ABSTRACT
Observations of diversity in alpine vegetation appear to be scale dependent. The relations of plant species richness with surface processes and geomorphology have been studied, but patterns of beta diversity are less known. In Glacier National Park, Montana, diversity has been examined within 1 m2 plots and for 16 m2 plots across two ranges, with within-plot and across-range explanatory factors, respectively. The slopes of species–area equations for nested 4, 8, 12, and 16 m2 plots were used as an indicator of beta diversity in Glacier National Park, where smaller and larger scales have been examined. The slopes were negatively related to a field assessment of surface stability and positively to the presence of talus—two sides of the same coin. A positive relationship with bedrock outcrops may be due to a misrepresentation of area for plants. The relationship of species–area slopes to plot-level gamma diversity was negative, weak, and marginally significant, and this variable did not enter the general linear model (GLM). Beyond simple differences in diversity with differences in environment, examination of beta diversity at a scale between that of earlier studies revealed surface processes and geomorphology as drivers that were also at a scale between those previously reported.
Introduction
Alpine plant communities are floristically diverse at multiple scales. In alpine plant communities, more functional types can be found in the same area given steep environmental gradients and high environmental heterogeneity, and alpine plants are small and thus more individuals and more species can fit per unit area (Körner Citation2003). These factors are scale dependent. Therefore, alpine plant community composition and diversity vary across and within regions and mountain ranges, and how the diversity of this habitat varies with spatial scale could inform fundamental theory in biogeography. In addition, understanding the underlying determinants of the diversity of alpine vegetation will help anticipate the effects of climate change (Amagai, Kudo, and Sato Citation2018; Malanson et al. Citation2019). Although increases in diversity in Europe have been reported (e.g., Pauli et al. Citation2012), species are mostly shifting their distributions upward, with an acceleration of such range shifts (e.g., Steinbauer et al. Citation2018). Thus, local diversity may be reduced by climate warming in the future (e.g., Dirnbock, Dullinger, and Grabherr Citation2003; Klanderud and Totland Citation2005; Engler et al. Citation2011). In the United States, Diaz and Eischeid (Citation2007) reported that the alpine type of climate probably will not exist in the American West in the near future. This change would endanger many species, although not necessarily eliminate all areas of alpine vegetation. A complicating factor is the multiscale heterogeneity caused by geomorphology (e.g., Johnson and Billings Citation1962; Malanson, Bengtson, and Fagre Citation2012; le Roux, Virtanen, and Luoto Citation2013; le Roux and Luoto Citation2014). We aim to fill a gap in the scales at which plant species diversity has been analyzed in the alpine vegetation of the Rocky Mountains, United States.
McGill (Citation2010) argued that a greater understanding of scale dependence was needed to explain biogeographic patterns. For species diversity, scale is foundational. In emphasizing the species–area relationship as perhaps the strongest biogeographic principle, Rosenzweig (Citation1995) focused on change over space, the slope of the species–area equation. Chase et al. (Citation2018) recently developed Rosenzweig’s argument that more useful information could be derived from the species–area equation per se than from analysis of alpha diversity alone. The slope of the species–area equation captures the beta diversity of the observed scale. Other measures of beta diversity are common, however, and may also be informative (Tuomisto Citation2010).
Studies in alpine diversity can be difficult to generalize because of the differences in their geographic and historical settings and resulting flora. Carlson et al. (Citation2013) noted the scale dependence of the determinants of alpine plant distributions, and Meier and Hofer (Citation2016) reported effects of plot size on observations of alpine diversity. Bhatta, Grytnes, and Vetaas (Citation2018) reported that the effects of regional gamma diversity did not differ with the scale of nested quadrats on an elevation gradient in the Himalaya. A number of studies point to topographically determined environmental heterogeneity as an important factor at multiple scales. At about 1 m scale, microtopography affects temperature (Scherrer and Körner Citation2011) and soil water (Litaor, Williams, and Seastedt Citation2008) and can indicate disturbance (Bruun et al. Citation2006). At this scale, species interactions and species characteristics, such as spread by cloning, will be important (Choler, Michalet, and Callaway Citation2001). At 10 to 100 m scale, factors such as roughness and slope aspect, angle, and shape (and their effect on snow and soil moisture) play a role (Randin et al. Citation2009; Malanson, Bengtson, and Fagre Citation2012; Jiménez‐Alfaro et al. Citation2014; Takahashi and Murayama Citation2014; Opedal, Armbruster, and Graae Citation2015; Wu et al. Citation2018). Across mountain ranges, variation in alpha and beta diversity again reflects general topography (Wehn, Lundemo, and Holten Citation2014) but in relation to broader factors such as synoptic climatology (Winkler et al. Citation2016). At yet larger scales, patterns of climate and evolutionary biogeography are probable factors but are not yet well described (Anthelme et al. Citation2014; Malanson, Cheney, and Kinney Citation2015).
