ABSTRACT
Plants exhibit several restrictions under waterlogging conditions, including stomatal limitations, negative impacts on gas exchange, lower nutrient uptake and reduced growth. 24-epibrassinolide (EBR) is a polyhydroxylated steroid, with the advantages to be a natural and biodegradable molecule, presenting beneficial roles in metabolic and physiological processes. The aim of this research is to investigate whether EBR can protect soybean plants against damage caused by waterlogging and evaluate the responses associated with the root and leaf anatomy, photosynthetic machinery and biomass. This study used a completely randomized factorial design with two water conditions (control and waterlogging) and three concentrations of 24-epibrassinolide (0, 5 and 10 nM EBR). This steroid stimulated the activities of enzymes linked to the antioxidant system and resulted in minor damage to the chloroplast membranes. EBR maximized the efficiency of photosystem II and improved the gas exchange, which was explained by the higher density and index of the stomata in addition to the increased chlorophyll content and electron transport rate. In root structures, EBR mitigated the impact of waterlogging on vascular cylinder and metaxilem, suggesting maintenance and functions of these structures in plants stressed.
Introduction
Soybean (Glycine max) is the main leguminous plant cultivated worldwide, and approximately 338 million tons were produced in the 2017/2018 harvest, with the USA, Brazil and Argentina the main producers.Citation1 Grains are widely used for human and animal food, and they are also used for the production of biofuels due to their high protein and oil content. Crop soybean is found in many agricultural regions, although such specie can suffer limitations due to environmental factors, such as waterlogging.
Waterlogging may be caused by inappropriate irrigation management, climate changes and irregular drainage. The main implication of waterlogging is the reduction of oxygen availability, which initially leads to hypoxia and can induce anoxic conditions. Under waterlogging conditions, plants exhibit several restrictions, including stomatal limitations, negative gas exchange changes, lower carbon dioxide (CO2) assimilation, lower nutrient uptake and reduced growth.Citation2 Plant cells grown under waterlogging conditions also have low oxygen availability that often causes a redox state imbalance and oxidative damage due to the accumulation of reactive oxygen species (ROS), such as superoxide (O2−) and hydrogen peroxide (H2O2). Studies of plants under waterlogging show that such conditions have a negative impact on plant metabolism. Study with four Vigna radiata genotypes and reported that waterlogging applied during the vegetative stage decreased the net photosynthetic rate (PN) and chlorophyll contents, resulting in reductions in leaf area and growth rate.Citation3 Physiological and biochemical behavior of Glycine max plants exposed to waterlogging indicated that reductions in growth were associated with limitations in stomatal conductance (gs) caused by waterlogging.Citation4
Available literature reveals the potential of the brassinosteroids (BRs) to induce plant tolerance to abiotic stresses, including heavy metal contamination, drought, salinity, high temperature and cold.Citation5 BRs, including 24-epibrassinolide (EBR), are polyhydroxylated steroids, which are natural and biodegradable molecules with beneficial roles in metabolic and physiological processes.Citation6,Citation7 These steroids regulate fermentative enzyme activities,Citation8 plant cell division and vascular differentiation,Citation9 stimulating reactive oxygen species (ROS) detoxificationCitation10 and increasing the photosynthetic apparatus efficiency.Citation11
Our hypothesis considered the negative impacts of hypoxia or anoxia on plants, and soybean was used as the model in this research due to its high sensitivity to waterlogging. Exogenous application of EBR in Solanum lycopersicum plants improved the gas exchange, including PN, due to the favorable regulation of stomatal characteristics.Citation12 Additionally, EBR treatment in Glycine max plants induced positive repercussions on root structures, promoting increment in vascular cylinder (VCD).Citation13 Therefore, the objective of this research is to investigate whether EBR can protect soybean plants against damages caused by waterlogging, evaluate the responses associated with the root and leaf anatomy, photosynthetic machinery and biomass, and define a better dose-response relationship.
Materials and methods
Location and growth conditions
The experiment was performed at the Campus of Paragominas of the Universidade Federal Rural da Amazônia, Paragominas, Brazil (2°55ʹ S, 47°34ʹ W). The study was conducted in a greenhouse with the temperature and humidity controlled. The minimum, maximum, and median temperatures were 23.4, 30.6 and 25.8°C, respectively. The relative humidity during the experimental period varied between 60% and 80%.
Plants, containers and acclimation
Seeds of Glycine max (L.) Merr. var. M8644RR acquired from Monsoy™ were germinated directly in pots (1.2-L, being 0.15 m in height and 0.10 m in diameter) filled with a mixed substrate of sand and vermiculite at a ratio of 3:1. The plants were cultivated under semi-hydroponic conditions with water aeration at 500 mL of distilled water for eight days. A modified nutrient solution 32 was used, with the ionic strength beginning at 50% (6th day after germination) and later modified to 100% after two days (8th day after germination). After this period, the nutritive solution remained at the total ionic strength.
Experimental design
The experiment followed a completely randomized factorial design with two water conditions (control and waterlogging) and three concentrations of 24-epibrassinolide (0, 5 and 10 nM EBR). Five replicates for each of six treatments were performed, and a total of 30 experimental units were used in the experiment, with one plant in each unit.
24-epibrassinolide (EBR) preparation and application
Ten-day-old seedlings were treated with 24-epibrassinolide (EBR) or Milli-Q water via the roots (containing a proportion of ethanol that was equal to that used to prepare the EBR solution)Citation14 for 28 days (days 10–38 after the start of the experiment). EBR solutions (0, 5 and 10 nM) were prepared with reagent acquired from Sigma-Aldrich (USA) and defined in concordance with study of Amzallag and Vaisman.Citation15
Plant cultivation and waterlogging
Plants received the following macro- and micronutrients from the nutrient solution.Citation16 To simulate waterlogging conditions, large 8-L pots (0.25 m in height and 0.20 in diameter) without drainage holes were used. The pots containing the plants were placed into larger pots and then waterlogging was applied (30th to 38th day after the start of the experiment). The water level was maintained at 3 cm above the root collar and was adjusted when necessary. Plants in the control treatment were maintained under semi-hydroponic conditions with water aeration. On day 38 of the experiment, physiological and morphological parameters were measured for all plants and leaf tissues were harvested for the anatomical and biochemical analyzes, with one plant per pot used to examine the plant parameters.
