ABSTRACT
The reemergence of smallpox as a bioterrorism attack is now an increasing and legitimate concern. Advances in synthetic biology have now made it possible for the virus to be synthesized in a laboratory, with methods publicly available. Smallpox introduction into a susceptible population, with increased immunosuppression and an aging population, raises questions of how vaccination should be used in an epidemic situation when supply may be limited. We constructed three modified susceptible-latent-infectious-recovered (SEIR) models to simulate targeted, ring and mass vaccination in response to a smallpox outbreak in Sydney, Australia. We used age-specific distributions of susceptibility, infectivity, contact rates, and tested outputs under different assumptions. The number of doses needed of second- and third-generation vaccines are estimated, along with the total number of deaths at the end of the epidemic. We found a faster response is the key and ring vaccination of traced contacts is the most effective strategy and requires a smaller number of doses. However if public health authorities are unable to trace a high proportion of contacts, mass vaccination with at least 125,000 doses delivered per day is required. This study informs a better preparedness and response planning for vaccination in a case of a smallpox outbreak in a setting such as Sydney.
KEYWORDS:
Introduction
The eradication of smallpox in 1980 was a triumph of public health. The first-generation smallpox vaccines using vaccinia virus, such as DryVax, were used for the World Health Organization (WHO) eradication program.Citation1 Mass vaccination was the primary strategy of choice during the era of endemic smallpox. However, when demand exceeded supply and achieving mass vaccination in India became a challenge, other more efficient vaccination approaches were considered. Ring vaccination, which combines surveillance and containment with vaccination of close contacts was the strategy used in the final phases of eradication.Citation2
The reemergence of smallpox as a bioterrorism attack is now an increasing and legitimate concern. Advances in synthetic biology have now made it possible for the virus to be synthesized in a laboratory,Citation3 with methods publicly available.Citation4 By 1980 first-generation smallpox vaccines ceased to be produced, and research subsequently began on second-generation vaccines produced via tissue culture to comply with modern manufacturing guidelines.Citation2 Third-generation replication deficient vaccines such as Imvamune (JYNNEOSTM) reduce risk of adverse events and can be used in people who are immunosuppressed.Citation2 Current stockpiles are mostly second generation vaccines,Citation5 with potential to cause serious adverse events.Citation2 Smallpox vaccine comprises a major proportion of the US Center for Disease Control (CDC) strategic national stockpile believed to be the largest national stockpile in the world.Citation6
Emergency response plans for most countries recommend the ring vaccination strategy and if this has been ineffective or if there are multiple sites of deliberate release of a smallpox, switching to mass vaccination strategy, while others suggest commencing with mass vaccination within 24 hours of confirmation of smallpox outbreak.Citation7,Citation8
The choice of vaccination strategy could differ depending on parameters such as initial size of attack, relevant efficiency of control measures, vaccine supplies, vaccine program capacity, level of residual vaccine-induced immunity, and local population and geographic factors.Citation9
Daniel Bernoulli’s 18th century model on smallpox was then refined by Kermack and McKendrick in the 20th century,Citation10 and it is now the most widely used category of models, the compartmental deterministic SIR (Susceptible-Infected-Recovered) models. Since then mathematical models have increasingly been used to understand transmission dynamics, forecasting epidemics and to assess the impact of intervention strategies.Citation11-13 The risk of infectious disease is dependent on characteristics of the infectious host, the organism, the susceptible host, and the environment,Citation14 however the traditional deterministic compartmental models usually assume homogeneous mixing with same probability of infection for everyone.Citation15
Studies assessing the impact of pharmaceutical and nonpharmaceutical interventions using modelling approaches have contributed to the evidence for control of smallpox outbreaks.Citation16 Several studies have compared ring and mass vaccination strategies and possible benefits of prior levels of herd immunity combined with control measures.Citation17-22 Some studies favor isolation of infected individuals and ring vaccination as the optimal strategy provided there are no resource constraints.Citation17,Citation19 In the presence of a large number of cases or higher reproduction number, mass vaccination may be a preferred option of outbreak control.Citation8,Citation23 Mass vaccination in a large scale outbreak may prevent more deaths than ring vaccination in resource-constrained settings where contact tracing cannot be conducted adequately, while the ring approach could be more suitable for the initial and final phase of an outbreak.Citation8 Combination strategies such as isolation along with targeted vaccination also may be appropriate in a small scale smallpox outbreak.Citation24-27
Smallpox introduction into a susceptible aged population, with increased immunosuppression could result in a 40% death rate.Citation28 In addition, different mixing patterns and immunosuppression lead to varying age specific transmission of disease and case fatality rates, with the highest transmitters in people aged 5–19 years, but the highest fatality rate in adults >65 years.Citation28 This raises questions of how vaccination should be used in an epidemic situation when supply may be limited and risk groups need to be prioritized.
In this study we focus on heterogeneity in host contact rates. While modelling studies focused on age structures and heterogeneous contact rates are common, studies on smallpox transmission that used a heterogeneous age-structure, mixing contacts and different susceptibility and infectivity levels as it is done in this study are less common. The aim of this study was to compare different vaccination strategies (mass, targeted and ring) for control of a smallpox outbreak in a highly populous, high-income city, Sydney (Australia), with high levels of contacts, elderly, and immunosuppressed.
Methods
The study used a previously developed model which simulated an epidemic of smallpox in Sydney, Australia.Citation28 Here we included three different outbreak responses to test different vaccination program options, under varying attack scenarios, for effectiveness of epidemic control and estimation of needed resources. Results are shown as incidence, deaths, and number of vaccine doses needed for each scenario tested.
