G-protein coupled receptor (GPCR) signaling contributes to normal cell functions including proliferation, survival and differentiation, as well as migration and homing. These signaling pathways can however be used also by cancer cells through aberrant expression of GPCRs and/or their ligands to promote tumor growth, metastatic spread and resistance to therapy by maintaining malignant cells in protective tissue niches.
The endocannabinoid system consists of 2 GPCRs, cannabinoid receptor 1 and 2 (CB1 and CB2), endogenous cannabinoids (endocannabinoids) and the enzymes participating in the synthesis and metabolism of endocannabinoids. CB1 is predominantly expressed in the central nervous system and regulates synaptic signaling. CB1 is also expressed at lower levels in other tissues including leukocytes under certain circumstances (reviewed inCitation1). However, the main cannabinoid receptor in immune cells is CB2, which regulates immune responses, development of B cells and localization of leukocytes in spleen.Citation2,3 The endocannabinoid system is dysregulated in many types of cancer and is involved in the regulation of survival and proliferation of cancer cells and cancer stem cells, in cancer metabolism, as well as in pro-metastatic events such as angiogenesis, migration and invasion.Citation4
Mantle cell lymphoma (MCL) is a non-curable B cell lymphoma that in several independent studies have been shown to express higher levels of CB1 and CB2 than non-malignant B cells (reviewed in Ref.Citation1). Previous in vitro studies of MCL cell lines and primary ex vivo isolated tumor cells have demonstrated that high (supraphysiological, μM) concentrations of cannabinoid receptor ligands induced proliferation arrest and programmed cell death. Furthermore, a synthetic cannabinoid receptor agonist decreased MCL tumor burden in a mouse xenograft model (reviewed in Ref.Citation1).
In a recent study, Wasik et al. correlated the mRNA expression of CB1, CB2 and the enzymes regulating levels, duration and magnitude of the signaling of one of the main endocannabinoids, anandamide, to clinical and pathological features. Mapping the expression pattern of the rate limiting enzymes participating in the anandamide synthesis (N-acyl phosphatidylethanolamine phospholipase D, NAPE-PLD) and anandamide metabolism (fatty acid amide hydrolyse, FAAH) and cannabinoid receptors (CB1 and CB2) in a large population based cohort of MCL showed that NAPE-PLD mRNA was highly expressed in MCL while FAAH mRNA was underexpressed as compared to normal B cells. Thus, these findings suggested accumulation of the polyunsaturated lipid anandamide in the MCL. CB1 was highly expressed in most, but not all, cases and CB2 was overexpressed in all MCL compared to non-malignant tissue cells. These findings imply an increased cannabinoid receptor signaling in MCL Citation5 (). Furthermore, the dissemination of tumor cells to the blood seemed to be negatively regulated by the endocannabinoid system since leukemic cases were characterized by low CB1 expression and high capacity to metabolize anandamide. Although the study by Wasik et al. did not investigate how CB1 signaling mediates MCL tissue retention, there are several other reports showing that CB1 can regulate adhesive/migratory properties of leukocytes.Citation6,7
Figure 1. A scheme depicting the perturbed anandamide pathway in mantle cell lymphoma. Red arrows show altered expression pattern in MCL, blue arrows show proposed effects. Altered expression of NAPE-PLD, an enzyme participating in anandamide synthesis from phospholipids, and FAAH, degrading anandamide, suggest accumulation of anandamide and together with overexpressed cannabinoid receptors, enhanced endocannabinoid signaling.
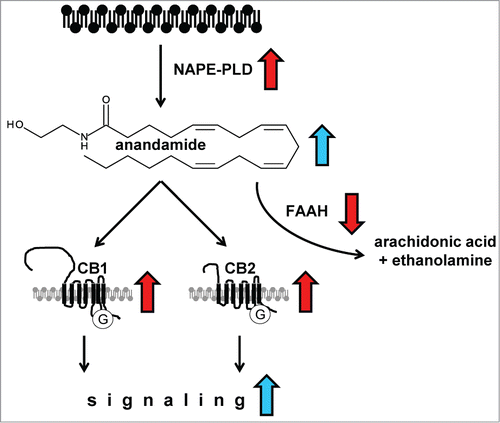
Thus, it can be concluded that the endocannabinoid system is perturbed towards enhanced anandamide signaling in MCL, suggesting a pro-oncogenic role. It therefore seems paradoxical that cannabinoids were reported to reduce proliferation in xenograft models and even induce cell death during in vitro culturing of primary tumor cells and MCL cell lines. The likely explanation for these apparently discrepant results is that the optimal concentration for inducing cell death in experimental settings is 1000-fold higher than the physiological concentration (mid nM) of endocannabinoids in vivo. All together, the data suggest that perturbations in the endocannabinoid system participate in the regulation of multi-functional cell responses regarding proliferation, migration and cell death control. Therefore, it can be concluded that further studies on pharmacological modulation of endocannabinoid accumulation and/or signaling offers an interesting option for novel anti-lymphoma therapy.
References
- Wasik AM, et al. Semin Cancer Biol 2011; 21:313-21; PMID:22024769; http://dx.doi.org/10.1016/j.semcancer.2011.10.004
- Muppidi JR, et al. J Exp Med 2011; 208:1941-8; PMID:21875957; http://dx.doi.org/10.1084/jem.20111083
- Pereira JP, et al. Nat Immunol 2009; 10:403-11; PMID:19252491; http://dx.doi.org/10.1038/ni.1710
- Chakravarti B, et al. Oncotarget 2014; 5:5852-72; PMID:25115386
- Wasik AM, et al. Oncoscience 2014; 1:550-7.
- Kianian M, et al. Clin Hemorheol Microcirc 2014; 58(2):333-42; http://dx.doi.org/10.3233/CH-131668
- Rossi B, et al. J Neuroimmunol 2011; 233:97-105; PMID:21216016; http://dx.doi.org/10.1016/j.jneuroim.2010.12.005