We focused on a local area for which we have a broader context. Rose and Malanson (Citation2012) examined species richness within 1 m2 plots in Glacier National Park (GNP), Montana. They found that microtopographic heterogeneity constrained rather than supported species richness. For the richness sampled in 16 m2 plots across GNP, Malanson, Fagre, and Zimmerman (Citation2018) reported that low diversity occurred only where precipitation was highest. However, those results were set within the context of similar size plots across the Rocky Mountains, and at that scale temperature was the determining factor. Here, we examined diversity at a scale between those two scales—that is, within the 16 m2 plots—by focusing on species–area equations. Our aim was to uncover the environmental factors affecting diversity at this in-between scale. We can interpret not only the specific results of an improved quantification but also how factors differ from other scales in the same vegetation.
Additional context for GNP indicates that this location has the environmental variability that could produce a wide range of species–area slopes. The plots in GNP were also part of a larger study that included similar plots in the Beartooth Range, Wyoming, the Indian Peaks Area, Colorado, and Taos, New Mexico (Malanson et al. Citation2017). Comparisons of the four regions are reported in Appendix A. The species–area slopes for GNP were higher than those for the other three locations (but not significantly from the Beartooth). The continuous environmental measures differed in some respects, but GNP was never the most extreme. GNP did tend to be more variable, however, with the highest coefficient of variation for soil depth and for the standard deviation of soil depth. In addition, the stratified sample design for the four regions was based on preliminary analyses in GNP, where the number of species in ninety-six plots exceeded the number reported by Damm (Citation2001) for a thorough coverage of the alpine with 525 releves. The total regional diversity was also relatively high, possibly because of its biogeographic position, with species from both the southern cordillera and the arctic floras (Harris Citation2002; Lesica and McCune Citation2004). Thus, GNP provided the best data to examine the question of beta diversity at the in-between scale.
Methods
We reexamined the data used by Malanson, Fagre, and Zimmerman (Citation2018): species identified in ninety-six 16 m2 quadrats distributed across the Lewis and Livingston Ranges in GNP. The original 16 m2 plots were recorded in four 2 × 2 m subplots. We used the species–area procedure in PC-ORD ver. 6 to compute the average number of species per single subplot, every pair of subplots, and every triplet of subplots; the total number per plot provided a fourth point. This approach constitutes an average nesting rather than a true one. We calculated the species–area equation for these four points with the usual log–log transformation from S = cAz to log S = log c + z log A (where S is species richness, A is area, and c and z are parameters). We used the slopes of these equations, z, as our initial dependent variable. Slope indicates the rate of increase in diversity among the four subplots and is an indicator of beta diversity at this scale. For comparison, we also calculated the simplest original indicator of beta diversity (Whittaker Citation1960), the total species per plot, or plot gamma diversity, divided by the average of the four subplots.
Following Damm (Citation2001), for each plot Malanson, Fagre, and Zimmerman (Citation2018) recorded the elevation, slope angle, and slope aspect, and they measured an indicator of soil depth at the center and four corners of the plot and calculated its mean and standard deviation. For indicators of surface condition and microtopography they recorded the following characteristics or conditions for each quadrat:
Slope relief: smooth, rolling, outcrop, convex, concave, irregular, terrace, other
Microclimate/exposure: cold air drainage, upslope warm air, wind blasted, snow catchment
Position: upper, middle, low or foot of a slope
Topography: ridge, saddle, valley bottom, slope, summit, other
Soil surface: stable–well vegetated, stable–poorly vegetated, unstable
Special features: talus, scree, wet meadow, swale, seep, avalanche, open water.
