?Mathematical formulae have been encoded as MathML and are displayed in this HTML version using MathJax in order to improve their display. Uncheck the box to turn MathJax off. This feature requires Javascript. Click on a formula to zoom.
?Mathematical formulae have been encoded as MathML and are displayed in this HTML version using MathJax in order to improve their display. Uncheck the box to turn MathJax off. This feature requires Javascript. Click on a formula to zoom.ABSTRACT
Background: There are several COVID-19 vaccines available and many are under different stages of development. However, vaccine hesitancy, including vaccination delays and refusals, represents a major hurdle for achieving herd immunity. The current study aims to evaluate COVID-19 vaccine hesitancy and the associated factors.
Method: This is a cross-sectional survey-based study that was conducted between Aug and Nov 2020.
Results: There were 417 respondents with nearly 61% females, more than 65% fall between the ages of 18 and 29 years, three-quarters holding a university degree, with more than 63% identified as single, and those who have no children represented more than 67% of the respondents. More than 36% of the respondents considered themselves COVID-19 vaccine hesitant. COVID-19 vaccine hesitancy appeared to be high among female respondents (p = .02), aged 30 years old and above (p < .001), widowed or divorced (p < .001) and those who have a child (p < .001). One of the most vaccine hesitancy influencing factors is the vaccines’ country of origin.
Conclusion: There appears to be a high COVID-19 vaccine hesitancy among the participants with several associated factors. The current finding provides a knowledge base for policymakers for communication improvement and confidence-building in relation to COVID-19 vaccines and vaccination.
Introduction
The current Coronavirus Infectious Disease of 2019 (COVID-19) pandemic is the largest public health crisis of our time that caused tremendous mortality and morbidity as well as significant economic loss worldwide. Evidently, the impact of the pandemic is continuously growing with some countries are experiencing a third wave of the viral emergence. One of the major means of controlling the pandemic is the availability of an effective vaccine capable of halting the spread of the deadly virus. Vaccination is one of the most important public health measures developed in the history of medicine that enabled the prevention of serious infectious diseases.Citation1
Nonetheless, there are several newly developed COVID-19 vaccines available, with some have already been rolled out in different countries. As of March 2021, there are more than 200 vaccine candidates being developed, of which 60 are in clinical trials, and seven different vaccines have been rolled out in many countries (WHO). These vaccines have either received full, emergency or provisional approval of use. For instance, one of the front-runner vaccines Pfizer-BioTech has received full approval in several countries, emergency use in the US, EU, UK, and a provisional approval in Australia.Citation2 Similarly, the Moderna vaccine approved for use in Switzerland and approved for emergency use in the US and EU. Also, the Oxford-AstraZeneca vaccine has a full approval in Brazil and several other countries, but received an emergency use approval in the UK and conditional marketing approval (CMA) in the EU.
While COVID-19 vaccine is the most powerful tool that we have in curbing the devastating COVID-19 pandemic, there is an increasing rate of vaccine refusal and skepticism. A recently published report by Pew Research Center stated that four in ten Americans will definitely or probably not take the vaccine and that 21% of American adults are “pretty certain” that they will not take the vaccine.Citation3 The increasing rate of vaccine refusal or hesitancy is driven by diverse factors in different countries or community groups. For instance, in our previous study by Akhmetzhanova et al. (2020) that investigated vaccine hesitancy in Kazakhstan, more than 30% of the participants consider themselves as vaccine hesitant, mostly due to poor vaccine knowledge.Citation4
However, our last survey was conducted prior to the current pandemic. Thus, we can safely assume that since the last survey, the majority of people are aware of the considerable suffering and disruption caused by the COVID-19 pandemic and that they are better informed of the importance of vaccination than before. Hence, the aim of the current study is to determine people’ perception of COVID-19 vaccination and the associated factors with COVID-19 vaccine hesitancy.
Patients and methods
Calculating sample size
The sample calculation formula, which was used in several previous studies,Citation3 is shown below. The applied formula calculated the sample size at 95% confidence interval (CI) with a 5% margin of error using our previous estimate of vaccine hesitancy of 35%:Citation4
where,
n = needed sample size,
Z = Z statistic for a level of confidence,
P = hypothesized vaccine hesitancy rate
d = margin of errors
For the level of confidence of 95%, which is conventional, Z value is 1.96.
(0.05)2 = 350, therefore 350 participants are needed to achieve the required confidence level.
Study setting and design
This is a cross-sectional electronic questionnaire-based study that was carried out in different regions of Kazakhstan, between Aug and Nov 2020. The questionnaire was designed in English then translated to Russian and Kazakh, which are the official spoken languages of Kazakhstan by proficient speakers of both languages and was revised to be suitable to the general population. To ensure accuracy of translation and retaining of the study concepts, the survey was back-translated to English and compared with the original version.
