ABSTRACT
Background: Disturbances are occurring in healthcare, trade, and travel, with countries closing off their borders due to the pandemic of coronavirus disease 2019(COVID-19) in the world. The pandemic has its impact on the delivery of important health services, including vaccinations. This study aimed to assess the impact of COVID-19 on vaccination coverage among children aged 15–23 months.
Methods: A community-based cross-sectional study was conducted from July 22 to August 7, 2020, in Dessie town, Ethiopia. 633 children with their mother/caregiver were interviewed. Interviewer administered pre-tested structured questioner was used. Data was entered into EPI data and analyzed by using SPSS (Statistical Package for Social Sciences). Both binary and multivariate logistic regression analyses were tested. In multivariate analysis, a significant association was declared at a p-value of < 0.05.
Result: Response rate was 96.4%. Based on the vaccination card plus recall, 350 (57.4%) of children finished all recommended vaccines. Age-eligible vaccination coverage during the COVID-19 outbreak was 12.5% lower than before the outbreak. Being a father main caregiver, mother/caregiver who can write and read, and mother/caregiver who educated from grade 1 to 8 mother/caregiver who is divorced, mother/caregiver who spend more than 30 minutes to reach health institution, and mother/caregiver who aware about the benefit of vaccination, about the campaign, and knowledge of COVID-19 each shows statistically significant association with full vaccination status of children.
Conclusion: Full vaccination coverage among children aged 15–23 months remains low in Dessie town, which further decreases after the COVID-19 pandemic. Hence, measures should be taken to increase consciousness about vaccination and the COVID-19 pandemic.
Background
COVID-19 (Corona Virus Disease −19) is a disease caused by a new strain of coronavirus called Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2). Formerly, COVID-19 disease was referred to as ‘2019 novel coronavirus’ or ‘2019-nCoV. This virus is the most recently discovered coronavirus causing COVID-19 and it was identified when the outbreak began in Wuhan, China, in December 2019.Citation1COVID-19 outbreak has been announced as a Public Health Emergency of International Concern (PHEIC) and has spread worldwide. Its emergence and global spread cause substantial danger to public health worldwide, particularly in low- and middle- income countries.Citation2,Citation3
COVID-19 pandemic has upended family life around the world. Especially, stress may occur in parents due to school closures, working remotely, and maintaining physical distancing. The settings for child growth and development can be interrupted due to contagious infections like COVID-19. Disturbances to families, friendships, daily routines, and the wider community can hurt a child’s welfare, growth, and safety.Citation4,Citation5
A public health emergency like COVID-19 can make every person in the community to be less capable in the business environment and hard to get access to health care facilities. Due to the lockdown measures, people may also ignore signs and symptoms of different illnesses, use self-prescribed medications and home remedies to treat themselves. So people should be informed enough on this pandemic to protect them.Citation6–8 The socio-economic impacts and the mitigation measures are taken for COVID-19 can affect all age group children in all countries. Children can be prevented from vaccine-preventable diseases through cost-effective and lifesaving public health interventions so-called vaccination service. These routine vaccination services are delivered by the government of Ethiopia through static, outreach, and mobile strategies since 1980.Citation9,Citation10
COVID-19 disease makes people stop attending pertinent healthcare services like antenatal care, newborn screening, or vaccination due to the fear of contracting the disease. The rise of vaccine-preventable diseases can occur if the people stopped to receive vaccination services and if they don’t attend the schedule for vaccinations due to fear of COVID-19. Many of these diseases, like measles and polio, may even be more severe than COVID-19, especially unvaccinated children. So, distress can happen to the people suffering from disease, their families, and to the health system.Citation11,Citation12
Disruption in life-saving vaccination services due to COVID-19 makes millions of children at risk of diseases like diphtheria, measles, and polio. The disturbance can be because of restrictions of movement, lack of information or fear of infection with the COVID-19 virus, absence of health workers because of restrictions on travel, move to COVID-19 response duties, and lack of protective equipment. A vaccination campaign’s targeting about 15 million children was scheduled in Ethiopia in April. But it was postponed due to COVID-19 to July and the campaign attained 96% coverage (14.4 million).Citation13,Citation14
Vaccination service access and use remain low with a high drop-out rate despite the enormous efforts taken to improve the service. COVID-19 pandemic may further exacerbate this problem. For 13 of the world’s least developed countries, an estimated 13.5 million people will not be vaccinated against measles, polio, and human papillomavirus.Citation10,Citation12
There are different studies conducted to measure the vaccination coverage in children aged 12 to 23 months before by which now the age-eligible for full vaccination coverage is changed into 15 to 23 months because of measle epidemic in the country.Citation15–19 But no study is conducted to assess the impact of COVID-19 on vaccination coverage among children aged 15 to 23 months in Ethiopia.