Measurement of chlorophyll fluorescence and gas exchange
Chlorophyll fluorescence was measured in fully expanded leaves under light using a modulated chlorophyll fluorometer (model OS5p; Opti-Sciences). Preliminary tests determined the location of the leaf, the part of the leaf and the time required to obtain the greatest Fv/Fm ratio; therefore, the acropetal third of the leaves, which was the middle third of the plant and adapted to the dark for 30 min, was used in the evaluation. The intensity and duration of the saturation light pulse were 7,500 µmol m–2 s–1 and 0.7 s, respectively. Gas exchange was evaluated in all plants and measured in the expanded leaves in the middle region of the plant using an infrared gas analyzer (model LCPro+; ADC BioScientific) in a chamber under constant CO2, photosynthetically active radiation, air-flow rate and temperature conditions at 360 μmol mol−1 CO2, 800 μmol photons m−2 s−1, 300 µmol s−1 and 28°C, respectively, between 10:00 and 12:00 h. This study estimated water-use efficiency (WUE)Citation17 and instantaneous carboxylation efficiency (PN/Ci).Citation18
Measurements of the anatomical parameters
Samples were collected from the middle region of the leaf limb of fully expanded leaves and roots 5 cm from the root apex. Subsequently, all collected botanical material was fixed in FAA 70 for 24 hours, dehydrated in ethanol and embedded in historesin (Leica, Nussloch, Germany). Transverse sections with a thickness of 5 μm were obtained with a rotating microtome (Leica RM 2245, Leica Biosystems) and stained with toluidine blue.Citation19 For stomatal characterization was used the epidermal impression method.Citation20 The slides were observed and photomicrographed under an optical microscope (Motic BA 310, Motic Group Co. LTD.) coupled to a digital camera (Motic 2500, Motic Group Co., LTD.). The images were analyzed with Motic plus 2.0, which was previously calibrated with a micrometer slide from the manufacturer.
Determination of the antioxidant enzymes, superoxide and soluble proteins
Antioxidant enzymes [superoxide dismutase (SOD), Catalase (CAT), ascorbate peroxidase (APX), and peroxidase (POX)], superoxide, and soluble proteins were extracted from leaf tissue.Citation21 The total soluble proteins were quantified.Citation22 The SOD assay was measured at 560 nm,Citation23 and the SOD activity was expressed in mg–1 protein. The CAT assay was detected at 240 nm,Citation24 and the CAT activity was expressed in μmol H2O2 mg–1 protein min–1. The APX assay was measured at 290 nm,Citation25 and the APX activity was expressed in μmol AsAmg–1 protein min–1. The POX assay was detected at 470 nm,Citation26 and the activity was expressed in μmol tetraguaiacol mg–1 protein min–1. Superoxide (O2−) was measured at 530 nm.Citation27
Quantification of hydrogen peroxide, malondialdehyde and electrolyte leakage
Stress indicators [hydrogen peroxide (H2O2) and malondialdehyde (MDA)] were extracted.Citation28 H2O2 was measured.Citation29 MDA was determinedCitation30 using an extinction coefficient of 155 mM−1 cm−1. Electrolyte leakage (EL) was measuredCitation31 and calculated by the formula EL (%) = (EC1/EC2) × 100.
Determination of photosynthetic pigments and biomass
The chlorophyll and carotenoid determinationsCitation32 were performed using a spectrophotometer (model UV-M51; Bel Photonics). Biomass of roots, stems and leaves was measured based on constant dry weights (g) after drying in a forced-air ventilation oven at 65°C.
Data analysis
The data were assessed via an ANOVA and the Scott–Knott test at a probability level of 5%. All statistical procedures were performed using Assistat software.
Results
EBR induced beneficial effects on the root and leaf anatomy in plants subjected to waterlogging
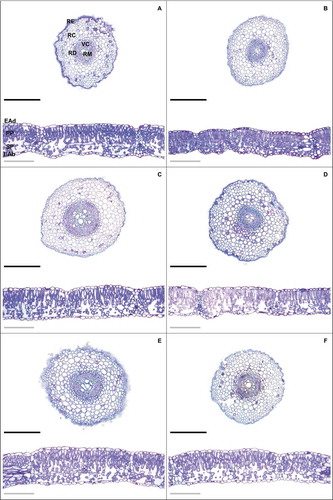
Waterlogging delayed the development of the roots () and reduced the root epidermis thickness (RET), root endodermis thickness (RDT), root cortex diameter (RCD), vascular cylinder diameter (VCD) and root metaxylem diameter (RDM) values; however, application of 10 nM EBR minimized the negative effects on these parameters, inducing significant increases of 39%, 58%, 57%, 28% and 28% (), respectively, compared to the waterlogging + 0 nM EBR treatment. For the leaf anatomy, plants subjected to waterlogging showed reductions in epidermis thickness from adaxial leaf side (ETAd), epidermis thickness from abaxial leaf side (ETAb), palisade parenchyma thickness (PPT) and spongy parenchyma thickness (SPT) (), whereas the utilization of 10 nM EBR led to increases in these variables of 27%, 26%, 18% and 39% (), respectively, compared to the waterlogging + 0 nM EBR treatment. The PPT/SPT ratio increased in plants subjected to waterlogging and reduced (14%) in the treatment with steroid application (10 nM), compared to the waterlogging + 0 nM EBR treatment.
