Abstract
Introduction: Atherosclerotic cardiovascular disease is a major contributor to morbidity and mortality in diabetic patients. Strict goal-directed lipid control in patients with diabetes is associated with better cardiovascular outcomes.
Aim: The main aim of this study is to describe the lipid profiles of a cohort of patients with type 2 diabetes mellitus in order to highlight the quality of lipid control by correlating the type and dose of lipid-modifying therapy used with lipid levels.
Method: A retrospective analysis was performed on 200 type 2 diabetic patients who attended the Charlotte Maxeke Johannesburg Academic Hospital diabetic clinic. Their lipid profiles and the type and dose of lipid-modifying therapy prescribed was assessed.
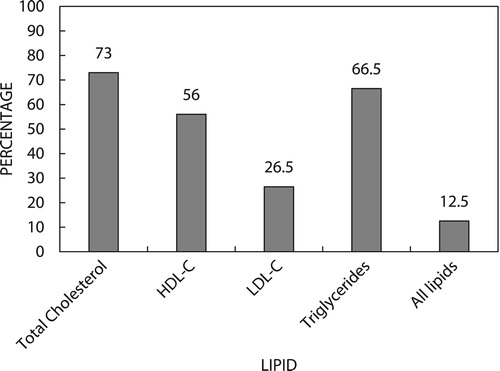
Results: Although the majority of participants (146 [73%]) were at the ideal level for total cholesterol, fewer (133 [66.5%]) were at the ideal level for triglycerides and 112 (56%) participants were at the ideal level for HDL cholesterol, only 53 (26.5%) participants were at target for LDL cholesterol, and very few, only 25 (12.5%), participants were at target for all four lipid parameters.
Conclusion: Higher doses of statins or the use of more potent statins with or without the addition of other lipid modifying drugs is recommended in order to achieve LDL cholesterol target in the majority of patients with type 2 diabetes.
Keywords:
Introduction
Type 2 diabetes mellitus is a metabolic disorder that results in persistently higher than normal serum glucose levels in untreated patients.Citation1 Long-term vascular complications are the cause of poor outcomes, such as death and disability,Citation2 with cardiovascular disease being the major cause.Citation3 Dyslipidaemia, which is an independent risk factor for cardiovascular disease, is present in the majority of patients with type 2 diabetes and affects all lipid fractions.Citation4
Elevated total and LDL cholesterol (LDL-C) as well as elevated levels of triglycerides together with low levels of HDL cholesterol (HDL-C) are found in the majority of patients with type 2 diabetes.Citation4
Reducing LDL-C levels confers significant protection against the higher risk of cardiovascular and cerebrovascular complications in dyslipidaemic patients.Citation4 A reduction in LDL-C by approximately 1 mmol/l decreases the five-year incidence of major coronary events by approximately 20%.Citation5
HDL-C levels are also used as a marker of cardiovascular disease risk with low levels being associated with a greater risk of cardiovascular disease, independent of LDL-C levels.Citation6,Citation7
In addition, high triglyceride levels in both fasted and non-fasted patients are also associated with a greater cardiovascular risk.Citation8
Treatment of diabetic dyslipidaemia with statins (or HMG CoA reductase inhibitors) has been shown to significantly reduce adverse cardiovascular events and these are therefore the drugs of choice in this setting.Citation2
Several statins with differing potencies are currently in use. For example in the Collaborative Atorvastatin Diabetes Study (CARDS) study a low dose of atorvastatin (10 mg daily) decreased LDL-C by 40% and triglycerides by 19%.Citation9 The Euro Aspire studies revealed a reduction in total cholesterol levels greater than 4.5 mmol/l from 94.5% to 46.2% attributable to statin therapy.Citation10
At maximal dose atorvastatin and rosuvastatin can potentially decrease LDL-C by as much as 60%.Citation5
Another class of lipid-lowering therapy called fibrates (or peroxisome proliferator-activated receptor-α agonists), in the form of the drug ezetimibe, may be necessary to reduce LDL-C, especially in patients at high risk for cardiovascular disease.Citation11 The role of other treatment options for this purpose is still under investigation.