Therefore, this new and the first study in the country will be helpful for the country. This study aims to assess the impact posed by COVID-19 on vaccination coverage among children aged 15 to 23 months in Dessie town, Northeast Ethiopia, 2020. The outcome of this study will help policymakers, program planning bodies, and service providers to get rid of the obstacles and improve child vaccination service to attain intended control of vaccine-preventable diseases in this time of COVID-19 pandemic.
Methods
Study design, period, and area
A community-based cross-sectional study design was conducted to assess the impact of COVID-19 on vaccination coverage among children aged 15 to 23 months in Dessie town, from July 22- August 7, 2020. Dessie town is located 401kms from Addis Ababa; 480 km from Bahirdar. Its astronomical site is 11°38’ north latitude and 37°15’ east longitude. The town has a national total population of close to 383,000. The town has 5 sub-cities and 18 kebeles.
Population
All children aged 15 to 23 months with their mothers/caregivers living in Dessie town were the source population. Those children aged 15 to 23 months with their mothers or caregiver living in the selected kebeles of Dessie town sub-cities was the study population. The inclusion criteria were children aged 15–23 months with their mothers or caregiver living in Dessie town for at least 12 months before the date of data collection and children aged 15 to 23 months and Caregivers aged 17 years and above were included in this study. But those children with their mothers or caregiver who had not been living in the study area for at least 12 months on the date of data collection were excluded.
Sample size determination
A single population proportion formula [n = (Z a/2)2 P(1-P)/d2] was used. The assumptions used to calculate the sample size were: 95% level of confidence interval which yields Z α/2 = 1.96 on the standard normal distribution curve, 5% margin of error, since there were no published data in Ethiopia about the impact of COVID-19 on vaccination coverage among children aged 15–23 months up to the date of data collection P of 50% proportion was used to get the greatest sample size. Then by considering the design effect of 1.5 and by adding a 10% non-response rate, the sample size was 633 children aged 15 to 23 months with their mother/caregiver.
Sampling technique
Multistage cluster sampling was used. Initially, Dessie town was divided into five sub-cities. Then, three sub-cities were selected by a lottery method. Then, from these randomly selected sub-cities, one kebele from each sub-city was selected using the lottery method. The total sample size was allocated proportionally to each kebele depending on the total number of children aged 15 to 23 months using the data of health extension workers’ registration books. In each kebeles, the first household was selected randomly from the central place of the kebele. The next household was selected according to the inclusion criteria based on the principle of the next nearest household. Households in the kebele were visited until the proportionally allocated sample size for each kebeles was fulfilled. For households having two and more children, one child was selected randomly.
Measures
Coverage by card only: is to show vaccination coverage among those indexed children only having vaccination cards on hand from the total participants.Citation18
Coverage by card plus history: is to refer vaccination coverage calculated using the data vaccination card plus mother’s report.Citation18
Fully vaccinated: a child aged between 15 to 23 months who received one dose of Bacille Calmette Guerin (BCG), three-doses of PCV(pneumococcal conjugate vaccine), three-doses of pentavalent, three doses of OPV,two-dose of Rota, and two-dose of measles vaccine by card plus mother history.Citation20
Index child: it is to refer to a child aged 15–23 months.Citation19
Knowledge on COVID-19: Correct answers were marked one and incorrect answers marked zero from nineteen knowledge questions. Those scoring greater than the mean were classified as having good knowledge. Those below the mean were classified as poor knowledge.
Not vaccinated: an indexed child who didn’t receive any vaccine before this study.Citation15
Pandemic: Is described as a global epidemic; an epidemic that spreads to more than one continent.