Table 1. Root and leaf anatomy in soybean plants treated with EBR and subjected to waterlogging
Figure 1. Root and leaf cross sections in soybean plants treated with EBR and subjected to waterlogging. Control + 0 nM EBR (a), waterlloging + 0 nM EBR (b), control + 5 nM EBR (c), waterlloging + 5 nM EBR (d), control + 10 nM EBR (e), waterlloging + 10 nM EBR (f). Legends: RE = Root epidermis; RC = Root cortex; RD = Root endodermis; VC = Vascular cylinder; RM = Root metaxylem; EAd = adaxial epidermis; EAb = Adaxial epidermis; PP = Palisade parenchyma; SP = Spongy parenchyma. Black bars = 500 µm and gray bars = 200 µm
Pre-treatment with steroid minimized the damages on photosynthetic pigments
Waterlogging decreased the Chl a (Chlorophyll a), Chl b (Chlorophyll b), Total Chl (Total chlorophyll) and Car (Carotenoids) contents, but EBR attenuated the negative impacts (), and the concentration of 10 nM EBR modulated significant increases of 14%, 47%, 19% and 111%, respectively, when compared to the waterlogging + 0 nM of EBR treatment. Chl a/Chl b and Total Chl/Car showed increases in the waterlogging condition, although their values were reduced (P < .05) by 22% and 44% after treatment with 10 nM EBR, respectively, when compared to the waterlogging without EBR treatment.
Table 2. Photosynthetic pigments, chlorophyll fluorescence and gas exchange in soybean plants treated with EBR and subjected to waterlogging
EBR mitigated the effects caused by waterlogging on photosynthetic machinery
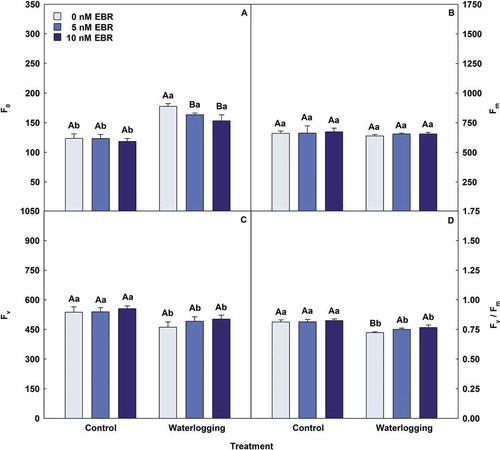
Waterlogging favored an increase in minimal fluorescence yield of the dark-adapted state (F0), which was minimized (14%) with the application of 10 nM EBR (). However, the values of maximal fluorescence yield of the dark-adapted state (Fm), variable fluorescence (Fv) and maximal quantum yield of PSII photochemistry (Fv/Fm) were lower under waterlogging conditions and increased after the application of 10 nM EBR by 3%, 9% and 6%, respectively. Plants under waterlogging presented reductions in the levels of effective quantum yield of PSII photochemistry (ΦPSII), photochemical quenching coefficient (qP) and electron transport rate (ETR) (); however, the application of 10 nM EBR promoted an increase in these variables by 35%, 37% and 34%, respectively, when compared to waterlogging without EBR. For nonphotochemical quenching (NPQ), relative energy excess at the PSII level (EXC) and ratio between the electron transport rate and net photosynthetic rate (ETR/PN), the waterlogging condition generated increases; however, the EBR significantly reduced these variables. For NPQ and EXC, the application of 10 nM EBR led to reductions of 28% and 19%, respectively, and for ETR/PN, the lowest value was observed for waterlogging + 5 nM EBR (reduction of 26%) when compared to the waterlogging + 0 nM EBR treatment. To gas exchange, waterlogging reduced the net photosynthetic rate (PN), transpiration rate (E), stomatal conductance (gs), water-use efficiency (WUE) and carboxylation instantaneous efficiency (PN/Ci), but the application of 5 and 10 nM EBR led to increases in these variables (). PN, E, gs, WUE and PN/Ci increases of 57%, 22%, 17%, 28% and 73% in the plants in the 5 nM EBR and waterlogging treatment, respectively, and increases of 37%, 5%, 17%, 30% and 45% in the plants in the waterlogging + 10 nM EBR treatment, respectively, when compared to the waterlogging + 0 nM EBR treatment. However, the levels of intercellular CO2 concentration (Ci) were higher in plants under waterlogging and lower with the use of the steroid, with the 5 and 10 nM EBR treatments decreasing the values by 10% and 6%, respectively, when compared to the waterlogging + 0 nM EBR treatment.
Figure 2. Minimal fluorescence yield of the dark-adapted state (F0), maximal fluorescence yield of the dark-adapted state (Fm), variable fluorescence (Fv) and maximal quantum yield of PSII photochemistry (Fv/Fm) in soybean plants treated with EBR and subjected to waterlogging. Columns with different uppercase letters between EBR levels (0, 5 and 10 nM EBR under equal water condition) and lowercase letters between water conditions (control and waterlogging under equal EBR concentration) indicate significant differences from the Scott-Knott test (P < .05). Columns corresponding to means from five repetitions and standard deviations
Steroid positively regulated the stomatal characteristics
Waterlogging caused reductions on stomatal density (SD), stomatal functionality (SF) and stomatal index (SI) on both faces (); however, EBR minimized these effects, with 10 nM EBR generating increases of 34%, 10% and 19% on the adaxial face, respectively, and 10%, 13% and 18% on the abaxial side, respectively. For polar diameter of the stomata (PDS) and equatorial diameter of the stomata (EDS), the values were higher under waterlogging conditions and reduced under the 10 nM EBR treatment by 17% and 22% on the adaxial face, respectively, and by 9% and 19% abaxial face, respectively, compared to the waterlogging + 0 nM EBR treatment.