They used these binary observations in categorical principal components analysis to produce two vectors for each of the six categories and an additional two for all combined; here, we used the binary variables directly after removing those for which fewer than five plots were recorded. For soil surface we collapse the categories into stable or unstable; our assessment was based on walking on the surface, not a visual estimate.
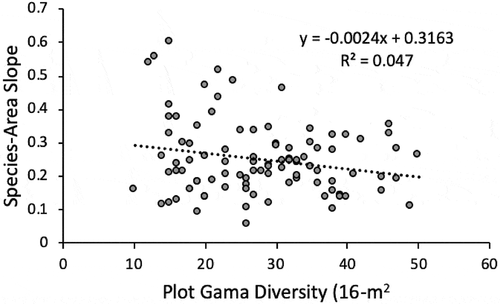
We first computed correlations among the two indicators of beta diversity, species–area slope and Whittaker’s beta; the intercept of the species–area equations; and the total species richness of the 16 m2 plots (plot gamma diversity). For the species–area slope, in Minitab ver. 19 we derived a general linear model (GLM) using a stepwise procedure with the categorical variables as factors and the continuous variables as covariates. We used GLM because of the mix of continuous and categorical variables. The stepwise procedure enters and removes variables at each step depending on p value, which we set at .05 to enter and .10 to remove. For the significant variables in the GLM, we used commonality analysis (Ray-Mukherjee et al. Citation2014) to partition the variance. Although it did not appear as a predictor in the model, we also examined the relationship between slope and plot gamma diversity.
Results
The descriptive statistics for the variables and correlations among species–area slope, Whittaker’s beta, plot gamma and alpha diversities, and the slope intercepts are shown in Appendix B. Because of a high correlation between the slopes and Whittaker’s beta, the intercept and alpha and gamma, and alpha and gamma we thereafter analyzed only species–area slope and gamma diversity. The species–area slopes ranged from 0.057 to 0.604, and their R2 ranged from 0.923 to 0.999.
The GLM and commonality analysis revealed that the independent variables explained only a modest proportion of the variance in beta diversity at this scale as represented by species–area slope ( and ). The surface stability indicator alone accounted for <15 percent of the variance in species area slope; talus added <10 percent and bedrock outcrops 3.25 percent. Shared effects of talus and outcrops with stability were <3 percent each, and their combination was <0.0, as was the interaction of all three variables (commonality analysis reveals negative contributions to explained variance). Species–area slope was negatively related to surface stability and the presence of talus, whereas it was positively related to the presence of outcrops. Although it was not significant in the GLM, when species–area slopes were regressed on plot gamma diversity (16 m2), the weak negative relationship was signficant (R2 = 0.047; p = .035; ).
Table 1. GLM results for the dependent species–area slope with the independent site variables. The contribution is the percentage that each source in the analysis of variance table contributes to the total sequential sums of squares
Table 2. Commonality analysis, which partitions the R2 in simple and multiple regressions, for the three independent variables that were significant in the GLM
Discussion
The species–area slopes are useful metrics of diversity at a small scale. Although much of the information is in the species richness of the plots, these slopes capture the internal heterogeneity of the plots and represent their beta diversity. The slopes are related to factors other than plot gamma diversity per se. The analysis of the other variables may help separate beta diversity from gamma diversity (cf. Tuomisto Citation2010; Ulrich et al. Citation2017). This analysis should perhaps complement those of Chase et al. (Citation2018).
The site-specific conditions that affected the species heterogeneity of plots were related to geomorphic heterogeneity at this scale. Of the potential explanatory factors, aspects of slope stability had larger effects. Surface stability, from an indicator based on field assessment, led to lower beta diversity. A moving surface creates diffferences in the subplots that account for this heterogeneity. Beta diversity is also higher with more talus, another indicator of slope instability. The influence of outcrops is an aspect of topography that differs from others. In subplots with an outcrop, the area for plants is actually less, and the steeper species–area slopes may misrepresent the relationship. A subquadrat with an outcrop has less inhabitable area (we did not include lichen in this study), and the area used as a basis for the analysis might not be meaningful.
