Chronic heart failure is characterized by exaggerated sympathoexcitation in both human patients and animal models. Despite major advances in therapy, the increased neurohumoral drive causes significant cardiovascular complications that contribute to increased morbidity and mortality. Blunted GABAergic inhibition in the hypothalamic paraventricular nucleus (PVN) has been suggested as a key integrating mechanism of the sympathoexcitation associated with cardiovascular-related disorders such as hypertension, diabetes, and heart failure. The GABAA receptor (GABAAR), a pentameric ligand-gated Cl− channel, mediates 2 inhibitory modalities in the PVN: a conventional inhibitory synaptic current (IPSC) mediated by synaptic GABAARs, and a persistent tonic inhibitory current (termed Itonic) generated by extrasynaptic GABAARs. As in other brain regions, Itonic mediates the dominant portion of GABAAR-mediated inhibition and thus has a major impact on PVN neurons projecting to the rostral ventrolateral medulla (PVN-RVLM) neuronal excitability.Citation1 However, the pathophysiological significance of Itonic in sympathoexcitation remains poorly understood. In a recent study using brain slice patch-clamping,Citation2 Sudip and colleagues showed that Itonic, defined as the holding current shift by the GABAAR antagonist bicuculline, was attenuated in the PVN-RVLM in rats with myocardial infarction (MI)-induced heart failure (HF). The authors suggested that this deficit in GABAergic tonic inhibition of the pre-sympathetic neurons and the resulting increased sympathetic outflow from the PVN during HF is attributable to enhanced astroglial GABA uptake.
Itonic generated by activation of extrasynaptic GABAARs is tightly controlled by extracellular GABA concentration as well as the expression and combination of extrasynaptic GABAARs in specific brain regions. In addition to vesicular GABA release responsible for activating IPSCs,Citation1,3 GABA released from glia contributes to Itonic generation in the brain. GABA transporter (GAT) reversal has been suggested in pathological conditions, while a debated role for the GABA-releasing anion channel bestrophin-1 (Best-1) has been proposed to explain physiological and pathophysiological GABA release from glia. Despite evidence for GAT reversal under pathophysiological conditions, GATs are generally believed to uptake extracellular GABA into cells. Of the 4 GATs (GAT-1, GAT-2, GAT-3, and BGT-1), GAT-1 and GAT-3 are most likely to be expressed in neurons and glia, respectively, and to be responsible for ambient GABA levels in the brain (Fig. 1).Citation4 Pharmacological or genetic inhibition of GAT-1 increases Itonic, which is associated with neurological and psychological disorders. However, the relationship between astroglial GABA clearance by GAT-3 and its effects on Itonic modulation and neuronal activity under pathological conditions are poorly understood. Sudip and colleagues showed that HF Itonic attenuation was reversed by a nonselective GAT blocker (nipecotic acid, NPA) and a GAT-3 selective blocker (SNAP-5114), but not by a GAT-1 blocker (NO-711), suggesting that astroglial GABA uptake plays a major role in Itonic regulation of HF in PVN-RVLM neurons,Citation2 as in the naïve PVN.Citation5 To exclude the involvement of BEST-1-mediated GABA release in HF Itonic attenuation, Sudip et al. showed that BEST-1 blockade did not affect Itonic in either sham-operated or post-MI rats.
Given that Itonic amplitude correlates with vesicular GABA release, HF Itonic attenuation may result from reduced ambient GABA concentrations related to a decrease in IPSC frequency in HF PVN-RVLM neurons.Citation6 Collectively, the finding that GAT blockers mask and reverse HF Itonic attenuation suggests that blockade of enhanced GAT activity could compensate and even overpower impaired vesicular GABA release in HF PVN-RVLM neurons. Using pharmacological probes, Sudip and colleagues also investigated possible changes in extrasynaptic GABAAR function in HF. Reduced Itonic sensitivity to THIP (4,5,6,7-tetrahydroisothiazolo-[5,4-c]pyridin-3-ol) supported decreased function of GABAAR δ subunits in HF, whereas similar Itonic sensitivity to benzodiazepines indicated that γ2 subunit-containing GABAARs do not differ between sham-operated and post-MI rats. Thus, despite reduced GABAAR δ subunit function, the increased impact on GABAAR γ2 subunits mediating Itonic may enable GAT blockade to reverse Itonic attenuation in HF PVN-RVLM neurons.