Table 3. Stomatal characteristics in soybean plants treated with EBR and subjected to waterlogging
EBR stimulated the antioxidant system and reduced oxidative stress generated by waterlogging
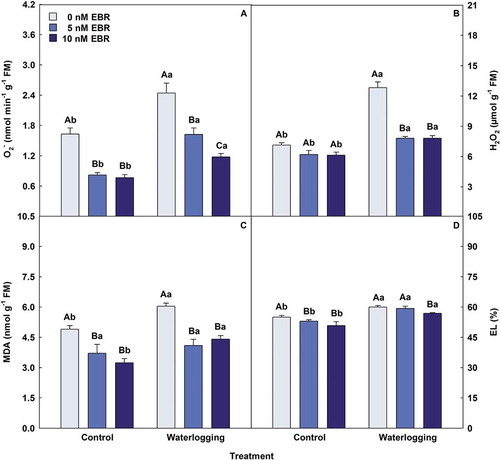
Waterlogged plants showed significant increases in all antioxidant enzymes (), when compared with control treatment. Exogenous EBR on waterlogging plants promoted significant increases in SOD (25%), APX (30%) and POX (59%) activities, respectively, as well as non-significant to CAT (14%). Plants submitted to waterlogging had increments related to stress indicators (). However, EBR minimized this effect, treatment using 10 nM EBR led to reductions (P < .05) in O2−, H2O2, MDA, and EL of 52%, 39%, 27% and 5%, respectively, compared with waterlogging + 0 nM EBR.
Figure 3. Activities of superoxide dismutase (SOD), catalase (CAT), ascorbate peroxidase (APX) and peroxidase (POX) in soybean plants treated with EBR and subjected to waterlogging. Columns with different uppercase letters between EBR levels (0, 5 and 10 nM EBR under equal water condition) and lowercase letters between water conditions (control and waterlogging under equal EBR concentration) indicate significant differences from the Scott-Knott test (P < .05). Columns corresponding to means from five repetitions and standard deviations
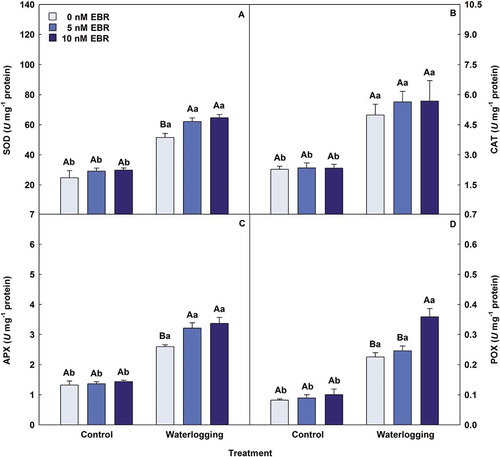
Figure 4. Superoxide (O2−), hydrogen peroxide (H2O2), malondialdehyde (MDA) and electrolyte leakage (EL) in soybean plants treated with EBR and subjected to waterlogging. Columns with different uppercase letters between EBR levels (0, 5 and 10 nM EBR under equal water condition) and lowercase letters between water conditions (control and waterlogging under equal EBR concentration) indicate significant differences from the Scott-Knott test (P < .05). Columns corresponding to means from five repetitions and standard deviations
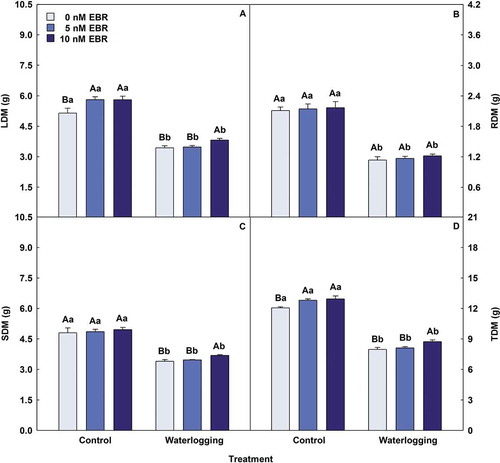
EBR alleviated the impacts of waterlogging on biomass
Waterlogging significantly reduced the leaf dry matter (LDM), root dry matter (RDM), stem dry matter (SDM) and total dry matter (TDM) (). However, the application of 10 nM EBR promoted significant increments in LDM (11%), SDM (9%) and TDM (9%), but non-significant in RDM (7%), comparing with treatment waterlogging + 0 nM EBR. Plants pretreated with 5 nM EBR and submitted to waterlogging had increments of 1%, 3%, 2% and 2% in LDM, RDM, SDM and TDM, respectively.
Figure 5. Leaf dry matter (LDM), root dry matter (RDM), stem dry matter (SDM) and total dry matter (TDM) in soybean plants treated with EBR and subjected to waterlogging. Columns with different uppercase letters between EBR levels (0, 5 and 10 nM EBR under equal water condition) and lowercase letters between water conditions (control and waterlogging under equal EBR concentration) indicate significant differences from the Scott-Knott test (P < .05). Columns corresponding to means from five repetitions and standard deviations
Discussion
Exogenous EBR had a positive effect on the root anatomy (RET, RDT, RCD, VCD and RDM) in plants subjected to waterlogging, thus revealing the ability of EBR to positively to modulate cell division and elongation. BRs show multiple actions on plant metabolism, including signaling, regulation and differentiation of stem cells in the root meristem, thereby promoting root growth. Exogenous EBR (50 nM) applied in young Eucalyptus urophylla plants under saline stress (250 mM) increased RET (17%), RDT (24%), RCD (11%,), VCD (9%) and RMD (10%) compared to plants under equal treatment without EBR.Citation33
Plants pre-treated with EBR subjected to waterlogging showed increases in ETAd, ETAb, PPT and SPT, evidencing the positive influence of this steroid on leaf anatomy. These increments can be related to the benefits promoted by EBR on PN/Ci, which were shown in this research, because a thicker spongy parenchyma can accumulate more CO2, thus maximizing the supplementation of this gas obtained from the intercellular spaces of the substomatal cavity in the direction of the external surface of the chloroplast cells. Thickening of the palisade parenchyma, which is associated with PPT, induces higher utilization of the incident radiation and indirectly increases PN.Citation34 Additionally, plants subjected to waterlogging and EBR presented reductions in PPT/SPT, being related to higher increments promoted by this steroid on SPT (39%), when compared with PPT (18%), suggesting that EBR acts of form more intense on SPT. Application of EBR on growth and metabolism of two Solanum lycopersicum genotypes resulted in increases of 10% ETAd, 33% ETAb and 5% PPT, when compared with plants of the same genotype without EBR.Citation12
Plants under waterlogging + EBR presented increases in Chl a, Chl b, Chl total and Car levels, suggesting that the EBR minimized damages on membrane, which was corroborated by the reductions of MDA in this study. EBR has the ability to protect the chloroplast ultrastructure against degradation caused by ROS during the stress.Citation35 Research on photosynthetic responses and stress tolerance during root hypoxia in five Phaseolus vulgaris cultivars revealed reductions in Chl a, Chl b and Car of 24%, 13% and 26%, respectively.Citation36 Physiological behavior of Chenopodium quinoa exposed to drought and waterlogging demonstrated that waterlogging significantly reduced Chl a (14%), Chl b (14%) and Chl total (19%) levels, compared to the control treatment.Citation37 Plants subjected to waterlogging and EBR showed reductions in Chl a/Chl b and Total Chl/Car, which are associated with higher increments on Chl b and Car, indicating that this steroid also acts on carotenoids, in which these pigments are intrinsically related to protection of the chlorophylls. Oxidative stress in Cajanus cajan genotypes induced by waterlogging and antioxidant activity found that after 6 days, there were drastic reductions in the levels of total Chl and increases in the Chl a/Chl b ratio.Citation38
