitors, ARBs, and others) (). The estimates from the experts agreed with the results obtained in the THAOS study, a registry that collected information worldwide on ATTR amyloidosis, which included Spanish patients [Citation14]. The prices of the drugs were taken from the Bot Plus database [Citation15]. As an example, the cost per patient of treatment with diuretics was calculated as follows: (i) on average, 83.8% of patients would receive diuretics; (ii) specifically, 81% of those treated with diuretics would be treated with furosemide (with a mean annual cost per patient of € 27.38), 11% with torasemide (€ 96.54/year), and 5% with thiazides such as hydrochlorothiazide (€ 42.89/year); (iii) the mean annual cost per patient of treatment with diuretics would be: (€ 27.38*81%) + (€ 96.54*11%) + (€ 42.89*5%) = € 35.25; (iv) in the overall pharmacological treatment computation, the one corresponding to diuretics would be: 83.8%*€ 35.25 = € 29.52 (). The cost of symptomatic treatment without the HF diagnosis (‘incorrect’ treatment) (€ 1,507.48 ± 153.82) was obtained from the Spanish study by Delgado et al [Citation16]. This is a multicentre, prospective and observational cost study with 12 months of follow-up conducted in a sample of 374 Spanish patients with HF.
2.5. Analyses performed
Patients with a correct diagnosis of ATTR-CM, the placebo group of the ATTR-ACT study, were compared with those undiagnosed. The study was conducted using probabilistic analyses, specifically second order Monte Carlo simulations, with the aim of analyzing the uncertainty of all the model variables [Citation17–19]. The probabilistic analyses conducted in our study included the probabilities (hospitalizations, death), adjusted for the beta distributions, and costs (diagnosis, pharmacological treatment, hospitalization), adjusted for the gamma distributions [Citation17,Citation18]. One thousand, second order Monte Carlo simulations were performed to analyze the uncertainty of all the indicated variables. The analysis results are presented from the healthcare perspective as the reduction in mortality and life-years gained (LYG) and from the economic perspective as savings per patient for the Spanish National Health System, resulting from correctly diagnosing ATTR-CM in comparison with not diagnosing the disease.
3. Results
3.1. Reduction in mortality and life-years gained in the diagnosed patient
The estimated reduction in ATTR-CM patient mortality associated with the correct disease diagnosis was 28.5%, 15.4%, 8.1%, and 3.9% at the end of 1, 5, 10, and 15 years, respectively ().
Table 3. Estimated reduction in mortality with and without diagnosis of ATTR-CM
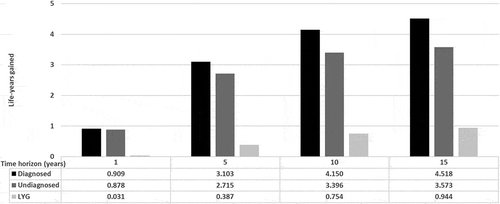
The LYGs in diagnosed patients versus undiagnosed patients are presented in . After 1, 5, 10, and 15 years, the correct diagnosis of ATTR-CM would result in a gain of 0.031 (95%CI 0.025, 0.038), 0.387 (95%CI: 0.329, 0.435), 0.754 (95%CI: 0.678, 0.781), and 0.944 (95%CI: 0.905, 0.983) life-years per patient, respectively, without taking into account the possibility of starting a specific treatment.
Figure 2. Life-years gained by a patient diagnosed with ATTR-CM receiving correct symptomatic treatment compared to the undiagnosed patient
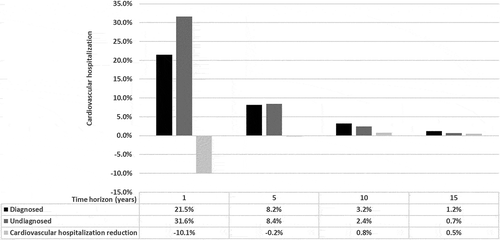
3.2. Hospitalizations avoided in diagnosed patients
As observed in , the correct diagnosis and symptomatic treatment entailed a 31.96%, 7.53%, 14.18%, 4.85%, and 2.38% reduction in the hospitalization rate for cardiovascular causes after 1, 2, 3, 4, and 5 years of follow-up, respectively. With a correct diagnosis for patients with ATTR-CM, an absolute reduction of 10.1% in hospitalizations for cardiovascular causes was obtained in the first year (). The decrease in the reduced hospitalization rate in years 4 and 5 was associated with a rapid increase in mortality in both of the compared groups, as observed in . In the first year, in a cohort of 1,000 patients, with a correct diagnosis and symptomatic treatment of the disease, 101 hospitalizations for cardiovascular causes would be avoided. Over a 5-year period, 139 hospitalizations would be avoided, generating a total savings in cardiovascular hospitalizations of € 640,976. The time horizon of the calculations was not extended, because most patients, without a specific treatment, die after 5 years () and since the differences in hospitalization for cardiovascular causes between the compared groups are insignificant starting in year 5.