Bhatta, Grytnes, and Vetaas (Citation2018) reported a gradient in diversity with elevation, but they were looking at patterns for broad gamma diversity among scales instead of environmental effects within a scale. Here, elevation had a small indirect effect on diversity. At the highest elevations, fewer species coexist in any case because of the more extreme conditions of exposure, more likely dry sites, and less soil. Though these factors did not individually affect diversity across their ranges, their combination at the highest elevations is a possible explanation; elevation can also represent spatiotemporal disequilibria (e.g., Roland and Schmidt Citation2015).
Rose and Malanson (Citation2012) analyzed diversity in 1 m2 plots distributed across GNP, and Malanson, Fagre, and Zimmerman (Citation2018) used 16 m2 plots similarly but not identically distributed. Here, we examined diversity in four 4 m2 plots comprising those 16 m2 plots. Analysis of the smaller plots revealed that diversity was lower where microtopographic variability was greatest, possibly because it increased the nonvegetated surface area. Analysis of the larger plots found that diversity was related to a precipitation gradient across GNP. This precipitation gradient is, however, also related to topography but at the scale of the major ranges that make up GNP: The climate of the northwestern portion of GNP is more directly affected by the Pacific Ocean, with a more maritime climate. The east side of GNP is in a rainshadow and is dominated by a more continental climate. Thus, topography at the scale of the mountain range may be a factor that differentiates diversity across GNP. The slope of the species–area equations based on the 4 m2 subplots within the 16 m2 plots reveals an intermediate scale of geomorphic patterns that control diversity. Where nonvegetated surface area reduced diversity in the 1 m2 plots, the presence of outcrops increased beta diversity within the 16 m2 plots, and it was further affected by mesoscale topography and slope instability.
That beta diversity varies with the environment is trivial, but identifying the specifics of scale dependence is a challenge (Carlson et al. Citation2013; Suding et al. Citation2016; Malanson, Fagre, and Zimmerman Citation2018). The factors that matter to ecological patterns must be those that affect processes at the individual plant level, but studies in alpine vegetation display scale dependence because of the multiple scales at which processes vary and interact. Spatial heterogeneity in environments creates complex intersections of these factors. These will include broad environmental patterns, such as continental gradients in precipitation (Malanson, Cheney, and Kinney Citation2015), but it is likely that the factors that matter, other than general temperature or precipitation, will be those that are scaled to modify those two drivers or their effects on individual plants (Carlson et al. Citation2013). Geomorphic and topographic features are likely to matter at this scale (e.g., Bruun et al. Citation2006; Litaor, Williams, and Seastedt Citation2008; Scherrer and Körner Citation2011; Malanson, Bengtson, and Fagre Citation2012; Rose and Malanson Citation2012; le Roux, Virtanen, and Luoto Citation2013; Jiménez‐Alfaro et al. Citation2014; Takahashi and Murayama Citation2014; Wehn, Lundemo, and Holten Citation2014; Winkler et al. Citation2016; Wu et al. Citation2018).
In this study, processes and patterns at a scale within slopes affected the within-plot heterogeneity, a scale-specific measure of beta diversity. Surface processes that led to nonvegetated area, notably talus, complemented the presence of bedrock outcrops. Together they produced low alpha diversity in some subquadrats, which in turn resulted in steeper species–area slopes. Although its importance was documented by Johnson and Billings (Citation1962), further study of the relations of surficial geomorphic processes and the diversity and structure of alpine vegetation is needed (cf. le Roux and Luoto Citation2014). These patterns can change with climate because freeze–thaw proceses are important drivers (Benedict Citation1970), and how they create the heterogeneity on which alpine diversity depends should be better understood.
Supplemental Material
Download Zip (917.5 KB)Supplemental data
Supplemental data for this paper can be accessed on the publisher’s website.
Disclosure statement
No potential conflict of interest was reported by the authors.
Additional information
Funding
References
- Amagai, Y., G. Kudo, and K. Sato. 2018. Changes in alpine plant communities under climate change: Dynamics of snow‐meadow vegetation in northern Japan over the last 40 years. Applied Vegetation Science 21:561–71.