The application of 10 nM EBR maximized the Fm, Fv and Fv/Fm values of plants subjected to waterlogging because EBR minimized photoinhibition and decreased the dissipation of excitation energy in the antennas of photosystem II. Plants subjected to waterlogging + EBR showed increases in the ΦPSII, qP and ETR values, which was related to the previously described benefits of EBR on Fv/Fm and F0. Reductions in ETR under waterlogging conditions can be indicative of a low efficiency of the plastoquinone (PQ), which is used in oxidation-reduction reactions. EBR led to increases in qP because this steroid favors the recovery of the D1 protein and minimizes the damage to reaction centers caused by photoinhibition.Citation39 EBR spray in Glycine max plants under water deficit increased ΦPSII, qP and ETR, because this steroid can induce the oxidation of plastoquinone (PQ) and improve the performance of the reaction centers from photosystem II (PSII).Citation16
EBR minimized NPQ, EXC and ETR/PN. The reductions in the levels of ETR/PN are associated with improvements promoted by EBR in PN as detected in this study. EBR optimizes the use of electrons produced in the photochemical phase, minimizes Mehler reactions and photorespiration, and increases the availability of these electrons for activities related to CO2 fixation during the biochemical step. Additionally, reductions in EXC were a consequence of the increase in photochemical efficiency triggered by EBR by minimizing the NPQ values.Citation40 Deleterious effects linked to waterlogging in two Medicago sativa varieties provoked significant reductions in ΦPSII and an increase in NPQ (15 days) in Wairau variety.Citation41
EBR mitigated the effects of waterlogging on gas exchange, increasing gs, E and PN as found in this study. Steroid stimulated the density and index linked to stomatal, reflecting on physiological variables and facilitating the capture and distribution of CO2 in leaves, confirmed by PN/Ci. Concomitantly, increments on E can have a positive effect on nutrient uptake.Citation42,Citation43 Additionally, EBR improves CO2 fixation through positive regulation under synthesis and the activation of enzymes that participate in the Calvin cycle, including ribulose-1,5-bisphosphate carboxylase/oxygenase. Gas exchange in Helianthus tuberosus plants subjected to two waterlogging levels (moderate and severe) was significantly affected, decreasing PN (87%) and gs (90%) after severe waterlogging (5 days).Citation44 EBR application in Cucumis sativus plants under hypoxia (8 days) led to an increase in PN of 12% and higher sugars supply to hypoxic roots, alleviating the cytosolic acidificationCitation8.
The reductions in Ci promoted by the pretreatment with EBR demonstrate the capacity of this steroid to maximize the fixation efficiency of carbon dioxide (CO2) through increments in SD and gs as detected in this research. EBR improved the CO2 diffusion, enhancing stomatal variables and leaf anatomy.Citation13 Changes induced by the waterlogging on gas exchange and oxidative compounds in Zea mays plants resulted in significant increases in Ci (72 and 96 hours).Citation45
EBR minimized the negative effects of waterlogging on SD, PDS, EDS, SF and SI. The increments in SD, SF and SI were related to ability of EBR positively regulate stoma formation.Citation46,Citation47 The increases in SI are consistent with the improvements in gas exchange described in this study. Positive relationship linked to SD with gs, PN and WUE suggested that this variable and the size of the guard cell suffered changes in response to waterlogging, because they are closely associated with the photosynthesis process and the use of water by the plant. Additionally, plants subjected to waterlogging + EBR showed reductions in PDS and EDS, because EBR induces the formation of stomata more elliptical, being elliptical shape more efficient and functional, if compared with rounded shape.Citation48 EBR actions on stomatal characteristics and growth in Solanum lycopersicum plants provided increments of 9%, 5% and 10% in SD, SF and SI (abaxial face), respectively, when compared to plants without EBR.Citation12
The increases in the activities of the SOD, CAT, APX and POX enzymes with EBR induced reductions in O2− and H2O2 in this study. EBR stimulates the activities of enzymes linked to ROS detoxification, thereby minimizing the oxidative damages generated by waterlogging. SOD is the first line of defense against ROS and is important in the detoxification of O2− to H2O2, which is subsequently catalyzed by CAT, APX and POX. Research using two contrasting Cucumis sativus seedlings (sensitive and tolerant to hypoxia) treated with exogenous EBR revealed that increases in SOD and POX activities under hypoxia stress induced reductions in O2− and H2O2.Citation10 Antioxidant system of Gossypium hirsutum plants under different temperatures and waterlogging times (3 and 6 days) presented significant increases in SOD, CAT and POX.Citation49
The application of EBR minimized the effects of waterlogging on O2−, H2O2, MDA, and EL, and these results are connected to the increases in the activities of antioxidant enzymes (SOD, CAT, APX and POX) detected in this study. Lower levels of H2O2 demonstrate higher efficiency of the enzymes CAT and APX, in which CAT acts to catalyze H2O2, which is produced during photorespiration and β-oxidation of fatty acids, while APX reduces H2O2 to H2O. Oxidative stress induced by waterlogging in two genotypes of Cajanus cajan resulted in a significant increase (53%) of O2− in the Pusa 207 genotype.Citation38