Partially/incompletely vaccinated: an index child who had missed one of the ten vaccines or even one dose of the fourteen vaccine doses.Citation19
Data collection tool and procedure
An interviewer-administered structured questionnaire was used to get the required data. The data collection was conducted by using a standardized questionnaire from a baseline survey tool on EPI and other COVID-19 related literature after some change has been made.Citation10,Citation21 The data collector visited the households with eligible children in selected kebeles until the proportionally allocated sample size is achieved. Vaccines received by the indexed child were copied by the data collector after asking mothers/ caregivers to bring the vaccination card of the indexed child. If no vaccination card, different questions were used to assess the vaccination status of the child for each specific vaccine. The mothers were asked to report the number of vaccines that the child had received. Questions about the site of administration (injection or oral), presence of a scar, and child age when vaccinate were included in the tool to decrease recall bias for mothers/caregiver history.
Data analysis
All filled questionnaires were checked for completeness, consistency, and accuracy before the analysis. Then the data was coded, and then cleaning and entry were carried out using EPI INFO software. Then, the data was cleaned for inconsistencies and missing values. The analysis was done using SPSS software version 23. Descriptive statistics such as frequencies, percentages, and cross-tabulations were used to summarize the data. Bivariate and multivariate logistic regression was done to find the contributing factor for the full vaccination status of children. The analysis focused on socio-demographic characteristics of mothers/caregivers and the indexed child, ownership of vaccination card, mothers’ knowledge about the vaccination-related issue, and COVID-19. The multivariate analysis was performed to test associations between the dependent variable (vaccination status) and the independent variables as predictors. In multivariable logistic regression, a significant association was declared at p < .05.
Data quality assurance
The training was given both to data collectors and supervisors. The training focus was on obtaining consent, maintaining neutrality, privacy issues, personal relation, and ethics in social research. By using 5% of the total sample size calculated, a pretest was done and the questionnaire was checked for its clarity, understandability, and simplicity in collecting for what it is aimed. After the pretest, the questionnaires were reformatted based on the inputs and comments generated. Then duplication of the final questionnaires was done. The quality of the data collected was assured by checking every questionnaire on the evening of the date of collection.
Ethical consideration
Ethical clearance was obtained from Wollo University College of medicine and health science research ethical review committee. The ethical clearance letter reference number is WU/324/T-01/2020. Communications with administrators of the selected kebeles in the town were held through a formal letter (reference number: WU/325/T-01/2020). The data collector elaborates on the importance of the study for the participants. Written consent was signed by each participant for their willingness to participate in the study before data collection Confidentiality of the collected data was maintained. . All paper and computer records of the study were kept in a secured place under lock and the name and/or other personal information wasn’t notified in any report.
Results
Socio-demographic characteristics of the child and mother/caregiver
From the calculated sample size of 633 mothers with children aged 15–23 months, 610 were interviewed, making the response rate of 96.37%. Of these 610 children, 50.8% were female. About 21.3% of the children were aged 15 months (Mean age = 18.4 months). Moreover, the majority of the children 274(44.9%) were first-born children in their family. Almost all (99.67%) of the respondents were the main caregiver of the indexed child. The mean age of the mothers/caregiver was 27.84 (SD = 4.855) years (Rang = 17–68 years). About 42.6% and 29.2% of the respondents attended grade 10 to 12 and primary education respectively. About (87%) were married and 55.4% were Muslim in religion. The majorities (65.4%) of the study population were housewives in occupations ().
Table 1. Socio-demographic characteristics of mothers and index children aged 15–23 months, Dessie town, Ethiopia. (N = 610)
Availability and accessibility of vaccination services
Most of the respondents 353(57.9%) stated the access to the health center that delivers vaccination services near to their home. About 365(59.8%) of the respondent states that they go by foot to the nearest health institution to get the service and 158(25.9%) states that the time taken to reach the nearest health institution to be 10 minutes ().
Table 2. Availability and accessibility of vaccination service in Dessie town, Ethiopia, 2020
Vaccination status of the child
Vaccination coverage by card plus recall
Of the surveyed children, only 426(69.8%) have a vaccination card. Based on vaccination card plus recall, 350 (57.4%, 95% CI: 53.48–61.32) of children completed all the recommended vaccines and 1.5% (95% CI: 0.5–2.4) having never been vaccinated. About 251 (41.1%) of the children were not fully/ partially vaccinated.