Population assumptions and parametrization
The population of Sydney was used for this study, estimated to be 5.25 million in 2016.Citation29 The population distribution of New South Wales (NSW) in 2016Citation30 was used to stratify the Sydney populationCitation29 into 18 age groups, 5-year wide, up to 80–84, and then combined for 85+ years old. Each age group was further divided into six immunity levels: healthy, mild and severe immunocompromised, vaccinated and not, and healthcare workers (HCW).Citation28 This was the basis of tested age-based targeted vaccination strategies (see below).
We categorized smallpox disease into four different types defined by infectivity (R0) and case fatality rates (CFR): hemorrhagic, flat, ordinary and vaccine modified. Estimation of residual immunity and probabilities of developing each disease type are age and immunological status dependent.Citation28 Because each smallpox type has a different infectivity, related to viral shedding and virus excretion,Citation31 we estimated a different probability of infection per contact β for each smallpox type in order to reproduce an R0 equal to 10 for hemorrhagic and flat, 7.96 for ordinary and 5.3 for modified smallpox. Because more severe cases are likely to stay at home from the first day of symptoms, we halved the contacts number for hemorrhagic and flat subtypes for the entire infectious period, while for ordinary and modified, the contacts number was considered halved after 2 and 3 days respectively from the appearance of the rash.Citation28,Citation32
Finally, we multiplied the force of infection by a parameter (α1, α2, α3, α4) to account for different population susceptibility levels. We did not consider births and deaths because the models run only for 300 days, which is not enough to have an impact from yearly birth and death rates. We also estimated the number of doses of non-replicating vaccine required for each comparative strategy using our previous minimal estimate of 17% of people living with immunosuppression in Sydney.Citation28 In comparing strategies, we also compared the expected occurrence of serious adverse events and deaths from vaccination, based on data from the United States,Citation2 estimated to be 14 and 1.1 per million, respectively. Each vaccination strategy starts at the same time of isolation of infectious cases. In a setting like Sydney, we assumed that 95%Citation1 of the new daily infectious cases will be hospitalized and once isolated we assume zero transmissions.Citation1 CFRs are based on expected distribution of hemorrhagic, flat, ordinary and modified smallpox.
Vaccine effectiveness has been shown to be reduced in persons already infected and in a latent state.Citation1,Citation33 For uninfected people, we assumed the vaccine to be 95% and 98% effective for never vaccinated, and previously vaccinated, respectively.Citation1,Citation34 For already exposed, latently infected people, studies describe a range of vaccine effectiveness, from 30%Citation1,Citation34,Citation35 to 80%.Citation36 We considered it to be 50% effective in latent infected as found in studies testing ACAM2000, which is assumed to be the predominant vaccine that would be used in Australia.Citation6,Citation37
In a pandemic emergency setting, the personnel to delivery vaccinations will be the authorized nurse, midwife, pharmacists and general practitioners (GPs); however, medical and nurse students can be authorized immunizer after appropriate vaccination training (provided by Health Protection New South Wales).Citation38 Each immunizer is capable of delivering between 80 and 100 doses per hourCitation38 in emergency situations, so we assumed up to 125,000 doses per day could be given in Sydney, for targeted and mass vaccination, however we tested the case of 50,000 and 300,000 doses per day for mass vaccination.
For the targeted vaccination strategy, we considered health care workers (HCWs) as first responders who would be prioritized for vaccination in the event of an epidemic. We estimated the number of HCWs in Sydney for 2016–2017Citation39,Citation40 based on the total estimated health workforce for NSWCitation41 and adjusted for the Sydney population.Citation29 We estimated 100,638 HCWs in Sydney, and applied an estimated age distribution.Citation39 We estimated the proportion of HCWs previously vaccinated and unvaccinated according to age. We assumed HCWs to have up to three times higher a risk of respiratory infections, based on past research on respiratory pathogensCitation42; however, we considered them wearing a respirator and personal protective equipment (PPE) from the start of intervention and estimated this to be 75% effective against transmission of respiratory pathogens.Citation43 Full description of the parameters used in the models can be found in the Appendix.
Mathematical model
We constructed three modified SEIR models to simulate targeted, ring and mass vaccination. The models run for 300 days. We started the epidemic with 100, 1000, or 10,000 infected in the latent untraced compartment, distributed following the total population age distribution and vaccination rates.
The model uses ordinary differential equations, to move the population into epidemiological states related to their smallpox infectious status and vaccination strategy compartments, following disease duration rates. The nonlinear age-specific disease transmission rate at which a susceptible person becomes infected () is a combination of β (probability of becoming infected per contact), c (the number of contacts per unit time) and the infectious prevalence. To include age-dependency, we used Euler’s discretization of the continuous variable “age,” so that the force of infection is represented by
Where k = 1, …, 4 represents the four infectious levels for hemorrhagic, flat, ordinary and modified smallpox types and i,y = 1, …, 18 represents the age groups. Individuals mix by age groups following the contact matrix.Citation44 As it was not available specifically for Australia, we adopted the contact matrix estimated for the United Kingdom based on more behavior similarity between the two countries. We started vaccination at day 15, 20, and 30 from the virus release, about a few days to a week or two after appearance of the first symptomatic cases. For targeted vaccination, we avoided immunocompromised people, who are at higher risk of adverse events and likelihood of a poor immune response,Citation45 while for ring vaccination in close contacts and mass vaccination we included the immunosuppressed population.