Reductions in MDA and EL promoted by EBR demonstrate the ability of this steroid to protect the integrity of membranes. In plants under waterlogging, increments detected in EL suggests a loss in the stability of the cell membrane, confirmed by MDA. MDA is a useful stress indicator due to low cost and fast determination, being used to determine oxidative lipid injury occasioned during abiotic and biotic stresses. Waterlogging and three temperatures (16°C, 22°C and 28°C) on stress indicators in Arabidopsis thaliana induced significant increases in MDA under temperatures of 22°C and 28°C.Citation50
EBR minimized the negative effects of the waterlogging on growth based on the improvements in the antioxidant system and photosynthesis as previously described. Reductions on RDM is clearly linked to hypoxia induced by waterlogging, because under these conditions there is an intense restriction of dissolved O2, which impairs the respiration process and induces oxidative stress, resulting in the inhibition of root growth. EBR alleviated the growth inhibition in Cucumis sativus seedlings exposed to waterlogging, improving CO2 assimilation and protecting leaves against oxidative damages.Citation11 Adverse effects of the waterlogging (seven days) in Gossypium hirsutum plants significantly reduced the RDM and SDM.Citation51 EBR actions on the growth and yield of Zea mays plants after 10 days of waterlogging attenuated the negative impacts and generated an increase in the LDM.Citation52
Conclusions
Our research clearly demonstrated that exogenous EBR protected soybean plants against oxidative stress caused by waterlogging, which was confirmed by the improvements in the root anatomy, photosynthetic machinery and biomass. This steroid stimulated the activities of enzymes linked to the antioxidant system (superoxide dismutase, catalase, ascorbate peroxidase and peroxidase), thus resulting in minor damage to the chloroplast membranes, which was corroborated by the reductions in malondialdehyde and maintenance of photosynthetic pigments. Additionally, EBR maximized the efficiency of photosystem II and mitigated the deleterious effects promoted by photoinhibition. Moreover, increases in gas exchange were due to direct effects related to the higher density and index of the stomata and indirectly connected to increases in the chlorophyll content and electron transport rate. In the root tissue, EBR mitigated the impact of waterlogging on the root anatomy, thus favoring the maintenance and functioning of the vascular cylinder and metaxylem, which are structures intrinsically related to the assimilation of water and essential nutrients for growth. Therefore, our results prove that pre-treatment with EBR improved the tolerance of soybean plants to waterlogging and that the application of 10 nM EBR produced better effects on biomass, mainly in the leaf and stem.
Author contribution statement
AKSL was the advisor of this project, planning all phases of this research. YCP and FRS conducted the experiment in the greenhouse and performed physiological, biochemical and morphological determinations, while BRSS measured anatomical parameters. FJRC and DJM critically revised and edited the manuscript. All authors read and approved final version of manuscript.
Data availability statement
Data are available upon request to the corresponding author.
Disclosure statement
The authors declare that they have no competing interests.
Additional information
Funding
References
- FAO. Food and agriculture organization of the united nations. Food outlook: biannual report on global food markets [Internet]. 2018 [accessed 2019 Dec 27]. www.fao.org/publications
- Ren B, Zhang J, Dong S, Liu P, Zhao B. Effects of duration of waterlogging at different growth stages on grain growth of summer maize (Zea mays L.) under field conditions. J Agron Crop Sci. 2016;202:1–12.
- Kumar P, Pal M, Joshi R, Sairam RK. Yield, growth and physiological responses of mung bean [Vigna radiata (L.) Wilczek] genotypes to waterlogging at vegetative stage. Physiol Mol Biol Plants [Internet]. 2013;19:209–220. http://link.springer.com/10.1007/s12298-012-0153-3.
- Garcia N, Da-Silva CJ, Cocco KLT, Pomagualli D, de Oliveira FK, da Silva JVL, de Oliveira ACB, Amarante LD. Waterlogging tolerance of five soybean genotypes through different physiological and biochemical mechanisms. Environ Exp Bot [Internet]. 2020;172:103975. https://linkinghub.elsevier.com/retrieve/pii/S0098847220300010.
- Ahanger MA, Ashraf M, Bajguz A, Ahmad P. Brassinosteroids regulate growth in plants under stressful environments and crosstalk with other potential phytohormones. J Plant Growth Regul [Internet]. 2018;37:1007–1024. http://dx.doi.10.1007/s00344-018-9855-2.
- Xia X-J, Wang Y-J, Zhou Y-H, Tao Y, Mao W-H, Shi K, Asami T, Chen Z, Yu J-Q. Reactive oxygen species are involved in brassinosteroid-induced stress tolerance in cucumber. Plant Physiol [Internet]. 2009;150:801–814. http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=2689980&tool=pmcentrez&rendertype=abstract.
- She J, Han Z, Kim TW, Wang J, Cheng W, Chang J, Shi S, Wang J, Yang M, Wang ZY, et al. Structural insight into brassinosteroid perception by BRI1. Nature [Internet]. 2011;474:472–477. http://dx.doi.10.1038/nature10178.
- Kang YY, Guo SR, Li J, Duan JJ. Effect of root applied 24-epibrassinolide on carbohydrate status and fermentative enzyme activities in cucumber (Cucumis sativus L.) seedlings under hypoxia. Plant Growth Regul. 2009;57:259–269.
- Fariduddin Q, Yusuf M, Ahmad I, Ahmad A. Brassinosteroids and their role in response of plants to abiotic stresses. Biol Plant. 2014;58:9–17.