The combined use of a statin with a fibrate may be more beneficial than statin therapy alone in patients with type 2 diabetes and dyslipidaemiaCitation12 but this has not been clearly demonstrated in cardiovascular outcome studies.Citation13
Globally, poor control of diabetic dyslipidaemia remains a problem and an unacceptably low proportion of patients with diabetes attain the currently recommended lipid targets.Citation10
Studies from countries such as Canada and the United States have demonstrated the difficulty of controlling dyslipidaemia, particularly LDL-C, in patients with diabetes.Citation14,Citation15 One contributory factor has been the underutilisation of high-intensity statins or high doses of statins.Citation16
The 2012 Society for Endocrinology, Metabolism and Diabetes of South Africa (SEMDSA) guidelines for the management of type 2 diabetes mellitus have allowed for the goal-directed management of diabetic dyslipidaemia. These were the current guidelines when this study was conducted.
According to the SEMDSA, the 2012 guidelinesCitation17 for lipid control in diabetes are as follows:
Total cholesterol < 4.5 mmol/l;
LDL-C < 1.8 mmol/l;*
HDL-C > 1.0 mmol/l (men) and > 1.2 mmol/l (women);
Triglycerides < 1.7 mmol/l.
* LDL-C goal is < 2.5 mmol/l in patients with diabetes who meet all of the following criteria:
No cardiovascular disease and no chronic kidney disease;
Less than 40 years old OR duration of diabetes less than 10 years;
No other cardiovascular risk factors.
Lipid guidelines are of benefit as they provide recommendations on treatment and lipid treatment targets. However, both local and international data have revealed an unexpected high prevalence of uncontrolled dyslipidaemia in diabetes. At the Charlotte Maxeke Johannesburg Academic Hospital Diabetic Clinic the introduction of Simvastatin 20 mg once daily, then up-titrating the dose or changing the intensity of the statin based on subsequent lipid profiles, has been the statin-prescribing strategy of choice. A study of the prevalence and control of diabetic dyslipidaemia, based on lipid-modifying therapy, was considered necessary to determine whether the current practices are adequate.
Methods and materials
Ethics clearance was obtained from the Human Research Ethics Committee at the University of the Witwatersrand. A retrospective observational study of patients with type 2 diabetes attending the diabetic clinic at the Charlotte Maxeke Johannesburg Academic Hospital was conducted. The only inclusion criterion was type 2 diabetes mellitus while subjects were excluded if they had new onset type 2 diabetes mellitus (< 3 months duration), type 1 diabetes mellitus, secondary diabetes, or secondary causes of dyslipidaemia. Participants were selected between March 1, 2017 and May 31, 2017.
A sample size of 200 was deemed to be sufficient for the purposes of this study.
Demographic information including age, gender, weight, height, duration of diabetes, smoking, established cardiovascular disease, chronic kidney disease, diabetic treatment, lipid-modifying therapy, and blood results for point-of-care HBA1C and formal lipograms (total cholesterol, triglycerides, HDL-C and LDL-C) were collected. The HBA1C and lipograms were documented only if they were done within 1 month of each other and if both had been done within the preceding 12 months.
A descriptive analysis was conducted with summary measures such as mean and standard deviation (SD) for age, gender, weight, duration of diabetes, hypertension, smoking status, presence of cardiovascular disease, chronic kidney disease (eGFR < 30 ml/min/m2), HBA1C and the different lipid parameters (total cholesterol, triglycerides, HDL-C and LDL-C). Frequency tables of patient use of medication for hyperglycaemia (oral hypoglycaemic agents/insulin), hypertension, and dyslipidaemia (no statin, low, moderate, and high-intensity statin therapy, dose, and other lipid-modifying therapy) were also produced. High-intensity statin therapy was considered to be daily doses of atorvastatin 40 mg, atorvastatin 60 mg, atorvastatin 80 mg, rosuvastatin 20 mg, or rosuvastatin 40 mg.
The percentage of patients achieving SEMDSA ideal lipid levels or targets was calculated according to the type and dose of lipid-modifying therapy used. Correlations between glycaemic control, lipid levels and intensity/dose of lipid modifying therapy were also assessed. A p-value of < 0.05 was considered significant.
Results
Characteristics of the study group
This cohort comprised 200 participants with a mean age of 58.9 years (SD ± 11.04) with 43% being male and 57% female. The duration of diabetes was more than 10 years in the majority (53%). Additional cardiovascular risk factors were common, namely hypertension (81%), excessive weight (20.5%) and smoking (12%). The lack of height measurements precluded calculating body mass index so a weight of more 100 kg was deemed to be overweight in this population. We acknowledge that an arbitrary weight of 100 kg is a limitation as a few participants may have had a normal body mass index. The demographics of the study cohort are shown in .