- Anthelme, F., D. Jacobsen, P. Macek, R. I. Meneses, P. Moret, S. Beck, and O. Dangles. 2014. Biodiversity patterns and continental insularity in the tropical High Andes. Arctic, Antarctic, and Alpine Research 46:811–28. doi:https://doi.org/10.1657/1938-4246-46.4.811.
- Benedict, J. B. 1970. Downslope soil movement in a Colorado alpine region: Rates, processes, and climatic significance. Arctic and Alpine Research 2:165–226. doi:https://doi.org/10.1080/00040851.1970.12003576.
- Bhatta, K. P., J. A. Grytnes, and O. P. Vetaas. 2018. Scale sensitivity of the relationship between alpha and gamma diversity along an alpine elevation gradient in central Nepal. Journal of Biogeography 45:804–14. doi:https://doi.org/10.1111/jbi.13188.
- Bruun, H. H., J. Moen, R. Virtanen, J. A. Grytnes, L. Oksanen, and A. Angerbjörn. 2006. Effects of altitude and topography on species richness of vascular plants, bryophytes and lichens in alpine communities. Journal of Vegetation Science 17:37–46. doi:https://doi.org/10.1111/j.1654-1103.2006.tb02421.x.
- Carlson, B. Z., C. F. Randin, I. Boulangeat, S. Lavergne, W. Thuiller, and P. Choler. 2013. Working toward integrated models of alpine plant distribution. Alpine Botany 123:41–53. doi:https://doi.org/10.1007/s00035-013-0117-4.
- Chase, J. M., B. J. McGill, D. J. McGlinn, F. May, S. A. Blowes, X. Xiao, T. M. Knight, O. Purschke, and N. J. Gotelli. 2018. Embracing scale‐dependence to achieve a deeper understanding of biodiversity and its change across communities. Ecology Letters 21:1737–51. doi:https://doi.org/10.1111/ele.13151.
- Choler, P., R. Michalet, and R. M. Callaway. 2001. Facilitation and competition on gradients in alpine plant communities. Ecology 82:3295–308. doi:https://doi.org/10.1890/0012-9658(2001)082[3295:FACOGI]2.0.CO;2.
- Damm, C. 2001. A phytosociological study of Glacier National Park, Montana, USA, with notes on the syntaxonomy of alpine vegetation in western North America ( Dissertation), Georg-August Universitaet, Goettingen, FRG.
- Diaz, H. F., and J. K. Eischeid. 2007. Disappearing alpine tundra Koppen climatic type in the western United States. Geophysical Research Letters 34 (L18707):1–4. doi:https://doi.org/10.1029/2007GL031253.
- Dirnbock, T., S. Dullinger, and G. Grabherr. 2003. A regional impact assessment of climate and land-use change on alpine vegetation. Journal of Biogeography 30:401–17. doi:https://doi.org/10.1046/j.1365-2699.2003.00839.x.
- Engler, R., C. F. Randin, W. Thuiller, S. Dullinger, N. E. Zimmermann, M. B. Araújo, P. B. Pearman, G. Le Lay, C. Piedallu, C. H. Albert, et al. 2011. 21st century climate change threatens mountain flora unequally across Europe. Global Change Biology 17:2330–41. doi:https://doi.org/10.1111/j.1365-2486.2010.02393.x.
- Harris, S. A. 2002. Biodiversity of the vascular timberline flora in the Rocky mountains of Alberta Canada. In Mountain biodiversity: A global assessment, ed. C. Körner and E. M. Spehn, 49–57. New York: Parthenon.
- Jiménez‐Alfaro, B., C. Marcenó, Á. Bueno, R. Gavilán, and J. R. Obeso. 2014. Biogeographic deconstruction of alpine plant communities along altitudinal and topographic gradients. Journal of Vegetation Science 25:160–71. doi:https://doi.org/10.1111/jvs.12060.
- Johnson, P. L., and W. D. Billings. 1962. The alpine vegetation of the Beartooth Plateau in relation to cryopedogenic processes and patterns. Ecological Monographs 32:105–35. doi:https://doi.org/10.2307/1942382.