- Kang Y, Guo S, Duan J, Hu X. Effects of 24-epibrassinolide on antioxidant system and anaerobic respiratory enzyme activities in cucumber roots under hypoxia stress. J Plant Physiol Mol Biol. 2006;32:535–542.
- Ma YH, Guo SR. 24-epibrassinolide improves cucumber photosynthesis under hypoxia by increasing CO2 assimilation and photosystem II efficiency. Photosynthetica [Internet]. 2014;52:96–104. http://link.springer.com/10.1007/s11099-014-0010-4.
- Maia CF, Silva BRS, Lobato AKS. Brassinosteroids positively modulate growth: physiological, biochemical and anatomical evidence using two tomato genotypes contrasting to dwarfism. J Plant Growth Regul [Internet]. 2018;37:1–14. http://link.springer.com/10.1007/s00344-018-9802-2.
- Santos LR, da Silva BRS, Pedron T, Batista BL, Lobato AKDS. 24-Epibrassinolide improves root anatomy and antioxidant enzymes in soybean plants subjected to zinc stress. J Soil Sci Plant Nutr. 2020;20:105–124.
- Ahammed GJ, Choudhary SP, Chen S, Xia X, Shi K, Zhou Y, Yu J. Role of brassinosteroids in alleviation of phenanthrene–cadmium co-contamination-induced photosynthetic inhibition and oxidative stress in tomato. J Exp Bot. 2013;64:199–213.
- Amzallag GN, Vaisman J. Influence of brassinosteroids on initiation of the root gravitropic response in Pisum sativum seedlings. Biol Plant. 2006;50:283–286.
- Pereira YC, Rodrigues WS, Lima EJA, Santos LR, Silva MHL, Lobato AKS. Brassinosteroids increase electron transport and photosynthesis in soybean plants under water deficit. Photosynthetica [Internet]. 2019;57:181–191. https://doi.10.32615/ps.2019.029.
- Ma CC, Gao YB, Guo HY, Wang JL. Photosynthesis, transpiration, and water use efficiency of Caragana microphylla, C. intermedia, and C. korshinskii. Photosynthetica [Internet]. 2004;42:65–70. http://link.springer.com/10.1023/B:PHOT.0000040571.63254.c2.
- Aragão RM, Silva EN, Vieira CF, Silveira JAG. High supply of NO3 − mitigates salinity effects through an enhancement in the efficiency of photosystem II and CO2 assimilation in Jatropha curcas plants. Acta Physiol Plant [Internet]. 2012;34:2135–2143. http://link.springer.com/10.1007/s11738-012-1014-y.
- O’Brien TP, Feder N, McCully ME. Polychromatic staining of plant cell walls by toluidine blue O. Protoplasma. 1964;59:368–373.
- Segatto FB, Bisognin DA, Benedetti M, Costa LC, Rampelotto MV, Nicoloso FT. A technique for the anatomical study of potato leaf epidermis. Ciência Rural [Internet]. 2004;34:1597–1601. http://www.scielo.br/scielo.php?script=sci_arttext&pid=S0103-84782004000500042&lng=pt&tlng=pt.
- Badawi GH, Yamauchi Y, Shimada E, Sasaki R, Kawano N, Tanaka K, Tanaka K. Enhanced tolerance to salt stress and water deficit by overexpressing superoxide dismutase in tobacco (Nicotiana tabacum) chloroplasts. Plant Sci [Internet]. 2004;166:919–928. http://linkinghub.elsevier.com/retrieve/pii/S0168945203005090.
- Bradford MM. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem [Internet]. 1976;72:248–254. http://linkinghub.elsevier.com/retrieve/pii/0003269776905273.
- Giannopolitis CN, Ries SK. Superoxide dismutases: I. occurrence in higher plants. Plant Physiol. 1977;59:309–314.
- Havir EA, McHale NA. Biochemical and developmental characterization of multiple forms of catalase in tobacco leaves. Plant Physiol [Internet]. 1987;84:450–455. http://www.plantphysiol.org/cgi/doi/10.1104/pp.84.2.450.
- Nakano Y, Asada K. Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in spinach chloroplasts. Plant Cell Physiol. 1981;22:867–880.
- Cakmak I, Marschner H. Magnesium deficiency and high light intensity enhance activities of superoxide dismutase, ascorbate peroxidase, and glutathione reductase in bean leaves. Plant Physiol [Internet]. 1992;98:1222–1227. http://www.plantphysiol.org/cgi/doi/10.1104/pp.98.4.1222.
- Elstner EF, Heupel A. Inhibition of nitrite formation from hydroxylammoniumchloride: A simple assay for superoxide dismutase. Anal Biochem [Internet]. 1976;70:616–620. http://linkinghub.elsevier.com/retrieve/pii/0003269776904887.
- Wu Q-S, Xia R-X, Zou Y-N. Reactive oxygen metabolism in mycorrhizal and non-mycorrhizal citrus (Poncirus trifoliata) seedlings subjected to water stress. J Plant Physiol [Internet]. 2006;163:1101–1110. http://linkinghub.elsevier.com/retrieve/pii/S0176161705003044.
- Velikova V, Yordanov I, Edreva A. Oxidative stress and some antioxidant systems in acid rain-treated bean plants protective role of exogenous polyamines. Plant Sci [Internet]. 2000;151:59–66. http://linkinghub.elsevier.com/retrieve/pii/S0168945299001971.
- Cakmak I, Horst WJ. Effect of aluminium on lipid peroxidation, superoxide dismutase, catalase, and peroxidase activities in root tips of soybean (Glycine max). Physiol Plant [Internet]. 1991;83:463–468. http://doi.wiley.com/10.1111/j.1399-3054.1991.tb00121.x.
- Gong M, Li Y-J, Chen S-Z. Abscisic acid-induced thermotolerance in maize seedlings is mediated by calcium and associated with antioxidant systems. J Plant Physiol [Internet]. 1998;153:488–496. http://dx.doi.10.1016/S0176-1617(98)80179-X.