Table 1: Characteristics of the study group
Description of the lipid profile
The lipid profiles are given in . Each lipid parameter had a wide range; however, the median for each, with the exception of LDL-C, reflected good control.
Table 2: Description of the lipid profile
Three of the four lipid parameters were controlled for most participants; however, LDL-C was not at target and only a small percentage had all four lipid parameters controlled ().
The median LDL-C was 2.21 mmol/l (IQR 1.13 mmol/l). Fifty-three participants had LDL-C at target. Participants requiring high-intensity statins had a median LDL-C of 2.34 mmol/l (IQR 1.37 mmol/l).
A total of 198 (99%) participants required a target LDL-C of 1.8 mmol/l and 2 participants (1%) required a target LDL-C of 2.5 mmol/l according to their individual risk factors.
Irrespective of a target level, 126 (63%) participants had a LDL-C of less than 2.5 mmol/l.
Lipid-modifying therapy
Among all participants, 83% were on lipid-modifying therapy while 17% were not. Only two types of statin, simvastatin and atorvastatin, were prescribed but at various dosages. In addition to statins a fibrate was prescribed in only 2% of patients (Supplementary table 1). Of those on statin therapy, the majority were on low-intensity statins ().
Table 3: Statin therapy according to intensity
The mean total cholesterol in participants not on statin therapy was 4.45 mmol/l compared with a mean of 3.96 mmol/l in all participants on statin therapy indicating a significant difference (p = 0.006), most likely attributable to statin therapy. In this regard the mean was used as total cholesterol was distributed normally. Supplementary table 2 shows the means of each lipid parameter according to statin intensity.
Glycated haemoglobin (HBA1C) and the lipid profile
The range of HBA1C was from 5.0% to 17.3% with a median of 8.3% (IQR = 3.2%).
shows how the HBA1C correlated with the lipid profile. The HBA1C showed a weak correlation with total cholesterol, LDL-C and HDL-C. It did, however, show a statistically significant positive correlation with triglycerides. Lower LDL-C levels were also associated with lower HBA1C levels (p = 0.007).
Table 4: Correlation of HBA1C and lipid profile.
Poor diabetic control therefore had a minor influence on lipid levels.
and describe the HBA1C in more detail according to gender and body weight.
Table 5: Breakdown of HBA1C
Table 6: Weight ≥ 100kg, lipid profile and HBA1C
HBA1C < 7% and the lipid profile
shows the correlation between HBA1C < 7% and the lipid profile.
HBA1C levels of less than 7% did not influence total cholesterol, LDL-C and HDL-C. However, good glycaemic control to an HBA1C of less than 7% was associated with lower triglyceride levels. With regard to having all four lipid parameters at target, an HBA1C of less than 7% showed no correlation.
Discussion
Overall, the results of this study reveal a disappointingly high prevalence of poorly treated diabetic dyslipidaemia in type 2 diabetics. The long-term cerebrovascular, cardiovascular and peripheral vascular complications in this population are potentially preventable with adequate lipid control. The study is a reflection of the global prevalence of uncontrolled diabetic dyslipidaemia. A study in China demonstrated that 54.9%, 43.7% and 37.4% of participants were not at goal (according to local practice guidelines) for LDL-C, triglycerides and HDL-C respectively.Citation18 Similar rates of prevalence have been reported in the CanadianCitation14 and GermanCitation19 arms of the DYSlipidemia International Study.
In the South African context, the prevalence of uncontrolled dyslipidaemia is reflected in studies done in the private sector only,Citation20 and in both the public and private sectors.Citation21,Citation22 In these studies, the prevalence of uncontrolled LDL-C ranged from 47.7% to 58.6%.
In this cohort, according to the SEMDSA guidelines, total cholesterol, triglycerides and HDL-C were controlled in more than half the population. However, only about a quarter of the participants had LDL-C at target. Of note, only one in eight patients have all lipid parameters controlled. Ideally, all four parameters should be at target or the ideal level to minimise the risk of vascular complications.Citation4 The strategy by which statin therapy was used in this study therefore did not result in optimal control of diabetic dyslipidaemia.
