The chromatin revolution in the past 2 decades has established the profound impacts of chromatin and epigenetic regulations in biology and medicine. There are 2 major mechanisms of chromatin modification: histone posttranslational modification and ATP-dependent chromatin remodeling. Interestingly, although histone-modifying complexes such as histone acetyltransferase (HAT) and histone methyltransferase complexes (HMT) are known to modify histones, non-histone substrates such as p53 have also been identified for these complexes. However, for ATP-dependent chromatin remodeling complexes such as SWI/SNF, the research to date is only focused on chromatin substrates (DNA, histones and nucleosomes). A recently published study revealed that the SWI/SNF chromatin remodeling complex directly interacts with and activates a key checkpoint kinase Mec1 (ATR in mammals) in an ATP-dependent fashion, suggesting that ATP-dependent chromatin remodeling complexes such as SWI/SNF can regulate the biochemical activities of the non-chromatin substrates.Citation1
Mec1 and Tel1 are phosphoinositide-3-kinase-related protein kinases and are called ATR and ATM, respectively, in mammals. Mutations in ATM/ATR cause genome instability, resulting in diseases such as Ataxia-telangiectasia and cancer.Citation2 As a major gatekeeper of genome integrity, the regulation of Mec1/ATR has been a major area of research in the DNA repair field. Currently, there are 4 established pathways to activate Mec1/ATR: (1) through Dpb11/TopBP1; (2) through the 9-1-1 complex; (3) through Dna2 and (4) compensation by Tel1/ATM. Given that in eukaryotes the DNA double-strand break (DSB) occurs on chromatin, and that the recruitment of ATP-dependent chromatin remodeling complexes at the DSBs sites occurs on a time scale similar to that of the H2A phosphorylation and recruitment of DNA damage repair and checkpoint proteins, it is possible that the chromatin remodeling complexes contributes in the regulation of DNA repair and checkpoint pathways.Citation3,4 The SWI/SNF-dependent Mec1 activation in this study provides a new and unconventional mechanism to regulate a key checkpoint kinase in response to DNA damage (). As such, revealing the mechanistic details of this novel mode of Mec1 activation will have a significant impact in the field of DNA repair. Moreover, this study also identified non-chromatin substrates for ATP-dependent chromatin remodeling complexes, which opens a major new research direction in the chromatin field as well.
Figure 1. Figure showing the mechanism of regulation of Mec1 kinase through SWI/SNF chromatin remodeling complex in response to DNA damage.
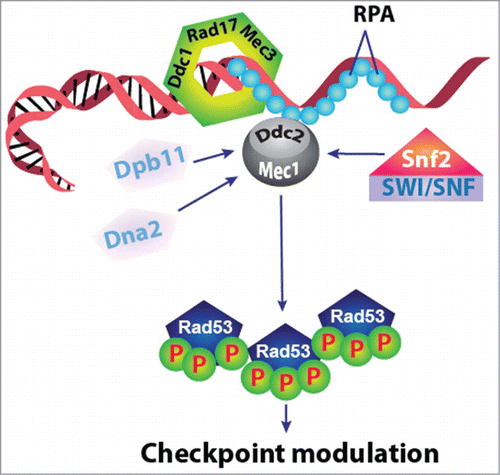
The SWI/SNF complex is a highly conserved multi-subunit assembly that plays a key role in the regulation of transcription. The yeast SWI/SNF complex consists of ˜12 different subunits and is required in vivo for the transcriptional induction of a large subset of genes and for the functioning of a variety of sequence-specific transcriptional activators. SWI/SNF complexes remodel nucleosome structure and are capable of mobilizing nucleosomes both by sliding and by catalyzing the ejection and insertion of histone octamers.Citation5 How does SWI/SNF engage a non-chromatin substrate? It has been shown that SWI/SNF can disrupt DNA-histone and histone-histone interactions during chromatin remodeling, and that several of the SWI/SNF subunits are required to interact with the nucleosome substrate. In contrast, the current study suggests that the Snf2 core ATPase is the main interaction partner for the non-chromatin Mec1 substrate, thereby suggesting distinct mechanisms for SWI/SNF complex to engage a nucleosome vs. non-chromatin substrate. How is Mec1 activated by the SWI/SNF chromatin remodeling complex? The established Mec1/ATR activators such as Dpb11 and TopBP1 contain unstructured Mec1/ATR-activation domains (AAD), which can bind to both Mec1/ATR and Ddc2/ATRIP, leading to Mec1/ATR activation. Authors in this study showed that SWI/SNF activates Mec1 in an ATP-dependent fashion, suggesting an unconventional mode of Mec1 activation, where ATP-dependent conformational changes in SWI/SNF could potentially unmask the Mec1-activation domain in Snf2 to activate Mec1.
The SWI/SNF complex is evolutionarily conserved from yeast to human (BAF complex). Given the prevalence of Snf2/Brg1 point mutations in different types of cancer cells, it is critical to understand which cellular processes they disrupt. Since the SWI/SNF complex was first established as a gene-regulator, cancer studies of SWI/SNF mostly focused on mis-regulation of genes, such as genes involved in cell proliferation.Citation6 The discovery of SWI/SNF as a novel DNA damage checkpoint regulator raises a new possibility to study SWI/SNF cancer mutations in the context of genome integrity. In human cells, the loss of Brg1 has been shown to reduce the level of γ-H2AX, suggesting a defect in ATM/ATR activation.Citation7 As such, SWI/SNF-mediated checkpoint regulation appears to be conserved from yeast to human. Taken together, these studies suggest that ATP-dependent chromatin remodeling complexes such as SWI/SNF can interact with and regulate the activity of non-chromatin substrates such as the Mec1/ATR kinases, and that the novel mode of Mec1/ATR regulation by SWI/SNF may provide a new link to SWI/SNF-related cancer.
References
- Kapoor P, et al. Genes Dev 2015; 29:591-602; PMID:25792597; http://dx.doi.org/10.1101/gad.257626.114
- Gobbini E, et al. DNA Repair (Amst) 2013; 12:791-9; PMID:23953933; http://dx.doi.org/10.1016/j.dnarep.2013.07.009
- Morrison AJ, Shen X. Nat Rev Mol Cell Biol 2009; 10:373-84; PMID:19424290; http://dx.doi.org/10.1038/nrm2693
- Bao Y. Epigenomics 2011; 3:307-21; PMID:22122340; http://dx.doi.org/10.2217/epi.11.14
- Bartholomew B. Annu Rev Biochem 2014; 83:671-96; PMID:24606138; http://dx.doi.org/10.1146/annurev-biochem-051810-093157
- Klochendler-Yeivin A, et al. Curr Opin Genet Dev 2002; 12:73-9; PMID:11790558; http://dx.doi.org/10.1016/S0959-437X(01)00267-2
- Park JH, et al. Embo J 2006; 25:3986-97; PMID:16932743; http://dx.doi.org/10.1038/sj.emboj.7601291
