Optimized Oil red O protocol
pp. 351–358
While the degree of adipocyte differentiation can be assessed through Oil red O, it is not clear whether this method of staining is valid for quantitative analysis. Authors Kraus et al. provide an optimized protocol for the use of oil red O staining after displaying that previous methods are unfit for quantitative analysis. This new protocol is shown to be culture plate independent, and in this research paper the authors propose that it is a good candidate for universal adaptation with regards to the quantitative analysis of adipocyte differentiation.
Modulating effects of branched short-chain fatty acids
pp. 359–368
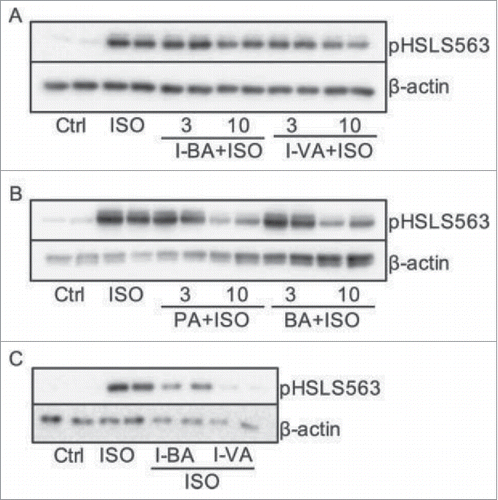
The anti-obesity properties of short-chain fatty acids (SCFAs) generated through the breakdown of dietary fibers are known, however the relation between branched SCFAs (BSCFAs) and adipocyte metabolism has not been extensively explored. In this research paper, authors Heimann et al. investigate the effect of BSCFAs, specifically isobutyric and isovaleric acid, on glucose and lipid metabolism in adipocytes. Their findings presented here show improved insulin sensitivity through BSCFAs effects on glucose and lipid metabolism in adipocytes ().
The trafficking of adipose stromal cells to tumors
pp. 369–374
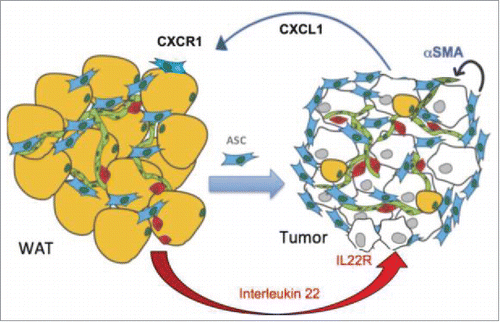
Adipose stromal cells (ASC) are known to promote cancer progression through the secretion of adipokines with pro-tumorigenic properties. While it is still unclear how ASC is trafficked, authors Zhang and Kolonin previously showed that interleukin (IL)-22 may be a mechanism of CXCL1 activation, which chemoattracts ASC in obesity. In this brief report, the authors present evidence showing IL-22R expression by tumors as well as IL-22 expression in white adipose tissue infiltrated leukocytes are obesity-dependent. The recruitment of these IL-22 expressing leukocytes caused by obesity-associated adipocyte death may be what triggers IL-22 signaling, resulting in ASC trafficking to tumors via CXCL1 secretion by cancer cells ().
Protective effects of subcutaneous abdominal fat
pp. 375–377
Although several studies have shown visceral adipose tissue as being a contributor to metabolic risk, research has also been presented supporting the idea that subcutaneous fat may be associated with favorable glucose and lipid profiles, and, also could protect from ailments such as atherosclerosis. This brief report by Niijima et al. compares fat area to insulin sensitivity in order to determine a relative range of subcutaneous and visceral abdominal fat area which may have clinical significance. Their research shows that in subjects with visceral fat area above 1002cm, increased subcutaneous abdominal fat area may have a protective effect and is a predictor of insulin sensitivity.
Retinoic acid levels in adipose tissue
pp. 378–383
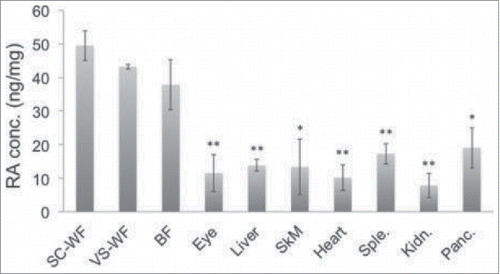
The vitamin A metabolite known as retinoic acid (RA) is known to be essential for stem cell differentiation, however RA's role in adipose tissue is not clear. In this brief report, authors Perumal, Sriram, Lim, Olivo and Sugii continue to look at the contributions of RA in adipose tissue, having previously shown how genes in stem cells may be differentially expressed depending on whether they are derived from visceral fat or subcutaneous fat. Using surface-enhanced Raman spectroscopy (SERS), the authors measured and analyzed tissue RA levels, showing that after a high fat diet, mouse adipose tissue contained significantly more endogenous RA when compared to other tissues ().
Inflammatory effects of HMGB1 explored
pp. 384–388
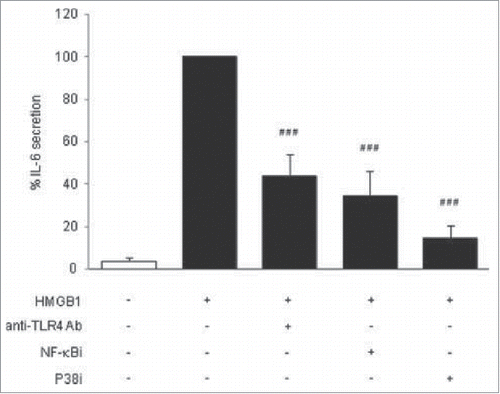
Authors Gunasekaran et al. investigate the role of high mobility group box protein 1 (HMGB1) on adipocyte inflammation in this brief report, an aspect not well known despite HMGB1's pro-inflammatory effects in obesity-related pathologies. When under stress, HMGB1 is known to promote inflammation and findings have shown that in obese subjects, the protein expression of HMGB1 is elevated. Here, the authors show that HMGB1 does have pro-inflammatory effects on adipocytes and can initiate inflammation dependent on toll-like receptor 4 (TLR4), leading to an increase in cytokine IL-6 secretion ().