3.3. Savings in diagnosed patients
As can be seen in , diagnosing patients with ATTR-CM, after 1, 5, 10, and 15 years, would result in a saving of € 212 (95%CI €-632, 633), € 2,289 (95%CI € 2,250, 2,517), € 2,859 (95%CI € 2,584, 3,149), and € 2,906 (95%CI € 2,669, 3,450) per patient, respectively, in comparison with not diagnosing the disease.
Table 4. Costs and savings generated per patient diagnosed with ATTR-CM who receives correct symptomatic treatment, compared to the undiagnosed patient. Detailed results
4. Discussion
This study points out the benefits and importance of correctly diagnosing a patient with HF if it is due to ATTR-CM, independently on any subsequent use of a specific treatment for the condition. In doing so, considering exclusively the resulting adjustment to symptomatic treatment, clinical and economic benefits would result, such as life-years gains, reduced number of hospitalizations, and reduced direct healthcare costs associated with the disease.
The delay in diagnosing ATTR-CM is a common problem in the real world of HF, with serious consequences for patients. In a study conducted in the United Kingdom [Citation20], in which 1,034 patients with ATTR-CM were studied between 2000 and 2017, a significant delay in diagnosing the disease was observed. The patients used hospital services on average 17 times during the 3 years prior to the diagnosis, with a deterioration in their quality of life during that period. In the specific case of patients with wild-type ATTR-CM, the diagnosis was delayed for more than 4 years after the presentation of the cardiac symptoms in 42% of the cases. This delayed diagnosis, as well as the elevated mortality of ATTR-CM, was also made clear in our country in the study by López-Sainz et al [Citation21].
Early diagnosis of ATTR-CM is crucial [Citation22]; first, because overall survival is very low once cardiac involvement is present [Citation23,Citation24] and second, because treatments are available that increase survival and reduce hospitalization rates [Citation8].
Other benefits of establishing a correct diagnosis of the disease include the possibility of receiving more accurate advice regarding the prognosis and possibility of diagnosing the genetic form of ATTR-CM, which may lead to an early diagnosis of family members carriers who will benefit from receiving reproductive and professional counseling and early follow-up for an earlier diagnosis.
One question that arises regarding the estimated LYGs in the study is whether these gains, which range between 0.031 and 0.944 LYGs, can be considered clinically significant. The clinical significance of life gained has mainly been studied in oncology (cancer patients have an average survival similar to that observed in patients with ATTR-CM), and a gain of 3 or more months has been deemed clinically significant according to the review by Salas et al. [Citation4] and the ESMO evaluation guidelines [Citation5]. Therefore, the life gain obtained by merely diagnosing the disease correctly in our analysis for a patient with ATTR-CM (4.6, 9.0, and 11.3 life-months gained at 5, 10, and 15 years) could be considered clinically significant.
To put these life-years gained into context, it is of interest to comment on a cost-effectiveness analysis of sacubitril/valsartan, in the context of patients with chronic HF with reduced ejection fraction [Citation25]. According to this study, considering a lifelong time horizon and a life expectancy between 6.2 and 6.7 years, with sacubitril/valsartan 0.50 years would be gained. In this analysis, a gain of 0.387 and 0.754 years are estimated for time horizons of 5 and 10 years. Therefore, the result obtained in our study is similar to the mentioned model, in very similar patients, with the difference being that in our analysis we are exclusively evaluating the effect of diagnosing and adjusting symptomatic treatment.
The economic evaluation performed has strengths and weaknesses. With regards to the strengths, it should be emphasized that the reliability of the economic analysis results is confirmed by 1,000 second order Monte Carlo simulations, which was used to analyze the uncertainty associated with the analyzed variables (transition probabilities and costs of the Markov states). This mathematical method makes it possible to reproduce the effect of simultaneous and random changes in those parameters, trying to simulate the clinical evolution in real life. The Monte Carlo simulation is a widely accepted and internationally recommended method [Citation17–19,Citation26]. Thus, the consistency of the results was confirmed in the 95% confidence intervals obtained in the probabilistic analysis (). It may be considered a strength of the model, the fact that the mortalities obtained at 1 and 5 years in diagnosed patients (15.8% and 60.3%, respectively) are similar to those described in a recently published Spanish retrospective study (14.9% and 68.6%, respectively) [Citation27], which are within the variability intervals considered by the model for this variable ().