- Klanderud, K., and O. Totland. 2005. Simulated climate change altered dominance hierarchies and diversity of an alpine biodiversity hotspot. Ecology 86:2047–54. doi:https://doi.org/10.1890/04-1563.
- Komarkova, V. 1979. Alpine vegetation of the Indian Peaks Area. Vaduz, Liechtenstein: J Cramer.
- Körner, C. 2003. Alpine plant life. Berlin: Springer.
- le Roux, P. C., and M. Luoto. 2014. Earth surface processes drive the richness, composition and occurrence of plant species in an arctic-alpine environment. Journal of Vegetation Science 25:45–54. doi:https://doi.org/10.1111/jvs.12059.
- le Roux, P. C., R. Virtanen, and M. Luoto. 2013. Geomorphological disturbance is necessary for predicting fine-scale species distributions. Ecography 36:800–08. doi:https://doi.org/10.1111/j.1600-0587.2012.07922.x.
- Lesica, P., and B. McCune. 2004. Decline of arctic‐alpine plants at the southern margin of their range following a decade of climatic warming. Journal of Vegetation Science 15:679–90. doi:https://doi.org/10.1111/jvs.2004.15.issue-5.
- Litaor, M. I., M. Williams, and T. R. Seastedt. 2008. Topographic controls on snow distribution, soil moisture, and species diversity of herbaceous alpine vegetation, Niwot Ridge, Colorado. Journal of Geophysical Research: Biogeosciences 113:G02008. doi:https://doi.org/10.1029/2007JG000419.
- Malanson, G. P., A. B. Cheney, and M. Kinney. 2015. Climatic and geographic relations of alpine tundra floras in western North America. Alpine Botany 125:21–29. doi:https://doi.org/10.1007/s00035-014-0144-9.
- Malanson, G. P., D. B. Fagre, and D. L. Zimmerman. 2018. Scale dependence of diversity in alpine tundra, Rocky Mountains, USA. Plant Ecology 219:999–1008. doi:https://doi.org/10.1007/s11258-018-0852-0.
- Malanson, G. P., D. L. Zimmerman, M. Kinney, and D. B. Fagre. 2017. Relations of alpine plant communities across environmental gradients: Multilevel versus multiscale analyses. Annals of the American Association of Geographers 107:41–53. doi:https://doi.org/10.1080/24694452.2016.1218267.
- Malanson, G. P., L. E. Bengtson, and D. B. Fagre. 2012. Geomorphic determinants of species composition of alpine tundra, Glacier National Park, USA. Arctic, Antarctic, and Alpine Research 44:197–209. doi:https://doi.org/10.1657/1938-4246-44.2.197.
- Malanson, G. P., L. M. Resler, D. R. Butler, and D. B. Fagre. 2019. Mountain plant communities: Uncertain sentinels? Progress in Physical Geography 43:521–43. doi:https://doi.org/10.1177/0309133319843873.
- McGill, B. J. 2010. Scale matters. Science 328:575–76. doi:https://doi.org/10.1126/science.1188528.
- Meier, E. S., and G. Hofer. 2016. Effects of plot size and their spatial arrangement on estimates of alpha, beta and gamma diversity of plants in alpine grassland. Alpine Botany 126:167–76. doi:https://doi.org/10.1007/s00035-016-0171-9.
- Opedal, Ø. H., W. S. Armbruster, and B. J. Graae. 2015. Linking small-scale topography with microclimate, plant species diversity and intra-specific trait variation in an alpine landscape. Plant Ecology & Diversity 8:305–15. doi:https://doi.org/10.1080/17550874.2014.987330.
- Pauli, H., M. Gottfried, S. Dullinger, O. Abdaladze, M. Akhalkatsi, J. L. B. Alonso, G. Coldea, J. Dick, B. Erschbamer, R. F. Calzado, et al. 2012. Recent plant diversity changes on Europe’s mountain summits. Science 336:353–55. doi:https://doi.org/10.1126/science.1219033.
- Randin, C. F., G. Vuissoz, G. E. Liston, P. Vittoz, and A. Guisan. 2009. Introduction of snow and geomorphic disturbance variables into predictive models of alpine plant distribution in the Western Swiss Alps. Arctic, Antarctic, and Alpine Research 41:347–61. doi:https://doi.org/10.1657/1938-4246-41.3.347.