Participants on low-intensity statins have lipid profiles closest to the guidelines. However, this may have been the result of the initiation of low-intensity statin therapy because of only mild hyperlipidaemia in these subjects. The participants not on a statin have lipid profiles similar to the patients on medium- and high-intensity statins. All levels of statin intensity failed to adequately control levels of LDL-C. Participants in whom high-intensity statin therapy was prescribed were more likely those with severe dyslipidaemia. These participants were, however, not at goal for their lipid targets and were not receiving supplementary lipid-lowering therapy, revealing a need for additional lipid-lowering therapy such as ezetimibe.
On average, participants not on statin therapy did not have significantly different triglyceride levels compared with those on statin therapy. However, this finding does not take into consideration the state of glycaemic control in these participants. Participants who were not on statin therapy have comparable levels of HDL-C to those on statin therapy. The benefit of statin therapy is reflected in the significantly lower LDL-C levels in statin-treated participants compared with participants not treated with statins. Despite the suboptimal prevalence of controlled LDL-C, statin therapy should have a positive impact in preventing cardiovascular complications by reducing LDL-C.Citation9
In this study an HBA1C of more than 7%, which may be considered to be poorly controlled diabetes mellitus, was associated with worse triglyceride levels, as the only difference when compared with an HBA1C of less than 7%. This association was also reported in a study directed specifically towards the relationship between glycaemic control and the serum lipid profile in type 2 diabetic patients,Citation23 and in a large scale Malaysian study of more than 28 000 participants.Citation24 These findings highlight the point that good glycaemic control is an important contributor to optimising lipid levels. This may obviate the need for escalating to more potent statins or adding other classes of lipid-lowering therapy, a benefit that would be appreciated in a resource-constrained setting. One may even consider a six-month trial period to optimise glycaemic control before modifying or up-titrating lipid-lowering therapy.
A limitation of the study was the timing of serum analysis. Not all participants had their lipograms and HBA1C done on the same day. Some participants had their lipograms done before their diabetic clinic appointments. Lipograms and HBA1C were paired only if they were done within 30 days of each other. It is possible that either or both of the lipid and HBA1C readings might have been slightly different had they been done on the same day.
However, in this study the selection of lipid-lowering therapy was suboptimal and compounded by a lack of appropriate dose titration. Various correctable factors may be postulated for the poor control of diabetic dyslipidaemia so that effective practical solutions can be enforced. The ability to choose and successfully titrate therapy may be influenced by the clinician’s knowledge of a statin’s potency, maximal dosage, need to escalate to a more potent agent or add another agent, and the experience of the clinician in this regard.Citation14 Trends towards Westernised diets and sedentary lifestyles present obstacles to good glycaemic and lipid control. Concerns for safety, toxicity and side effects, as well as ethnicity and lower socio-economic status, have been cited as patient factors for poor statin adherence.Citation20–22 In the public sector the lack of drug availability, due to formulary restrictions and at times stock shortage, is a common problem while staff shortage in large diabetic clinics impairs the holistic management of diabetic dyslipidaemia.
Supplemental Material
Supplemental data for this article can be accessed at https://doi.org/10.1080/16089677.2019.1686869
Supplemental Material
Download PDF (17.5 KB)Disclosure statement
No potential conflict of interest was reported by the authors.
References
- Powers AC. Diabetes mellitus. In: Longo DL, Fauci AS, Kasper DL, Hauser SL, editors. Harrison’s principles of internal medicine. 18th ed. New York: McGraw-Hill; 2012. p. 2968–3003.