Vaccination coverage by card only
From the surveyed children, 69.8% took oral polio vaccine 1 and Pneumococcal conjugate vaccine 2 vaccines, followed by pentavalent 2 (69.7%). Measles vaccine 2 was taken by only 55.2% of children and 54.6% were fully vaccinated by card only ().
Table 3. Vaccination coverage by card and card plus mother/caregiver recall, at Dessie town, Ethiopia, 2020. (N = 610)
Reasons for not or incomplete vaccination among children
Mothers/caregivers who did not take their children for the vaccinations at all and not complete vaccinations per schedule had mentioned the following reasons: unaware about the need for vaccination(17.9%), Mother has no enough time (16.1%), Unaware about the need for returning for 2/3rd appointment (15.7%) and fear of contracting COVID-19COVID-19 (11.3%) ().
Table 4. Mothers’/caregivers’ of children aged 15–23 months reasons for not or incomplete vaccination, Dessie town, Ethiopia, 2020
Impact due to COVID-19 pandemic on vaccination coverage rates
To assess the vaccination coverage rate change occurred due to the pandemic, those respondents with vaccination card was only included for this specific goal because more in formations like date of birth, vaccination date, vaccine type, dose and frequency, appointment and all other information can be easily found in the vaccination card and it prevents the recall bias if it was taken from mother/caregiver history. The time frame was considered after March 11, 2020, when FMOH declare the investigation of the COVID-19 case in the country.Citation22
So based on this, Before COVID-19 Pandemic is identified in the country, the age-eligible vaccination rate was 79.2% (95% CI: 72.7.0, 85.7), during the COVID-19 pandemic, the age-eligible vaccination rate was 66.7% (95% CI: 61.02, 72.38). Therefore, age-eligible coverage during the COVID-19 outbreak was 12.5% (95% CI: −27.1% to 2.1%) lower than before the COVID-19 outbreak as described in the table below ().
Table 5. Number of children aged 15 to 23 months old vaccinated by card before the COVID-19 outbreak, and during the COVID-19 outbreak, in Dessie town, Ethiopia, 2020
Mothers’ knowledge about vaccination
From the total 610 mothers/caregivers, about 598 (98%) and 584 (95.7%) know child age to start vaccination and knows that vaccine can prevent VPDs respectively. About 495(81.1%) of the respondent know the frequency for going to health institutions to complete child vaccination as shown by the table below ().
Table 6. Mothers’ knowledge about vaccination for children at Dessie town, Ethiopia, 2020
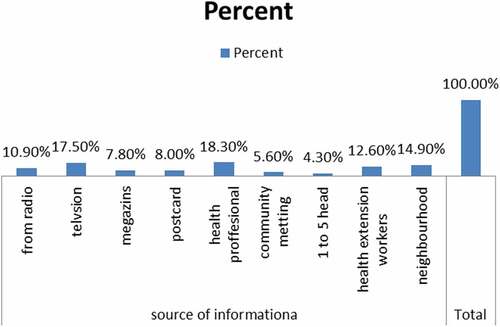
Source of information about vaccinations
Most of the respondents hear about vaccination from health professionals and television as shown by the figure below ().
Knowledge about COVID-19
Of all respondents, 72.5% of the respondent have good knowledge about COVID-19(mean = 15.36, SD = 3.059). Almost all the respondents hear about the COVID-19 pandemic and 94.3% of them know that the disease is contiguous. About 89.8% of the respondent states cough as a sign of COVID-19 and 92.6% of the respondents state need for confirmed case admission to the isolation center as described by the table below ().
Table 7. Knowledge of the respondent about COVID-19, Dessie, Ethiopia, 2020
Factors associated with children full vaccination status
Socio-demographic variables that show association with full vaccination status of the children
The bivariate analysis shows that being a father to be the main caregiver, being able to read and write, being educated from grade 1 to 8, being divorced in marital status, and time taken to reach the nearest health institution show association with complete vaccination status of the child. So all these variables with p < .2 were entered into multivariate analysis.Then, the multivariate analysis shows that father main caregiver (0.382, 95% CI: 0.170, 0.861), those who can write and read (7.822, 95% CI: 1.243, 49.225) and those educated from grade 1 to 8(5.297, 95% CI: 1.263, 22.22), being divorced (0.2, 95% CI, 0.065, 0.981), and those who spend more than 30 minutes to reach health institution(1.619, 95% CI: 1.065, 2.461) were significantly associated with complete vaccination status of children ().