The questionnaire used in the current study was adapted from that of Opel et al., 2011 and developed to assess vaccine hesitancy and its determinants among participants by answering questions to the best of their knowledge.Citation5 The questionnaire contains 44 questions that were divided into different sections, including a section about the participant’s demographic data and other sections that were designed to test the participant’s knowledge, attitude, and practice toward childhood immunization. The COVID-19 vaccine hesitant respondents were determined as those who answered “No” to the question “Would you vaccinate yourself against COVID-19?” whereas those who responded “Yes” as COVID-19 vaccine acceptant.
The questionnaire was validated with a pilot run using a group of 28 randomly selected individuals (16 females and 12 males) who were surveyed to ensure reliability and suitability of the survey. The results of the pilot test indicated minor changes and based on the results of the pilot run, the final corrected version of the questionnaire was used to execute the current study.
Participants were recruited by convenience sampling method through mass e-mail invitations, to students and staff at Nazarbayev University, local hospitals, and contacts in different cities as well as invitations through social medias. Participants received an e-mail with full explanation about the study, its aims, and objectives. The e-mail included a link to the survey, which asks them to consent to participate in the study before granted access. Those who do not consent will not have access to the survey. All participants were made aware that this study is for research purposes only and their participation was voluntary. They were not asked for their names, e-mail address, or contact information, ensuring the privacy of survey respondents.
Study population (inclusion/exclusion)
Inclusion: Participants, males and females, who are at least 18 years of age or older at the time of the survey were included in the study. Exclusion: Those who are under 18 years of age, or those who did not consent to participate were excluded from the study.
Data storage
All data collection forms were kept in a secure setting, only available to the principal investigator.
Statistical analysis
All statistical analyses were performed using STATA version 16.1. In descriptive statistics, skewed numeric variables were presented in medians and interquartile ranges whereas categorical variables were presented as frequencies and percentages. To investigate relationships between categorical variables, Pearson’s chi-square test was used. If one of the assumptions for the chi-square test was violated, Fisher’s exact test was applied. Univariate and multivariable logistic regression analyses were performed to examine associations between independent variables and the COVID-19 vaccine hesitancy. In the final multivariable logistic regression model, only statistically significant and public health important variables were included. Multicollinearity was determined using a rule of thumb, variance inflation factor level > 10.
Ethics approval
The research project was submitted and approved by the Nazarbayev University School of Medicine (NUSOM), Ethics Committee, Kazakhstan # NUSOM-IREC-SEP-2019-1.
Results
Response rate
There were 440 surveys distributed to cover for the required 350 participants at 95% confidence level, plus 10% attrition rate (i.e., to consider nonrespondents). There were 417 completed surveys making the survey response rate to be 94.7%.
Demographic data
A total of 417 were included in the statistical analysis. The majority of the respondents were females (60.9%), between 18–29 years old (65.6%), with University degree (78.8%), single (63.1%), not having a child (67.4%) and were from central or southern regions of Kazakhstan (70.6%) ().
Table 1. Socio-demographic characteristics of the study participants and COVID-19 vaccine hesitancy rate
Prevalence of vaccine hesitancy and knowledge
Interestingly, the majority of respondents with one child or more considered themselves vaccine hesitant, with more than 50% would not vaccinate their daughters against HPV (p < .001), and more than 73% of the respondents were hesitant to vaccinate their children (p < .001) or themselves (p < .001) against seasonal flu. However, the majority of the respondents with children were against vaccinating a child 88% (p < .001) or themselves 76% (p < .001) against COVID-19 (). 36.1% of the respondents considered themselves COVID-19 vaccine hesitant (). The rate of COVID-19 vaccine hesitancy was showing to be high among female respondents (p = .02), aged 30 years old and above (p < .001), widowed or divorced (p < .001) and those who have a child (p < .001) ().
Table 2. Vaccine hesitancy rates among those who have a child and those who do not
Attitudes toward vaccine
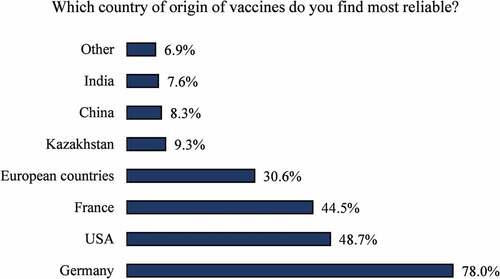
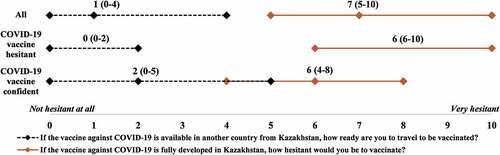
The vaccine country of origin was identified as one of the influencing factors that drove participants to be vaccine hesitant. Thus, participants were provided with a list of countries with vast vaccine productivity and asked to record their confidence level in the producing country. Out of the listed countries, the majority of the participants (78%) selected Germany as the most trustworthy vaccine origin, with India being the least (). The respondents would most likely accept a COVID-19 vaccine that is developed outside of Kazakhstan (). Interestingly, both COVID-19 hesitant and acceptant groups showed significant hesitancy toward a Kazakhstani developed vaccine, and appeared to be more acceptant of those that are developed outside of the country ().