- Baigent C, Keech A, Kearney PM, et al. Efficacy and safety of cholesterol lowering treatment: prospective meta-analysis of data from 90,056 participants in 14 randomised trials of statins. Lancet [Internet]. 2005 Oct;366:1267–78. [cited 2016 Jun 10] Available from: http://www.thelancet.com/pdfs/journals/lancet/PIIS0140-6736(05)67394-1.pdf. doi: 10.1016/S0140-6736(05)67394-1
- Taskinen M, Boren J. New insights into the pathophysiology of dyslipidemia in type 2 diabetes. Atherosclerosis [Internet]. 2015 Apr;239:483–95. [cited 2016 Jan 16] Available from: http://www.atherosclerosis-journal.com/article/S0021-9150(15)00088-X/fulltext. doi: 10.1016/j.atherosclerosis.2015.01.039
- Reiner Z, Catapano AL, De Backer G, et al. ESC/EAS guidelines for the management of dyslipidaemias: the task force for the management of dyslipidaemias of the European Society of Cardiology (ESC) and the European Atherosclerosis Society (EAS). Eur Heart J. 2011 Jul;32:1769–818. [cited 2016 Jan 23]; Available from: http://eurheartj.oxfordjournals.org/content/32/14/1769.long. doi: 10.1093/eurheartj/ehr158
- Bramlage P, Lanzinger S, Rathmann W, et al. Dyslipidaemia and its treatment in patients with type 2 diabetes: A joint analysis of the German DIVE and DPV registries. Diabetes Obes Metab. 2016 Sep;19(1):61–9. [cited 2017 Apr 19] Available from: http://onlinelibrary.wiley.com/doi/10.1111/dom.12783/full. doi: 10.1111/dom.12783
- Gordon T, Castelli WP, Hjortland MC, et al. High density lipoprotein as a protective factor against coronary heart disease. the framingham study. Am J Med. 1977 May;62:707–14. [cited 2016 Jan 24]; Available from: http://www.sciencedirect.com/science/article/pii/0002934377908749. doi: 10.1016/0002-9343(77)90874-9
- Assmann G, Schulte H, Cullen P, et al. Assessing risk of myocardial infarction and stroke: new data from the Prospective cardiovascular münster (PROCAM) study. Eur J Clin Invest. 2007 Dec;37:925–32. [cited 2017 Apr 28]; Available from: http://onlinelibrary.wiley.com/doi/10.1111/j.1365-2362.2007.01888.x/epdf. doi: 10.1111/j.1365-2362.2007.01888.x
- Sarwar N, Danesh J, Eiriksdottir G, et al. Triglycerides and the risk of coronary heart disease 10,158 incident cases among 262,525 participants in 29 Western prospective studies. Circulation. 2007 Jul;115(4):450–8. [cited 2017 Apr 30] Available from: http://circ.ahajournals.org/content/115/4/450. doi: 10.1161/CIRCULATIONAHA.106.637793
- Colhoun HM, Betteridge DJ, Durrington PN, et al. Primary prevention of cardiovascular disease with atorvastatin in type 2 diabetes in the collaborative atorvastatin diabetes study (CARDS): multicentre randomised placebo-controlled trial. Lancet. 2004 Aug;364(9435):685–96. [cited 2017 Jan 29] Available from: http://0-search.proquest.com.innopac.wits.ac.za/docview/199004507/fulltextPDF/C836D43AB6AC4227PQ/31?accountid=15083. doi: 10.1016/S0140-6736(04)16895-5
- Pérez A, Blanco CG, Hernández-Presa MA, et al. Therapeutic approach to dyslipidemia and goal achievement in a Spanish population with type 2 diabetes without cardiovascular disease. Endocrinol Nutr. 2011 Mar;58:289–90. [cited 2016 Jan 18] Available from: http://www.sciencedirect.com/science/article/pii/S2173509311700617/pdf?md5=e955d90a7e671a70f3c93c571f1a3cb8&pid=1-s2.0-S2173509311700617-main.pdf. doi: 10.1016/j.endonu.2011.03.005
- Fruchart JC, Sacks FM, Hermans MP, et al. The residual risk reduction initiative: a call to action to reduce residual vascular risk in dyslipidaemic patient. Diab Vasc Dis Res. 2008 Nov;5:319–35. [cited 2017 Jan 29]; Available from: http://journals.sagepub.com/doi/pdf/10.3132/dvdr.2008.046.
- Krauss RM. Lipids and lipoproteins in patients with type 2 diabetes. Diabetes Care. 2004 Jun;27(6):1496–504. [cited 2017 Apr 22] Available from: http://care.diabetesjournals.org/content/27/6/1496.full-text.pdf. doi: 10.2337/diacare.27.6.1496
- Goff Jr DC, Gerstein HC, Ginsberg HN, et al. Prevention of cardiovascular disease in persons with type 2 diabetes mellitus: current knowledge and rationale for the action to control cardiovascular risk in diabetes (ACCORD) trial. Am J Cardiol. 2007 Jun;99(12):S21–S33. [cited 2017 Nov 15] Available from: https://doi.org/10.1016/j.amjcard.2007.03.002.