Table 8. Socio-demographic characteristics of mothers and children associated with full vaccination status of children aged 15–23 months, in Dessie, Ethiopia. (N = 610)
Association of mothers’ knowledge on vaccination and COVID-19 with full vaccination status of the children
In bivariate analysis knowing the benefit of vaccination (0.619, 95% CI : 0.444, 0.862), knowing child age-eligible for vaccination(0.671, 95% CI : 0.480, 0.938), knowing place for regular vaccination(0.7, 95% CI : 0.507, 0.966)), knowing the appointment times for vaccination(0.618, 95% CI : 0.430, 0.889), knowing about campaign(0.530, 95% CI : 0.374, 0.753), and good knowledge level on COVID-19(1.562, 95% CI :1.093, 2.234) show association. So all these variables (p < .2) were entered into multivariate analysis. After that 3 variables show statistically significant association: those who know the benefit of vaccination (0.693, 95% CI: 0.483, 0.996), knowing about the campaign (0.564, 95% CI: 0.394, 0.806)) and good knowledge level on COVID-19(1.447, 95% CI: 1.0, 2.093) shows statistically significant association with full vaccination status of the children ().
Table 9. Association between mothers’ knowledge on vaccination related issues and COVID-19 in relation to complete vaccination coverage among children aged 15–23 months, Dessie Ethiopia, 2020
Discussion
This study assessed the impact posed by the COVID-19 Pandemic toward full vaccination coverage and factors associated with vaccination coverage among children aged between 15–23 months old in Dessie town, Ethiopia. Before COVID-19 Pandemic is identified in the country, the age-eligible vaccination rate was 79.2 (95% CI: 72.7.0, 85.7), during the COVID-19 pandemic, the age-eligible vaccination rate was 66.7% (95% CI: 61.02, 72.38) in Dessie town. Therefore, the age-eligible coverage rate during the COVID-19 outbreak was 12.5% (95% CI: −27.08% to 2.08%) lower than before the COVID-19 outbreak. The possible explanation for this may be fear of contracting COVID-19 while going to a health care facility for vaccination service.
Based on vaccination card and recall, 57.4% of children were fully vaccinated. This result was higher than the study conducted in ambo (35.4%) (18, but lower than the study conducted in Tehuldera wereda,Citation23 in Sinan district,Citation19 and the study conducted in Ghana.Citation24 This discrepancy may be due to the fear of the COVID-19 pandemic because all those studies were conducted before the COVID-19 pandemic. On the other hand, it may be due to child age variation between these studies.
Of the respondents, only 426(69.8%) showed the vaccination card of the indexed children. This result was higher than the study in Ambo.Citation18 This discrepancy may be due to the current high awareness about vaccination for children and child age variation between the two studies.
However, in this study children having a father’s main caregiver were less likely to be fully vaccinated (OR = 0.382) than children having a mother’s main caregiver. This finding was similar to the study done in Mozambique in which children delivered at home were less likely to complete vaccination.Citation13 The explanation related to this may be that mothers are more prone to their child and most of them are housewives in their occupation, so they are conscious about their child.
Children from mother/caregivers who can write and read were more likely to be fully vaccinated than children with can’t read and write mothers/caregivers (AOR = 7.822). The possible explanation may be those who can read and write may easily know the appointment time for the next vaccination, so they can finish based on the appointment date.
Those children of mothers/caregivers educated from grade 1 to 8 were more likely to be fully vaccinated than those children of mothers/caregivers who can’t read and write (AOR = 5.297). this is in line with what was shown in Jijiga.Citation25 This is maybe related to an increase in health-seeking behavior of a family when the educational status of the family gets upgraded, which in turn improves the child vaccination.
On the other hand, children from a divorced mother/caregiver were less likely to be fully vaccinated than children of a married mother/caregiver (AOR = 0.253). The possible explanation may be being a divorce may make the mother /caregiver negligent for the child and child and they may not take the child for vaccination.
Children from mothers/caregivers who spend more than 30 minutes to reach the health institution were more likely to be fully vaccinated than those who spend less than 30 minutes(AOR = 1.619). this is in line with the study conducted in the Bale zone.Citation19
Regarding the knowledge related factors, children from mothers/caregivers who don’t know the benefit of vaccination were less likely to be fully vaccinated than know the benefit of vaccination (AOR = 0.693). This may be being unaware of the benefit of vaccination makes them be care less about the full vaccination status of their children which affects the vaccination status of the child.