Sudip et al. found that Itonic attenuation increased membrane input resistance (IR) and firing discharge rate in HF PVN-RVLM neurons, indicating that Itonic, as the dominant portion of GABAAR-mediated inhibition, has a major impact on PVN-RVLM neuronal excitability.Citation1 The direct impact of Itonic on membrane IR, and thus the membrane time constant, may affect synaptic efficacy and integration in neurons.Citation7 Accordingly, Sudip and colleagues observed a leftward shift in the input-output (I-O) function of HF PVN-RVLM neurons, reversed by NPA, suggesting that Itonic attenuation significantly impacts neuronal sensitivity to incoming excitatory and/or inhibitory synaptic inputs in the HF PVN-RVLM. Therefore, the increased impact on membrane IR and the I-O function would enable GAT blockade to correct the altered synaptic efficacy and integration in HF PVN-RVLM neurons. This conclusion is further supported by the finding that NPA efficiently inhibits the increased spontaneous firing in HF PVN-RVLM neurons.
In conclusion, Sudip and colleagues showed that enhanced astroglial GABA uptake attenuates Itonic and, in turn, increases neuronal firing of pre-sympathetic PVN neurons in heart failure. The data demonstrate a link between pathophysiology and GAT-3 uptake modulation of GABAAR tonic inhibition in the brain during altered autonomic nerve activity and highlight the potential of targeting astroglial GABA clearance to reduce sympathoexcitation associated with cardiovascular disorders.
Disclosure of Potential Conflicts of Interest
No potential conflicts of interest were disclosed.
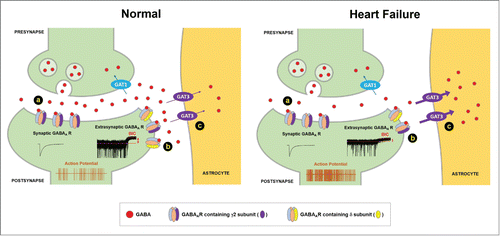
Figure 1. Regulation of GABAA tonic inhibition of the pre-sympathetic PVN neurons in normal rats and following heart failure. Combined with decreased vesicular GABA release (A) and reduced function of extrasynaptic GABAARs containing δ subunits (B), enhanced astroglial GABA clearance via GAT-3 (C) attenuates GABAA tonic inhibition and increases neuronal firing in heart failure.
References
- Park JB, et al. The Journal of physiology 2007; 582:539-51; PMID:17495040; http://dx.doi.org/10.1113/jphysiol.2007.133223.
- Pandit S, et al. Journal of neurophysiology 2015; 114(2):914–26; PMID:26063771; http://dx.doi.org/10.1152/jn.00080.2015
- Glykys J, et al. The Journal of physiology 2007; 582:1163-78; PMID:17525114; http://dx.doi.org/10.1113/jphysiol.2007.134460.
- Dalby NO. Eur J Pharmacol 2003; 479:127; PMID:14612144; http://dx.doi.org/10.1016/j.ejphar.2003.08.063.
- Park JB, et al. The Journal of physiology 2009; 587:4645-60; PMID:19703969; http://dx.doi.org/10.1113/jphysiol.2009.173435.
- Han TH, et al. American journal of physiology Regulatory, integrative and comparative physiology 2010; 299:R129-39; PMID:20164200; http://dx.doi.org/10.1152/ajpregu.00391.2009.
- Semyanov A, et al. Trends Neurosci 2004; 27:262-9; PMID:15111008; http://dx.doi.org/10.1016/j.tins.2004.03.005.