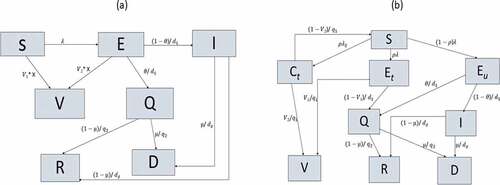
For mass and targeted vaccination ()), a proportion of susceptible (S) or latent (E) individuals can be vaccinated and move to compartment V. Vaccination rates distribution; in different age-groups and immunity levels, as well as in symptomatic or latent people for mass vaccination model; are detailed from the matrix X. The model diagram is the same for mass and targeted vaccination, however the vaccine distribution rates (matrix X) differs between the two strategies (full details in the supplementary materials). Once symptomatic, the infected not vaccinated can be isolated (Q compartment) or proceed to the infectious compartment I, where they will be able to transmit the disease. For ring vaccination ()) a susceptible individual, if infected, can be traced (Et) or untraced (Eu); while if not infected, can be a traced contact (Ct), which can be effectively vaccinated and move to the V compartment or come back to the S compartment. Traced infected (Et) can be successfully vaccinated and move to V compartment or isolated (Q) once symptomatic; while a proportion of latent untraced, once symptomatic, will get isolated in Q compartment and the rest will be infectious in the I compartment. Isolated (Q) or not (I), infected people will then recover (R) or die (D). The following is a short description of the model used for each vaccination strategy simulated for the outbreak response. For each strategy, sensitivity analysis was done on time starting intervention as 15, 20, and 30 days from virus release, furthermore for mass and ring vaccination we conducted a sensitivity analysis on daily vaccinations distributed.
Strategy 1: targeted vaccination ())
We previously determined that the highest risk of disease transmission is in the age group 5–19 years, and the highest risk of death in >65 years.Citation28 This was the basis of tested age-based targeted vaccination strategies and first responders. Therefore, we modeled the different outcomes if the vaccination is prioritized to:
5–19 age group (being the age group with the highest number of contacts/transmissionsCitation28),
60–79 age group (being the age group with highest risk of complications and deathCitation28)
All HCWs and all 5–19 year old
All HCWs only
Vaccination doses are distributed in 7 days for the first two scenarios and 8 for the third one, using the first day to vaccinate HCWs.Citation38,Citation46 The number of doses delivered per day is estimated to be 125,000.Citation38 Doses were distributed proportionally to the size of the age groups in the targeted group and between previously vaccinated and not.
Strategy 2: ring vaccination ())
In order to simulate a response with tracing/vaccinating contacts and case isolation, we used the model structure from Ref. Citation34 but accounting for different immunity levels and infectivity for each smallpox type as in Ref. Citation28.
Isolation of cases and percentage of traced vaccinated contacts are based on available pre-eradication dataCitation1 and assumed to be 95% in the base case scenario, which is likely for cases hospitalized and isolated in a high-income setting such as Sydney. However, we explored and showed results for three different daily proportion of contacts traced and vaccinated (70%, 80%, and 95%).
Strategy 3: mass vaccination ())
In order to calculate the vaccine coverage needed to stop a smallpox outbreak and reduce transmissions to <1 per infected we used the formulaCitation47
where Vc is the vaccine coverage and Ve is the vaccine effectiveness; however, we used the estimated R0 = 4.6, obtained as previously explained.
In order to implement mass vaccination, we fixed the number of doses delivered per day and calculated the duration of the campaign (in days) needed to reduce transmissions to zero; however, the distribution changes daily. We keep the distribution of doses proportional to the weighted size of each age group, immunity level, and susceptible or latent group. The number of doses needed to reach the required coverage to stop transmission depends from the proportion of latent and susceptible people (each with a different vaccine effectiveness VE as shown in Technical Appendix), which varies over the epidemic. We started mass vaccination in an ongoing smallpox outbreak, therefore the VE was varied for latent and susceptible people (see Technical Appendix). Sensitivity analysis has been done on number of daily doses delivered as 125,000 doses per day for the base case scenario, with a sensitivity analysis of 300,000 and 50,000 doses per day. Results are shown by time of starting intervention after virus release.
Results
Targeted vaccination
The higher the initial number infected, the more rapid and severe the epidemic.
In we compare the age specific incidence of infection after 50 days from virus release (a) and the total number of deaths (b) for each targeted vaccination scenario and the case of only isolation of cases without vaccination. The number of doses used to cover each targeted group are 100,000, 850,000, 900,000, and 1 million respectively for the four vaccination scenarios. Vaccination of HCWs has minimal effect on the population epidemic. Vaccinating the 5–19 age group produces a higher reduction in transmissions ()) and consequently a smaller number of deaths by the end of the epidemic. Deaths are sensitive to the time starting intervention, quadrupling between starting intervention at day T = 15 and T = 30 ().