- Lichtenthaler HK, Buschmann C. Chlorophylls and carotenoids: measurement and characterization by UV‐VIS spectroscopy [Internet]. In: Current protocols in food analytical chemistry. Hoboken (NJ, USA): John Wiley & Sons, Inc.; 2001. p. 431–438. doi:https://doi.org/10.1002/0471709085.ch21.
- Oliveira VP, Lima MDR, Silva BRS, Batista BL, Lobato AKS. Brassinosteroids confer tolerance to salt stress in Eucalyptus urophylla plants enhancing homeostasis, antioxidant metabolism and leaf anatomy. J Plant Growth Regul. 2019;38:557–573.
- Asmar SA, Castro EM, Pasqual M, Pereira FJ, Soares JDR. Changes in leaf anatomy and photosynthesis of micropropagated banana plantlets under different silicon sources. Sci Hortic (Amsterdam) [Internet]. 2013;161:328–332. http://dx.doi.10.1016/j.scienta.2013.07.021.
- Tanveer M, Shahzad B, Sharma A, Khan EA. 24-Epibrassinolide application in plants: an implication for improving drought stress tolerance in plants. Plant Physiol Biochem [Internet]. 2019;135:295–303. https://doi.10.1016/j.plaphy.2018.12.013.
- Velasco NF, Ligarreto GA, Díaz HR, Fonseca LPM. Photosynthetic responses and tolerance to root-zone hypoxia stress of five bean cultivars (Phaseolus vulgaris L.). South African J Bot [Internet]. 2019;123:200–207. https://doi.10.1016/j.sajb.2019.02.010.
- González JA, Gallardo M, Hilal M, Rosa M, Prado FE. Physiological responses of quinoa (Chenopodium quinoa Willd.) to drought and waterlogging stresses: dry matter partitioning. Bot Stud. 2009;50:35–42.
- Kumutha D, Ezhilmathi K, Sairam RK, Chinnusamy V, Meena RC. Waterlogging induced oxidative stress and antioxidant enzyme activities in pigeon pea. Biol Plant. 2009;53:75–84.
- Yuan L, Shu S, Sun J, Guo S, Tezuka T. Effects of 24-epibrassinolide on the photosynthetic characteristics, antioxidant system, and chloroplast ultrastructure in Cucumis sativus L. under Ca(NO3)2stress. Photosynth Res. 2012;112:205–214.
- Lima MDR, Barros Junior UO, Batista BL, Lobato AKS. Brassinosteroids mitigate iron deficiency improving nutritional status and photochemical efficiency in Eucalyptus urophylla plants. Trees [Internet]. 2018;32:1681–1694. http://dx.doi.10.1007/s00468-018-1743-7.
- Irving LJ, Sheng YB, Woolley D, Matthew C. Physiological effects of waterlogging on two lucerne varieties grown under glasshouse conditions. J Agron Crop Sci. 2007;193:345–356.
- Ali B, Hayat S, Fariduddin Q, Ahmad A. 24-Epibrassinolide protects against the stress generated by salinity and nickel in Brassica juncea. Chemosphere [Internet]. 2008;72:1387–1392. https://linkinghub.elsevier.com/retrieve/pii/S0045653508004724.
- Inoue SI, Iwashita N, Takahashi Y, Gotoh E, Okuma E, Hayashi M, Tabata R, Takemiya A, Murata Y, Doi M, et al. Brassinosteroid involvement in arabidopsis thaliana stomatal opening. Plant Cell Physiol. 2017;58:1048–1058.
- Yan K, Zhao S, Cui M, Han G, Wen P. Vulnerability of photosynthesis and photosystem I in Jerusalem artichoke (Helianthus tuberosus L.) exposed to waterlogging. Plant Physiol Biochem [Internet]. 2018;125:239–246. https://doi.10.1016/j.plaphy.2018.02.017.
- Yordanova RY, Popova LP. Flooding-induced changes in photosynthesis and oxidative status in maize plants. Acta Physiol Plant [Internet]. 2007;29:535–541. http://link.springer.com/10.1007/s11738-007-0064-z.
- Kim T-W, Michniewicz M, Bergmann DC, Wang Z-Y. Brassinosteroid regulates stomatal development by GSK3- mediated inhibition of a MAPK pathway. Nature. 2012;482:419–422.
- González D, Fuentes S, Serna L. Interactions among gibberellins, brassinosteroids and genes regulate stomatal development in the arabidopsis hypocotyl. Int J Dev Biol. 2017;61:383–387.
- Khan PSSV, Kozai T, Nguyen QT, Kubota C, Dhawan V. Growth and water relations of Paulownia fortunei under photomixotrophic and photoautrophic conditions. Biol Plant [Internet]. 2003;46:161–166. http://link.springer.com/10.1023/A:1022844720795.
- Wang H, Chen Y, Hu W, Snider JL, Zhou Z. Short-term soil-waterlogging contributes to cotton cross tolerance to chronic elevated temperature by regulating ROS metabolism in the subtending leaf. Plant Physiol Biochem [Internet]. 2019;139:333–341. https://doi.10.1016/j.plaphy.2019.03.038.
- Xu L, Pan R, Shabala L, Shabala S, Zhang W-Y. Temperature influences waterlogging stress-induced damage in Arabidopsis through the regulation of photosynthesis and hypoxia-related genes. Plant Growth Regul [Internet]. 2019;89:143–152. http://link.springer.com/10.1007/s10725-019-00518-x.
- Ashraf MA, Ahmad MSA, Ashref M, Al-Qurainy F, Ashraf MY. Alleviation of waterlogging stress in upland cotton (Gossypium hirsutum L.) by exogenous application of potassium in soil and as a foliar spray. Crop Pasture Sci. 2011;62:25–38.
- Otie V, Ping A, Udo I, Eneji E. Brassinolide effects on maize (Zea mays L.) growth and yield under waterlogged conditions. J Plant Nutr [Internet]. 2019;42:954–969. https://doi.10.1080/01904167.2019.1584220.