- Ray‐Mukherjee, J., K. Nimon, S. Mukherjee, D. W. Morris, R. Slotow, and M. Hamer. 2014. Using commonality analysis in multiple regressions: A tool to decompose regression effects in the face of multicollinearity. Methods in Ecology and Evolution 5:320–328. doi:https://doi.org/10.1111/2041-210X.12166.
- Roland, C. A., and J. H. Schmidt. 2015. A diverse alpine species pool drives a ‘reversed’ plant species richness-elevation relationship in interior Alaska. Journal of Biogeography 42:738–50. doi:https://doi.org/10.1111/jbi.12446.
- Rose, J. P., and G. P. Malanson. 2012. Microtopographic heterogeneity constrains alpine plant diversity, Glacier National Park, MT. Plant Ecology 213:955–65. doi:https://doi.org/10.1007/s11258-012-0056-y.
- Rosenzweig, M. L. 1995. Species diversity in space and time. Cambridge: Cambridge University Press.
- Scherrer, D., and C. Körner. 2011. Topographically controlled thermal‐habitat differentiation buffers alpine plant diversity against climate warming. Journal of Biogeography 38:406–16. doi:https://doi.org/10.1111/jbi.2011.38.issue-2.
- Steinbauer, M. J., J. A. Grytnes, G. Jurasinski, A. Kulonen, J. Lenoir, H. Pauli, C. Rixen, M. Winkler, M. Bardy-Durchhalter, E. Barni, et al. 2018. Accelerated increase in plant species richness on mountain summits is linked to warming. Nature 556:231–34. doi:https://doi.org/10.1038/s41586-018-0005-6.
- Suding, K. N., E. C. Farrer, A. J. King, L. Kueppers, and M. J. Spasojevic. 2016. Vegetation change at high elevation: Scale dependence and interactive effects on Niwot Ridge. Plant Ecology and Diversity 8:713–25. doi:https://doi.org/10.1080/17550874.2015.1010189.
- Takahashi, K., and Y. Murayama. 2014. Effects of topographic and edaphic conditions on alpine plant species distribution along a slope gradient on Mount Norikura, central Japan. Ecological Research 29:823–33. doi:https://doi.org/10.1007/s11284-014-1168-8.
- Tuomisto, H. 2010. A diversity of beta diversities: Straightening up a concept gone awry. Part 1. Defining beta diversity as a function of alpha and gamma diversity. Ecography 33:2–22. doi:https://doi.org/10.1111/eco.2010.33.issue-1.
- Ulrich, W., A. Baselga, B. Kusumoto, T. Shiono, H. Tuomisto, and Y. Kubota. 2017. The tangled link between beta- and gamma-diversity: A Narcissus effect weakens statistical inferences in null model analyses of diversity patterns. Global Ecology and Biogeography 26:1–5. doi:https://doi.org/10.1111/geb.12527.
- Wehn, S., S. Lundemo, and J. I. Holten. 2014. Alpine vegetation along multiple environmental gradients and possible consequences of climate change. Alpine Botany 124:155–64. doi:https://doi.org/10.1007/s00035-014-0136-9.
- Whittaker, R. H. 1960. Vegetation of the Siskiyou Mountains, Oregon and California. Ecological Monographs 30:279–338. doi:https://doi.org/10.2307/1943563.
- Winkler, M., A. Lamprecht, K. Steinbauer, K. Hülber, J. P. Theurillat, F. Breiner, P. Choler, S. Ertl, A. Gutiérrez Girón, G. Rossi, et al. 2016. The rich sides of mountain summits–A pan‐European view on aspect preferences of alpine plants. Journal of Biogeography 43:2261–73. doi:https://doi.org/10.1111/jbi.12835.
- Wu, M., H. He, S. Zong, X. Tan, H. Du, D. Zhao, K. Liu, and Y. Liang. 2018. Topographic controls on vegetation changes in alpine tundra of the Changbai Mountains. Forests 9 (12):756. doi:https://doi.org/10.3390/f9120756.