Figure 2. The impact of alternative targeted vaccination strategies on the incidence of infectious people (left) and cumulative deaths (right) over time in the case of a smallpox outbreak starting with 100 infected people in the Sydney population of just over 5 million, with the response commencing on day 20 after the attack
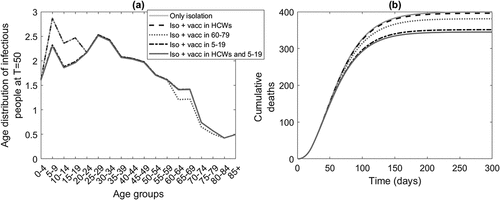
Figure 3. Total number of deaths at the end of the epidemic by time starting intervention and targeted group for vaccination, with 95% case isolation. Results are showed for 100 initial infected
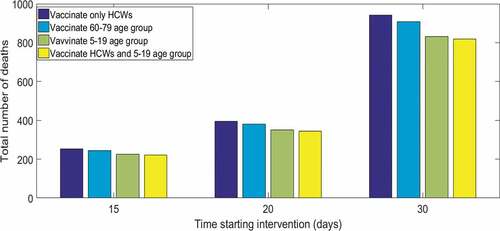
For the targeted vaccination strategy, the higher the initial number infected, the smaller the difference between targeting different age groups for vaccination. However the total number of deaths at the end of the epidemic is proportional to the initial number infected, which in this case is 2.2–2.5, 3.5–4, and 8.1–9 times the initial number of infected respectively starting intervention at T = 15, 20 and 30 days after virus release.
Ring vaccination
With ring vaccination we explored the influence of percentage of contacts traced/vaccinated and time to starting intervention on epidemic size, cumulative deaths and number of doses needed, starting outbreak with 100 (base case), 1000, and 10,000 initial infected.
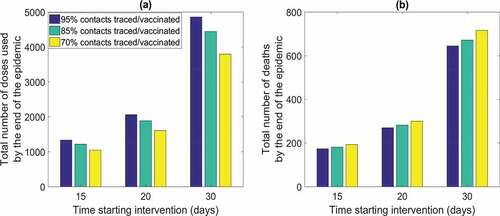
With 100 initial infected starting ring vaccination at day 20 with 95% of new infected isolated each day, the outbreak will be controlled in about 100 days, regardless the proportion of contacts traced. However, the number of doses needed and total deaths at the end of the epidemic is dependent from time of starting intervention and contacts vaccinated. shows the impact of timing of the response varying from 15, 20, to 30 days postattack, corresponding to 3, 8, and 18 days after the first symptomatic patient presents, and contact traced. The results are most sensitive to timing of response (). If the starting intervention is 30 days (T = 30) after virus release, the number of doses needed and total deaths will be more than doubled () compared to starting at 20 days (T = 20) A ring response with 100 initial infected, with isolating and vaccinating a high proportion (95%) of new infectious cases and contacts respectively, will end the epidemic with a total of 270 deaths (2.7 times the initial number of infected) using about 2060 doses starting the intervention at T = 20 days following the start of the epidemic compared with about 4800 doses used and 620 deaths if intervention starts 30 days following the start of the epidemic
Mass vaccination
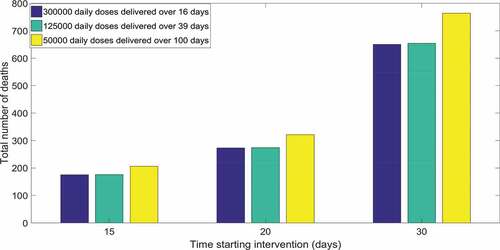
For mass vaccination, assumed an average reproduction number as 4.6, vaccine coverage of at least 82% is required with vaccine effectiveness being 95%. Reaching 82% coverage in Sydney will need about 4.3 million doses, if delivered before the start of the outbreak. In the best-case scenario of 300,000 doses delivered daily, we found that will need, to vaccinate daily for 16 days, 468 vaccinators a day (considering that each vaccinator can delivery 80 doses per hourCitation38 and 640 per day) reaching a total of 4.8 million doses to reduce transmission to zero. If the daily number of doses is 125,000, 4.875 million doses will be needed to stop transmission, in a 39 day continuous campaign. If capacity to vaccinate is only 50,000 doses a day, about 5 million doses in 100 days would be needed by the end of the epidemic. We show how the number of daily doses delivered and the time starting intervention influences number of total deaths at the end of the outbreak in . The number of daily doses () becomes more influential when the doses number drop to 50,000. In the worst case scenarios of delivering only 50,000 doses a day there will be a total number of deaths as 2, 3.2, and 7.6 times the initial infected respectively by starting intervention at day 15, 20, and 30. However even for mass vaccination the most important variable is timing.
Figure 5. Total number of deaths by time starting intervention and number of doses delivered daily. Result showed for 100 initial infected
summarizes all the results for each vaccination strategy explored for the base case scenario with 100 infected people and starting intervention after about 8 days from the first symptomatic cases.
Table 1. Comparison of vaccination strategies for the base case scenario of 100 initial infected with the response commencing at day 20 postrelease and 95% of cases effectively isolated
Conclusion
Understanding the most appropriate public health vaccination strategy for epidemic control is critical for preventing population morbidity and mortality of reemergent smallpox. Reducing cost, wastage, and adverse events are also considerations. We showed that the time to commencing the response is critical. However, we used time from the attack, which includes an average incubation period of 12 days prior to smallpox being recognized.Citation32 This means in practice that for a response to start at day 15 postattack, vaccination must commence 3 days after the first case becomes symptomatic. For 20 days, this would be 8 days after the first symptoms begin. Given smallpox is eradicated, and failures in recognition of serious emerging infections is common in the emergency room, 20 or 30 days, or even longer, may be the reality. For example, the last European outbreak of smallpox in Yugoslavia resulted from failure to diagnose the index case, who had returned from the Middle East.Citation48 In that instance, smallpox was not recognized until second generation cases began appearing, which would correspond to a response time of over 25 days after the initial infection. Given recent notable failures to recognize Ebola in the United StatesCitation49 and Nigeria,Citation50 and MERS in South Korea,Citation51 failure in diagnosis may be a weak point in health systems. Among the targeted vaccination strategies, vaccination for the 5–19 age group is slightly more successful in reducing transmission and delaying the epidemic, but ring vaccination will result in better epidemic control with a smaller number of doses. In all cases, starting the response as early as possible is best for epidemic control, but remains better with higher proportions of contacts traced and vaccinated. The difference between targeted vaccination options delivered to very small proportions of the population is small, because overall vaccine coverage remains low in all of these strategies. In practice, vaccination will be required for HCWs, who are unlikely to come to work if not offered vaccination. Therefore, for the city of Sydney, for core clinical HCWs, at least 100,000 doses of vaccine should be available to ensure workforce willingness to work and to meet occupational safety requirements. Stockpiling for other first responders such as paramedics, emergency services and police should also be considered.