- Goodman SG, Langer A, Bastian NR, et al. Prevalence of dyslipidemia in statin-treated patients in Canada: results of the DYSlipidemia international study (DYSIS). Can J Cardiol. 2010 Nov;26:330–5. [cited 2016 Jan 17] Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2989357/. doi: 10.1016/S0828-282X(10)70454-2
- Kennedy AG, MacLean CD, Littenberg B, et al. The challenge of achieving national cholesterol goals in patients with diabetes. Diabetes Care. 2005 May;28(5):1029–34. [cited 2016 Jan 25]; Available from: http://care.diabetesjournals.org/. doi: 10.2337/diacare.28.5.1029
- Leiter LA, Lundman P, da Silva PM, et al. Persistent lipid abnormalities in statin-treated patients with diabetes mellitus in Europe and Canada: results of the dyslipidaemia international study. Diabet Med. 2011 Jun;28:1343–51. [cited 2016 Jan 24]Available from: http://onlinelibrary.wiley.com/doi/10.1111/j.1464-5491.2011.03360.x/epdf. doi: 10.1111/j.1464-5491.2011.03360.x
- Amod A, Ascott-Evans BH, Berg GI, et al. The 2012 SEMDSA guidelines for the management of type 2 diabetes (revised). JEMDSA. 2012 Aug;17(2):S1–S95. [cited 2016 May 13] Available from: http://www.semdsa.org.za/images/2012_SEMDSA_Guideline_July_FINAL.pdf.
- Zhao S, Wang Y, Mu Y, et al. Prevalence of dyslipidaemia in patients treated with lipid-lowering agents in China: results of the DYSlipidemia international study (DYSIS). Atherosclerosis. 2014 Aug;235:463–9. [cited 2016 Nov 12]; Available from: https://www.sciencedirect.com/science/article/pii/S0021915014011666. doi: 10.1016/j.atherosclerosis.2014.05.916
- Gitt AK, Jünger C, Smolka W, et al. Prevalence and overlap of different lipid abnormalities in statin-treated patients at high cardiovascular risk in clinical practice in Germany. Clin Res Cardiol. 2010 Nov;99:723–33. [cited 2016 Jan 23] Available from: http://link.springer.com/article/10.1007%2Fs00392-010-0177-z. doi: 10.1007/s00392-010-0177-z
- Raal F, Schamroth C, Blom D, et al. CEPHEUS SA: a South African survey on the under-treatment of hypercholesterolaemia. Cardiovasc J Afr. 2011 Oct;22(5):234–40. [cited 2019 Oct 15]; Available from: https://0-www-ncbi-nlm-nih-gov.innopac.wits.ac.za/pmc/articles/PMC3721857/. doi: 10.5830/CVJA-2011-044
- Blom DJ, Raal F, Amod A, et al. Management of low-density lipoprotein cholesterol levels in South Africa: the international ChoLesterol management Practice study (ICLPS). Cardiovasc J Afr. 2019 Jan;30(1):15–23. [cited 2019 Oct 15]; Available from: http://0-resolver.ebscohost.com.innopac.wits.ac.za/openurl?sid=Entrez%3aPubMed&id=pmid%3a30720848&site=ftf-live. doi: 10.5830/CVJA-2018-054
- Raal FJ, Blom D, Naidoo S, et al. Prevalence of dyslipidaemia in statin-treated patients in South Africa: results of the DYSlipidaemia international study (DYSIS). Cardiovasc J Afr. 2013 Sep;24(8):330–8. [cited 2019 Oct 15] Available from: https://0-www-ncbi-nlm-nih-gov.innopac.wits.ac.za/pmc/articles/PMC3821092/pdf/cvja-24-330.pdf. doi: 10.5830/CVJA-2013-071
- Mahato RV, Gyawali P, Raut PP, et al. Association between glycaemic control and serum lipid profile in type 2 diabetic patients: glycated haemoglobin as a dual biomarker. Biomed Res. 2011 Mar;22(3):375–80. [cited 2017 Apr 22]; Available from: http://www.alliedacademies.org/articles/association-between-glycaemic-control-and-serum-lipid-profile-in-type-2-diabetic-patients-glycated-haemoglobin-as-a-dual-biomarker.pdf.
- Chew BH, Ismail M, Lee PY, et al. Determinants of uncontrolled dyslipidaemia among adult type 2 diabetes in Malaysia: the Malaysian diabetes registry 2009. Diabetes Res Clin Pract. 2012 Jun;96:339–47. [cited 2016 Jan 27] Available from: http://0-www.sciencedirect.com.innopac.wits.ac.za/science/article/pii/S0168822712000393. doi: 10.1016/j.diabres.2012.01.017