Those children from mothers/caregivers who aren’t aware of campaigns were less likely to be fully vaccinated than those from mothers/caregivers aware of the campaign (OR = 0.564). This may be due to those who are not aware of the campaign commonly given at home to a home visit. It is difficult to come to the health institution away from their home.
On the other hand, children of mothers/caregivers with good knowledge of COVID-19 were more likely to be fully vaccinated than those children from mothers/caregivers with poor knowledge of COVID-19(AOR = 1.447). The possible explanation for this may be, those who know about COVID-19 related to the sign symptom, transmission, and prevention technique may able to protect themselves and will come on the appointment date for child vaccination without any fear.
Limitations
This study was conducted among children aged 15 to 23 months and we discuss or compare the overall vaccination coverage with other study done before the COVID-19 pandemic because as to our knowledge there is no published data conducted on the impact of COVID-19 on vaccination coverage among children aged 15 to 23 months till the preparation of this manuscript. Recall bias may affect the study because mothers may not exactly remember the vaccines taken, the frequency, and the route.
Conclusion
Full vaccination coverage among children aged 15–23 months remains very low in Dessie town. The age-eligible coverage rate during the COVID-19 outbreak was 12.5% (95% CI: −27.1% to 2.1%) lower than before the COVID-19 outbreak. In addition to this, low awareness about child age-eligible for vaccination, about the side effect of vaccines and measures taken for vaccine side effects, and about transmission and prevention mechanism of COVID-19 pandemic and its fatal outcome(death) was shown among mothers. Children from a father main caregiver, being a mother/caregiver who can write and read and educated from grade 1 to 8, mother/caregiver who spend more than 30 minutes to reach health institution, mother/ caregiver having awareness about the benefit of vaccination, about the campaign, and mother knowledge level on COVID-19 was found to have a significant association with full child vaccination status of children in the town. So prompt action must be taken to increase the consciousness of mothers/caregivers on vaccine benefits, a vaccination campaign, and about the current pandemic of COVID-19 by designing proper health education. Leaflets on vaccination and COVID-19 prevention measures should be prepared in local languages and should be provided to the community through the home to home visit and in health care facilities of the town.
Abbreviations
| ANC | = | Antenatal care |
| BCG | = | Bacille Calmette Guerin |
| CI | = | Confidence interval |
| COVID-19 | = | Coronavirus Disease 19 |
| DC | = | Data collector |
| DOR. | = | Dropout rate |
| EPI | = | Expanded Program on Immunization |
| FMOH | = | Federal Minister of Health |
| HPV | = | Human Papilloma Virus |
| I | = | Investigators |
| IRC | = | Institutional Review Committee |
| IV | = | Intravenous |
| OPV | = | Oral Polio Vaccine |
| PAHO | = | Pan African Health Organization |
| PHEIC | = | Public Health Emergency of International Concern |
| SARS | = | Severe Acute Respiratory Syndrome |
| SPSS | = | Statistical Package for the Social Sciences |
| VPDs | = | Vaccine-preventable diseases |
| WHO | = | World Health Organization |
| WU | = | Wollo University |
Ethics approval and consent to participate
Ethical clearance was obtained from Wollo University College of medicine and health science research ethical review committee. The ethical clearance letter reference number is WU/324/T-01/2020. Communications with administrators of the selected kebeles in the town were held through a formal letter (reference number: WU/325/T-01/2020). The data collector elaborates on the importance of the study for the participants. Written consent was signed by each participant. for their voluntariness to participate in the study before data collection. Confidentiality of the collected data was maintained.
Availability of data and materials
All datasets generated and analyzed during the current study are presented in the paper.
Competing interest
We affirm that we have no competing interests.
Authors’ contributions
DGM participated in the development proposal with methods and objectives, prepared tools, collected data, analyzed, and wrote the first draft of the manuscript. ZAA and SGA supervised the development of the study design, and carried out the literature review, and helped in manuscript writing. All authors developed and supervised the research, edit and ready the manuscript for publication. All authors read and approved the final manuscript.