We estimated an overall reproductive number of R0 = 4.6, consistent with other estimates.Citation52,Citation53 For mass vaccination, this translates to requiring 82% of the population to be effectively vaccinated to achieve herd immunity and stop transmission. Reaching vaccine coverage of 82% of susceptible people requires almost 5 million doses, including over 828,000 doses of nonreplicating vaccine for immunosuppressed people, for minimal net benefit over ring vaccination, except for reduction in the duration of the epidemic from 100 to 60 days, or just over a month. The risk of adverse events and deaths from vaccination also increases with mass vaccination. However in the case of not being able to trace and vaccinate a high percentage of contacts, mass vaccination with at least 125,000 doses delivered per day could prevent more deaths. This would require a large scale program with human resources and vaccine supplies. Although there is lack of knowledge of the use of bifurcated needle for vaccination, and training of vaccinators would take some time, we estimated more than 100,000 HCWs in Sydney.Citation54 A study found that each immunizer in Sydney is capable of delivering between 80 and 100 doses per hourCitation38 in emergency situations, so delivery of at least 125,000 doses per day seems a realistic target. For every vaccination strategy the final number of deaths is directly proportional to the size of initial infected, and it should be noted that our results use a base case of 100 initial infected, which is an optimistic assumption. Larger attack size will result in a longer and larger epidemic.
The results of this study are consistent with other modelling studies that look at vaccination strategies for a smallpox outbreak,Citation19,Citation21,Citation23-27 all suggesting that ring vaccination is the best strategy to contain an epidemic. However past studies do not consider heterogeneous mixing patterns, residual immunity from previous vaccination or population immunosuppression when estimating deaths or doses of nonreplicating vaccine needed in different scenarios. We estimate the number of nonreplicating vaccine doses required for the different vaccination options ranges from under 300 for ring vaccination to over 800,000 for a mass vaccination strategy. To ensure the safety of immunosuppressed contacts, a relatively modest investment in stockpiling of third generation vaccine will be adequate.
Only two previous studies showed number of doses needed for different scenarios.Citation21,Citation25 ZenihanaCitation21 showed that with 20 initial infected, ring vaccination ends with 38 deaths, which corresponds to 190 deaths if the initial infected are 100, while we found 270 deaths for the same scenario. However, this could be explained by our model considering the immunocompromised population as being at higher risk of death. LegrandCitation25 found for 100 initial infected, starting intervention at T = 25 days, the epidemic will end with 730 deaths and 5440 doses used. Those higher values can be explained from the lower percentages of isolation (60%) and contacts vaccinated (80%) compered to the 95% assumption in our study. Indeed if we use the same percentages of isolation and vaccination we find just over 7000 doses used and 1500 deaths, which is likely due to consideration on 17% of the population immunosupressed. We assumed high levels of case isolation as a highly effective adjunct strategy to vaccination. We expect in a setting such as Sydney that case isolation rates close to 95% will be achieved, and this underpins epidemic control.
The main limitation of this study is the unavailability of data regarding smallpox transmission in a contemporary population. Therefore there is significant uncertainty regarding how effective a public health will be in managing a terror attack with variola virus. Furthermore, we did not account for the beneficial use of newer antivirals as treatment of smallpox cases, which would reduce the number of deaths. Another limitation is that we assumed HCWs partially to be protected from PPE based on data from respiratory transmission of pathogens, but there are no direct data on PPE use for smallpox. The lack of PPE data for smallpox reinforces the need for vaccination for HCWs. Including age-specific mixing patterns, accounting for residual immunity from past vaccination and age-specific estimation of immunosuppression rates is a strength of our model.
In summary, rapid implementation of vaccination response, combined with high rates of case isolation are critical to smallpox epidemic control, which can be achieved using ring vaccination in a setting such as Sydney. Further research is needed to estimate health system capacity for managing cases, for rapidly delivering vaccination at scale, tracing contacts and monitoring, to inform preparedness planning for a smallpox outbreak.
Supporting information
S1. Models Description
S2. Targeted and mass vaccination models
S3. Ring vaccination model
Supplemental Material
Download MS Word (48.5 KB)Supplementary material
Supplemental data for this article can be accessed online at http://dx.doi.org/10.1080/21645515.2020.1800324.
References
- Fenner F, Henderson D, Arita I, Ježek ZLI Smallpox and its eradication.
- Kennedy RB, Lane JM, Henderson DA, Poland GA. Smallpox and vaccinia. Vaccines: Sixth Edition 718-745 (Elsevier Inc., 2012). doi:10.1016/B978-1-4557-0090-5.00010-0.