As far as weaknesses are concerned, firstly, it should be taken into account that a theoretical model was used, which is, by definition, a simplified simulation of reality. Secondly, in the model it was necessary to make assumptions due to a lack of data in patients with ATTR-CM who have not been diagnosed. Finally, it was assumed that a correct diagnosis implies a correct treatment, which is not necessarily true in 100% of cases. These assumptions were clinically validated by the panel of experts.
By being focused on the effect of a correct diagnosis, our study does not take into account the expenses associated with starting a specific pharmacological therapy to treat the ATTR-CM. It also does not assess the possible additional savings derived from the beneficial effects of those treatments. The cost-effectiveness of treatment with tafamidis has recently been questioned in the U.S. due to the cost of the drug in that country [Citation28]. Nevertheless, the results of this economic study have also been questioned [Citation29], mainly because the analysis and cost-effectiveness thresholds are inappropriate for orphan drugs and due to the different methodological problems, such as inadequate extrapolation of overall survival used in the model. In any case, more studies will be needed in our country, once the price of tafamidis is known in our setting.
5. Conclusions
Early diagnosis of ATTR-CM and resulting correct symptomatic treatment of the disease produce a significant gain in life-years and cost savings, due to the avoided hospitalizations for cardiovascular causes, for the Spanish NHS.
Article highlights
ATTR-CM is an underdiagnosed disease with a clinical presentation similar to that of much more prevalent cardiovascular diseases, with serious consequences for patients, increasing the mortality and hospitalisations for cardiovascular causes.
Based on this model, the diagnosis of ATTR-CM and resulting correct symptomatic treatment of the disease produce a significant gain in life-years and cost savings for the National Health System, specifically due to the hospitalisations for cardiovascular causes that would be avoided.
Declaration of interest
Carlos Rubio-Terrés and Darío Rubio-Rodríguez are employees of Health Value and have received consultancy payments from Pfizer in connection with the conduct of this study and the development of the manuscript. CP, PT and AL work at Pfizer SLU. CP and PT own shares in Pfizer SLU. PGP has received fees for lectures and/or advisory activity from Akcea, Alnylam, Eidos, Neuroimmune, and Pfizer. The PGP center has received research and/or training funding from Akcea, Alnylam, Eidos, Pfizer, and Prothena. ANR, FF and FJMS have received advisory fees from Pfizer. The authors have no other relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript apart from those disclosed.
Reviewers disclosure
Peer reviewers on this manuscript have no relevant financial relationships or otherwise to disclose.
Additional information
Funding
References
- Garcia-Pavia P, Domínguez F, Gonzalez-Lopez E. Transthyretin amyloid cardiomyopathy. Med Clin (Barc). 2021;156:126–134.
- González-López E, López-Sainz A, García-Pavía P. Diagnóstico y tratamiento de la amiloidosis cardiaca por transtiretina. Progreso y esperanza. Rev Esp Cardiol. 2017;70:991–1004.
- Castaño A, Drachman BM, Judge D, et al. Natural history and therapy of TTR-cardiac amyloidosis: emerging disease-modifying therapies from organ transplantation to stabilizer and silencer drugs. Heart Fail Rev. 2015;20:163–178.
- Salas-Vega S, Iliopoulos O, Mossialos E. Assessment of overall survival, quality of life, and safety benefits associated with new cancer medicines. JAMA Oncol. 2017;3:382–390.
- Cherny NI, Sullivan R, Dafni U, et al. A standardised, generic, validated approach to stratify the magnitude of clinical benefit that can be anticipated from anti-cancer therapies: the European society for medical oncology magnitude of clinical benefit scale (ESMO-MCBS). Ann Oncol. 2015;26:1547–1573.
- Vyndaqel (tafamidis). Ficha técnica. [ cited 2020 Sept 22]. Available from: https://cima.aemps.es/cima/pdfs/es/ft/11717001/FT_11717001.pdf
- TreeAge pro healthcare. [ cited 2020 May 6]. Available from: https://www.treeage.com/shop/treeage-pro-healthcare/
- Maurer MS, Schwartz JH, Gundapaneni B, et al.; ATTR-ACT Study Investigators. Tafamidis treatment for patients with transthyretin amyloid cardiomyopathy. N Engl J Med. 2018;379:1007–1016.