Acknowledgments
We are thankful for Dessie town community members, data collectors, and supervisors. We are grateful to Wollo University for supporting us to research on the current pandemic disease.
Additional information
Funding
References
- World health organization. 2020. COVID-19. Explained to the parents of young children.
- UNICEF WHO. 2020 March. Key messages and actions for prevention and control in schools.
- National primary health care development agency. 2020. Preparedness and response to Coronavirud Disease 2019 at primary health care and community level.
- UNICEF. 2020. UNICEF Global COVID-19 Situation report No.2. 2:1–17.
- The alliance for child protection in humanitarian action, technical note: protection of children during the Coronavirus pandemic, Version 1, March 2019.
- Melugbo D; Ujunwa1 Ogbuakanne, Maryann Uche1 Jemisenia JO 1Centre. Entrepreneurial potential self-assessment in times of COVID-19: assessing readiness, engagement, motivations, and limitations among young adults in Nigeria. Ianna J Interdiscip Stud. 2020;2(1):12–28.
- Odii A, Ngwu MO, Aniakor MC, Owelle IC, Aniagboso MC, Uzuanwu OW. Effect of COVID-19 lockdown on poor urban households in Nigeria: where do we go from here? Ianna J Interdiscip Stud. 2020;2:75–85.
- Victoria A. A library-based model for explaining information exchange on Coronavirus disease in Nigeria. Ianna J Interdiscip Stud. 2020;2(1):1–11.
- UN. The policy brief: impact of COVID-19 on children; 2020 April 15.
- Ababa A. 2015 June. Extended Program on Immunization (EPI) coverage in selected Ethiopian zones : A baseline survey for L10K’s Routine Vaccination Improvement Initiative. [accessed 2020 May 20]. www.l10k.jsi.com
- Hirabayash DK. 2020 April. The impact of COVID-19 on routine vaccinations. UNICEF East Asia and Pacific.
- ReAct. Opinions and News: impact of COVID-19 on vaccine-preventable diseases and antibiotic resistance in 2020.
- World Health Africa. 2020. Ethiopia vaccinates nearly 15M children against measles.
- WHO. 2020. At least 80 million children under one at risk of diseases such as diphtheria, measles, and polio as COVID-19 disrupts routine vaccination efforts, warn Gavi, WHO, and UNICEF.
- Feleke A, Worku W, Sharma HR 2014 August. Immunization coverage of 12 – 23 months old children and associated factors in Jigjiga District, Somali National Regional State, Ethiopia.
- Hailu S, Astatkie A, Johansson KA, Lindtjørn B. Low immunization coverage in Wonago district, southern Ethiopia: A community-based cross-sectional study. PLoS ONE. 2019;14(7):e0220144. https://doi.org/10.1371/journal.pone.0220144
- Tesfaye TD, Temesgen WA, Kasa AS. Vaccination coverage and associated factors among children aged 12 – 23 months in Northwest. Hum Vaccin Immunother [Internet]. 2018;14(10):2348–54. doi:10.1080/21645515.2018.1502528.
- Etana B, Deressa W. Factors associated with complete immunization coverage in children aged 12 – 23 months in Ambo Woreda, Central Ethiopia. BMC Public Health [Internet]. 2012;12:1.
- Legesse E, Dechasa W. An assessment of child immunization coverage and its determinants in Sinana District, Southeast. Internet. BMC Pediatrics. 2015;15:31.
- Federal Democratic Republic of Ethiopia, Ministry of Health. Health Sector Development Program 2010/11 – 2014/15. FINAL DRAFT October 2010.
- PAHO W. 2020 March. The immunization Program in the Context of the COVID-19 Pandemic.
- FMOH. 2020 April. National comprehensive COVID-19 management handbook.
- Beyene W. 2015 September. Childhood vaccination coverage in Tehuldera district, Northeast Ethiopia : A community-based cross-sectional study.
- Adokiya MN, Baguune B, Ndago JA 2017. Evaluation of vaccination coverage and its associated factors among children 12 – 23 months of age in Techiman Municipality. 1–10.
- Mohamud AN, Feleke A, Worku W, Kifle M, Sharma HR. Immunization coverage of 12 – 23 months old children and associated factors in Jigjiga District, Somali National Regional State, Ethiopia. BMC Public Health. 2014;14:865.