- Koblentz GD. The De Novo synthesis of horsepox virus: implications for biosecurity and recommendations for preventing the reemergence of smallpox. Heal Secur [ Internet]. 2017 Aug 24 [ accessed 2017 Oct 16];15(6):620–28. http://www.ncbi.nlm.nih.gov/pubmed/28836863.
- Kupferschmidt K. How Canadian researchers reconstituted an extinct poxvirus for $100,000 using mail-order DNA. Science (80-) [ Internet]. 2017 Jul 6 [ accessed 2018 Apr 9]; http://www.sciencemag.org/news/2017/07/how-canadian-researchers-built-poxvirus-100000-using-mail-order-dna.
- Yen C, Hyde TB, Costa AJ, Fernandez K, Tam JS, Hugonnet S, Huvos AM, Duclos P, Dietz VJ, Burkholder BT. The development of global vaccine stockpiles. Lancet Infect Dis [ Internet]. 2015 Mar [ accessed 2018 Sep 17];15(3):340–47. http://www.ncbi.nlm.nih.gov/pubmed/25661473.
- Nalca A, Zumbrun EE. ACAM2000: the new smallpox vaccine for United States strategic national stockpile. Drug Des Devel Ther [ Internet]. 2010 May 25 [ accessed 2017 Aug 11];4:71–79. http://www.ncbi.nlm.nih.gov/pubmed/20531961.
- CDC-Centers for Disease Control and Prevention. Public health response activities | smallpox | CDC [Internet]. [accessed 2018 Sep 17]. https://www.cdc.gov/smallpox/bioterrorism-response-planning/public-health/index.html.
- Kaplan EH, Craft DL, Wein LM. Emergency response to a smallpox attack: the case for mass vaccination. Proc Natl Acad Sci U S A [ Internet]. 2002 Aug 6 [ accessed 2018 Sep 17];99(16):10935–40. http://www.ncbi.nlm.nih.gov/pubmed/12118122.
- Dasaklis TK, Rachaniotis N, Pappis C. Emergency supply chain management for controlling a smallpox outbreak: the case for regional mass vaccination. Int J Syst Sci Oper Logist [ Internet]. 2017 Jan 2 [ accessed 2018 Sep 17];4(1):27–40. https://www.tandfonline.com/doi/full/10.1080/23302674.2015.1126379.
- Kermack WO, McKendrick AG. A Contribution to the Mathematical Theory of Epidemics. Proc R Soc A Math Phys Eng Sci. 1927 Aug 1;115(772):700–21.
- Heesterbeek H, Anderson RM, Andreasen V, Bansal S, De Angelis D, Dye C, Eames KTD, Edmunds WJ, Frost SDW, Funk S. Modelling infectious disease dynamics in the complex landscape of global health. Science [ Internet]. 2015 Mar 13 [ accessed 2017 Oct 16];347(6227):aaa4339. http://www.ncbi.nlm.nih.gov/pubmed/25766240.
- Huppert A, Katriel G. Mathematical modelling and prediction in infectious disease epidemiology. Vol. 19, Clinical Microbiology and Infection. Blackwell Publishing Ltd; 2013. p. 999–1005. doi:10.1111/1469-0691.12308.
- Rodrigues HS. Application of SIR epidemiological model: new trends. Int. J. Appl. Math. Info; 2016;10:92–97.
- Trauer JM, Dodd PJ, Gomes MGM, Gomez GB, Houben RMGJ, McBryde ES, Melsew YA, Menzies NA, Arinaminpathy N, Shrestha S. The importance of heterogeneity to the epidemiology of tuberculosis. Clin Infect Dis [ Internet]. 2019 Jun 18 [ accessed 2019 Nov 25];69(1):159–66. https://academic.oup.com/cid/article/69/1/159/5154892.
- Kong L, Wang J, Han W, Cao Z. Modelling heterogeneity in direct infectious disease transmission in a compartmental model. Int J Environ Res Public Health. 2016 Feb 24;13:3. doi:10.3390/ijerph13030253.
- Kaplan EH, Craft DL, Wein LM. Analyzing bioterror response logistics: the case of smallpox. Math Biosci [ Internet]. 2003 Sep 1 [ accessed 2018 Sep 17];185(1):33–72. https://www.sciencedirect.com/science/article/pii/S0025556403000907.
- Egan JR, Hall IM, Leach S. Stamping out fires! controlling smallpox with targeted mass vaccination. Med Decis Mak [ Internet]. 2011 Jan 18 [ accessed 2018 Sep 17];31(1):69–78. http://journals.sagepub.com/doi/10.1177/0272989X10369003.
- Eubank S, Guclu H, Anil Kumar VS, Marathe MV, Srinivasan A, Toroczkai Z. Modelling disease outbreaks in realistic urban social networks. Nature [ Internet]. 2004 May 13 [ accessed 2018 Sep 17];429(6988):180–84. http://www.ncbi.nlm.nih.gov/pubmed/15141212.
- Hall IM, Egan JR, Barrass I, Gani R, Leach S. Comparison of smallpox outbreak control strategies using a spatial metapopulation model. Epidemiol Infect [ Internet]. 2007 Oct [ accessed 2018 Sep17];135(7):1133–44. http://www.ncbi.nlm.nih.gov/pubmed/17222358.
- Halloran ME, Longini IM, Nizam A, Yang Y. Containing bioterrorist smallpox. Science [ Internet]. 2002 Nov 15 [ accessed 2018 Sep 17];298(5597):1428–32. http://www.ncbi.nlm.nih.gov/pubmed/12434061.