- De Giuli F, Khaw KT, Cowie MR, et al. Incidence and outcome of persons with a clinical diagnosis of heart failure in a general practice population of 696.884 in the United Kingdom. Eur J Heart Fail. 2005;7:295–302.
- Sayago-Silva I, García-López F, Segovia-Cubero J. Epidemiology of heart failure in Spain over the last 20 years. Rev Esp Cardiol (Engl Ed). 2013;66:649–656.
- Solomon SD, Rizkala AR, Lefkowitz MP, et al. Baseline characteristics of patients with heart failure and preserved ejection fraction in the PARAGON-HF trial. Circ Heart Fail. 2018;11:e004962.
- Carrasco-Sánchez FJ, Páez-Rubio MI, García-Moreno JM, et al. Predictive variables for mortality in elderly patients hospitalized due to heart failure with preserved ejection fraction. Med Clin (Barc). 2013;141:423–429.
- Petitti DB. Meta-analysis, decisión análisis and cost-effectiveness analysis. Methods for quantitative synthesis in medicine. New York (NY): Oxford University Press; 1994.
- Damy T, Kristen AV, Suhr OB, et al. Transthyretin cardiac amyloidosis in continental Western Europe: an insight through the transthyretin amyloidosis outcomes survey (THAOS). Eur Heart J. 2019;ehz173. DOI:https://doi.org/10.1093/eurheartj/ehz173
- BotPlus. [ cited 2019 Dec 29]. Available from: https://botplusweb.portalfarma.com/
- Delgado JF, Oliva J, Llano M, et al. Costes sanitarios y no sanitarios de personas que padecen insuficiencia cardiaca crónica sintomática en España. Rev Esp Cardiol. 2014;67:643–650.
- Briggs A, Claxton K, Sculpher M. Decision modelling for health economic evaluation. Oxford: Oxford University Press; 2007.
- Gray AM, Clarke PM, Wolstenholme JL, et al. Applied methods of cost-effectiveness analysis in health care. Oxford: Oxford University Press; 2012.
- Rubio-Terrés C, Rubio-Rodríguez D. Probabilistic Analysis: sensitivity analysis or main result? (Editorial). Pharmacoeconomics. 2016;1:2.
- Lane T, Fontana M, Martinez-Naharro A, et al. Natural history, quality of life, and outcome in cardiac transthyretin amyloidosis. Circulation. 2019;140:16–26.
- López-Sainz A, Hernández-Hernández A, González-López E, et al. Perfil clínico y evolución de la amiloidosis cardiaca en un centro español de referencia. Rev Esp Cardiol. 2020. DOI:https://doi.org/10.1016/j.rec.2019.12.020
- Oerlemans MIFJ, Rutten KHG, Minnema MC, et al. Cardiac amyloidosis: the need for early diagnosis. Neth Heart J. 2019;27:525–536.
- Merlini G, Dispenzieri A, Sanchorawala V, et al. Systemic immunoglobulin light chain amyloidosis. Nat Rev Dis Prim IEEE Trans Med Imaging. 2018;4:38.
- Falk RH, Alexander KM, Liao R, et al. AL (light-chain) cardiac amyloidosis: a review of diagnosis and therapy. J Am Coll Cardiol. 2016;68:1323–1341.
- Ademi Z, Pfeil AM, Hancock E, et al. Cost-effectiveness of sacubitril/valsartan in chronic heart-failure patients with reduced ejection fraction. Swiss Med Wkly. 2017;147:w14533.
- Isla D, De Castro J, Juan O, et al. Costs of adverse events associated with erlotinib or afatinib in first-line treatment of advanced EGFR-positive non-small cell lung cancer. Clinicoecon Outcomes Res. 2016;9:31–38.
- Barge-Caballero G, Vázquez-García R, Barge-Caballero E, et al. Amiloidosis cardíaca por cadenas ligeras y por transtirretina: características clínicas, historia natural y predictores pronósticos. Med Clín (Barc). 2021. DOI:https://doi.org/10.1016/j.medcli.2020.04.031
- Kazi DS, Bellows BK, Baron SJ, et al. Cost-effectiveness of tafamidis therapy for transthyretin amyloid cardiomyopathy. Circulation. 2020;141:1214–1224.
- Rozenbaum MH, Kemner J, Parasuraman B. Letter by rozenbaum et al regarding article, “cost-effectiveness of tafamidis therapy for transthyretin amyloid cardiomyopathy”. Circulation. 2020;142:e210–e211.