- Zenihana T, Ishikawa H. Effectiveness assessment of countermeasures against bioterrorist smallpox attacks in Japan using an individual-based model. Environ Health Prev Med [ Internet]. 2010 Mar 26 [ accessed 2018 Sep 17];15(2):84–93. http://link.springer.com/10.1007/s12199-009-0111–z.
- Sato H, Sakurai Y. The contribution of residents who cooperate with ring-vaccination measures against smallpox epidemic. Disaster Med Public Health Prep [ Internet]. 2012 Oct 8 [ accessed 2018 Sep 17];6(03):270–76. http://www.journals.cambridge.org/abstract_S1935789300004493.
- Ohkusa Y, Taniguchi K, Okubo I. Prediction of smallpox outbreak and evaluation of control-measure policy in Japan, using a mathematical model. J Infect Chemother Internet. 2005;11(2):71–80. accessed 2018 Sep 17 http://linkinghub.elsevier.com/retrieve/pii/S1341321X05710072.
- Kretzschmar M, van den Hof S, Wallinga J, van Wijngaarden J. Ring vaccination and smallpox control. Emerg Infect Dis [ Internet]. 2004 May [ accessed 2018 Sep 17];10(5):832–41. http://www.ncbi.nlm.nih.gov/pubmed/15200816.
- LEGRAND J, VIBOUD C, BOELLE PY, VALLERON AJ, FLAHAULT A. Modelling responses to a smallpox epidemic taking into account uncertainty. Epidemiol Infect [ Internet]. 2004 Feb [ accessed 2018 Sep 17];132(1):19–25. http://www.journals.cambridge.org/abstract_S0950268803001390.
- Longini IM, Elizabeth Halloran M, Nizam A, Yang Y, Xu S, Burke DS, Cummings DAT, Epstein JM. Containing a large bioterrorist smallpox attack: a computer simulation approach. Int J Infect Dis [ Internet]. 2007 Mar 1 [ accessed 2018 Sep 17];11(2):98–108. https://www.sciencedirect.com/science/article/pii/S1201971206000968.
- Porco TC, Holbrook KA, Fernyak SE, Portnoy DL, Reiter R, Aragón TJ. Logistics of community smallpox control through contact tracing and ring vaccination: a stochastic network model. BMC Public Health [ Internet]. 2004 Dec 6 [ accessed 2018 Sep 17];4(1):34. http://www.ncbi.nlm.nih.gov/pubmed/15298713.
- MacIntyre CR, Costantino V, Chen X, Segelov E, Chughtai AA, Kelleher A, Kunasekaran M, Lane JM. Influence of population immunosuppression and past vaccination on smallpox reemergence. Emerg Infect Dis [ Internet]. 2018 Apr [ accessed 2018 Mar 20];24(4):646–53. http://wwwnc.cdc.gov/eid/article/24/4/17-1233_article.htm.
- Sydney Population 2018 [ Internet]. [accessed 2018 Feb 12]. http://www.population.net.au/sydney-population/.
- Statistics c=AU; o=Commonwealth of A ou=Australian B of. Details - Key Figures. [ accessed 2018 Feb 12]. http://www.abs.gov.au/AUSSTATS/[email protected]/DetailsPage/3101.0Jun 2017?OpenDocument.
- Sarkar JK, Mitra AC, Mukherjee MK, De SK, Mazumdar DG. Virus excretion in smallpox. 1. Excretion in the throat, urine, and conjunctiva of patients. Bull World Health Organ [ Internet]. 1973 May [ accessed 2019 May 16];48(5):517–22. http://www.ncbi.nlm.nih.gov/pubmed/4359678.
- Costantino V, Kunasekaran MP, Chughtai AA, MacIntyre CR. How valid are assumptions about re-emerging smallpox? A systematic review of parameters used in smallpox mathematical models. Mil Med [ Internet]. 2018 Feb 7 [ accessed 2018 Mar 20];183(7–8):e200–e207. http://www.ncbi.nlm.nih.gov/pubmed/29425329.
- Keckler MS, Reynolds MG, Damon IK, Karem KL. The effects of post-exposure smallpox vaccination on clinical disease presentation: addressing the data gaps between historical epidemiology and modern surrogate model data. Vaccine [ Internet]. 2013 Oct 25 [ accessed 2018 Sep 7];31(45):5192–201. http://www.ncbi.nlm.nih.gov/pubmed/23994378.
- Gani R, Leach S. Transmission potential of smallpox in contemporary populations. Nature [ Internet]. 2001 Dec 13 [ accessed 2017 Aug 11];414(6865):748–51. http://www.nature.com/doifinder/10.1038/414748a.
- Mack TM. Smallpox in Europe, 1950-1971. J Infect Dis [ Internet]. 1972 Feb [ accessed 2018 Mar 20];125(2):161–69. http://www.ncbi.nlm.nih.gov/pubmed/5007552.
- Massoudi MS, Barker L, Schwartz B. Effectiveness of postexposure vaccination for the prevention of smallpox: results of a delphi analysis. J Infect Dis [ Internet]. 2003 Oct 1 [ accessed 2018 Jan 8];188(7):973–76. https://academic.oup.com/jid/article-lookup/doi/10.1086/378357.
- Sato H. Countermeasures and vaccination against terrorism using smallpox: pre-event and post-event smallpox vaccination and its contraindications. Environ Health Prev Med [ Internet]. 2011 Sep [ accessed 2018 Sep 7];16(5):281–89. http://www.ncbi.nlm.nih.gov/pubmed/21431786.
- Mass Vaccination Clinics during an Influenza Pandemic Policy manual Not applicable Functional group Clinical/Patient Services-Governance and Service Delivery, Infectious Diseases Population Health-Communicable Diseases [ Internet]. 2018 [accessed 2018 Sep 7]. https://www1.health.nsw.gov.au/pds/ActivePDSDocuments/GL2018_008.pdf.
- Downloads | AHPRA annual report 2016/17 [ Internet]. [accessed 2018 Feb 1]. http://www.ahpra.gov.au/annualreport/2017/downloads.html#data.
- Workforce Statistics - NSW Ambulance [ Internet]. [accessed 2018 Feb 1]. http://www.ambulance.nsw.gov.au/Our-performance/Workforce-Statistics.html.
- Population of New South Wales 2018 [ Internet]. [accessed 2018 Feb 12]. http://www.population.net.au/population-of-new-south-wales/.
- Macintyre CR, Seale H, Yang P, Zhang Y, Shi W, Almatroudi A, MOA A, WANG X, LI X, PANG X. Quantifying the risk of respiratory infection in healthcare workers performing high-risk procedures. Epidemiol Infect [ Internet]. 2014 Sep [ accessed 2018 Apr 13];142(9):1802–08. http://www.ncbi.nlm.nih.gov/pubmed/24308554.
- MacIntyre CR, Chughtai AA, Rahman B, Peng Y, Zhang Y, Seale H, Wang X, Wang Q. The efficacy of medical masks and respirators against respiratory infection in healthcare workers. Influenza Other Respi Viruses [ Internet]. 2017 Nov 1 [ accessed 2017 Dec 11];11(6):511–17. doi:10.1111/irv.12474.
- Social Contacts and Mixing Patterns Relevant to the Spread of Infectious Diseases: e74 - ProQuest.
- CDC. Clinical guidance for smallpox vaccine use in a postevent vaccination program [ Internet]. 2015 [ accessed 2018 Jan 8]. https://www.cdc.gov/mmwr/preview/mmwrhtml/rr6402a1.htm.
- Carr C, Durrheim D, Eastwood K, Massey P, Jaggers D, Caelli M, Sonya Nicholl LW. Australia’s first pandemic influenza mass vaccination clinic exercise: hunter New England area health service, NSW, Australia. AJEM [ Internet]. 2011 [ accessed 2018 Sep 7];26(1). https://ajem.infoservices.com.au/items/AJEM-26-01–09.
- Anderson RMM RM. Infectious diseases of humans: dynamics and control - Roy M. Anderson, Robert M. May - google books [ Internet]. 1995 [accessed 2018 Oct 12]. https://books.google.com.au/books?hl=en&lr=&id=HT0–xXBguQC&oi=fnd&pg=PA25&dq=infctious+diseaes+in+humans+anderson+and+May&ots=IdjiMMTSig&sig=ZTzFoEoxUrP9qZA3nWBxh8zO5EU#v=onepage&q=infctious diseaes in humans anderson and May&f=false.
- Ilic M, Ilic I. The last major outbreak of smallpox (Yugoslavia, 1972): the importance of historical reminders. Travel Med Infect Dis [ Internet]. 2017 May 1 [ accessed 2018 Oct 22];17:69–70. https://www.sciencedirect.com/science/article/pii/S1477893917300819.
- Smith CL, Hughes SM, Karwowski MP, et al., Addressing needs of contacts of ebola patients during an investigation of an ebola cluster in the United States — Dallas, Texas, 2014. Morb Mortal Wkly Rep (MMWR)-CDC [ Internet]. 2018 [ accessed 2018 Oct 22]. https://www.cdc.gov/mmwr/preview/mmwrhtml/mm6405a2.htm.
- Fasina FO, Shittu A, Lazarus D, Tomori O, Simonsen L, Viboud C. Transmission dynamics and control of Ebola virus disease outbreak in Nigeria, July to September 2014. Eurosurveillance [ Internet]. 2014 Oct 9 [ accessed 2018 Oct 22];19(40):20920. http://www.eurosurveillance.org/content/10.2807/1560-7917.ES2014.19.40.20920.
- Korea Centers for Disease Control and Prevention. Middle east respiratory syndrome coronavirus outbreak in the republic of Korea, 2015. Osong Public Heal Res Perspect [ Internet]. 2015 Aug [ accessed 2018 Oct 22];6(4):269–78. http://www.ncbi.nlm.nih.gov/pubmed/26473095.
- Eichner M, Dietz K. Transmission potential of smallpox: estimates based on detailed data from an outbreak. Am J Epidemiol [ Internet]. 2003 Jul 15 [ accessed 2017 Aug 30];158(2):110–17. https://academic.oup.com/aje/article-lookup/doi/10.1093/aje/kwg103.
- Del Valle S, Hethcote H, Hyman JM, Castillo-Chavez C. Effects of behavioral changes in a smallpox attack model. Math Biosci [ Internet]. 2005 Jun 1 [ accessed 2018 Oct 11];195(2):228–51. https://www.sciencedirect.com/science/article/pii/S0025556405000593.
- MacIntyre CR, Costantino V, Kunasekaran MP. Health system capacity in Sydney, Australia in the event of a biological attack with smallpox. PLoS One [ Lau EH, editor]. 2019 Jun 14 [ Internet];14(6):e0217704. http://dx.plos.org/10.1371/journal.pone.0217704.