Practice toward vaccine
The respondents were asked whether they follow the vaccination plan identified by the Ministry of Health (MOH). More than 80% of the respondents stated that they do follow the recommended plan with two-thirds agreed with the compulsory plan (). While more than two-thirds of the COVID-19 vaccine acceptant were willing to pay for a vaccine against COVID-19, only 24.2% of the respondents were willing to participate in a trial of vaccination against COVID-19. However, the respondents who were identified as COVID-19 vaccine hesitant, were not willing to pay for the vaccine against COVID-19 (p < .001) and not willing to participate in a trial of vaccination against COVID-19 (p < .001).
Table 3. Barriers and enablers associated with future COVID-19 vaccination
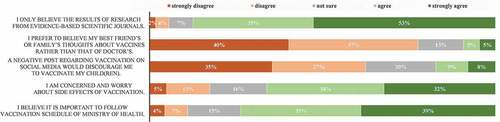
Less than 24% of the respondents believed friends or family’s thoughts about vaccines, but more than 50% of the respondents believed the results from evidence-based scientific journals (). While 17% of respondents are likely to be discouraged by a negative post about vaccination on social media, the majority (76%) of the participants were concerned and worried about potential vaccine side effects ().
Multivariate analyses for vaccine hesitancy
In the univariate logistic regression, sex (p = .02) and age (p < .001) were associated with COVID-19 vaccine hesitancy (); however, in the multivariable model, both of these variables, sex and age, were not associated with COVID-19 vaccine hesitancy. The results of the multivariable regression model showed that having a child (OR = 2.73, p = .03), higher hesitancy level to the vaccine developed in Kazakhstan (OR = 1.20, p < .01), not free COVID-19 vaccine (OR = 4.28, p < .001) and a discouraging negative post on social media (OR = 1.44, p < .01) were independently and positively associated with COVID-19 vaccine hesitancy. Furthermore, the results demonstrated that COVID-19 vaccine that would be developed outside of Kazakhstan (OR = 0.85, p < .01), willingness to participate in a trial of vaccination against COVID-19 (OR = 0.26, p < .01) and reliance on evidence-based scientific journals (OR = 0.61, p < .01) were negatively associated with COVID-19 vaccine hesitancy ().
Table 4. Univariate and multivariable logistic regression models investigating factors associated with COVID-19 vaccine hesitancy
Discussion
We have previously shown that 35% of the respondents considered themselves as vaccine hesitant, which is comparable to the WHO estimated prevalence of vaccine hesitancy.Citation6 However, we hypothesized that the significant impact of the current COVID-19 pandemic might have changed people’s attitude toward vaccination. Thus, this study was developed to measure the impact of the current COVID-19 pandemic on people’s perception and attitude toward vaccination in general and COVID-19 vaccine in particular, among respondents from the republic of Kazakhstan.
The results from the current study show similar rate of vaccine hesitancy (36.1%) and that the hesitancy rate appears, with some degree to be vaccine dependent. Out of all the surveyed individuals, those who have at least one child were shown to be the most hesitant toward vaccinating themselves or children against seasonal flu, or vaccinate their daughters against HPV. This finding is similar to our previous findings in Akhmetzhanova et al. (2020) as well as others including that of Opel et al. who suggest parents were more vaccine hesitant than their childless peers.Citation4,Citation6,Citation7
However, the majority of the respondents opposed participating in COVID-19 trials or receiving the vaccine themselves or for their children, of whom the majority were identified as females within the 30 year old and older group. While there are no global data on COVID-19 vaccine hesitancy available, a recent report from the US suggested that more than half of surveyed black Americans and 30% of Latin American individuals were reluctant to take a free COVID-19 vaccine.Citation8 Nonetheless, the report attributed the high hesitancy rate among the participants to the novelty of the virus, the rapid vaccine development timeline, as well as its politicization.Citation9 Additionally, vaccine side effects appeared to be a concerning factor for more than 66% of the respondents. This is a valid reason of concern that seems to be driven by vaccine misinformation, thus it is important to address valid reasons and respond to vaccine misinformation.Citation10
Interestingly, the viral novelty and fast timeline for vaccine development may have impacted the vast majority of vaccine hesitant individuals worldwide, but the respondents from Kazakhstan have an additional concern that have affected their decision about COVID-19 vaccination that is the vaccine country of origin. A sizable proportion of the respondents stated that they will more likely take a vaccine that is produced outside of Kazakhstan. This concern is directly related to vaccine quality, which might have been exacerbated by previously reported incidents in Kazakhstan, including the infamous incident of lymphadenopathy cases in children who received a Serbian-manufactured BCG vaccine.Citation11 However, out of all the listed vaccine country of origins, Germany was the most trusted origin with close to 80% of the participants expressed their willingness of taking a German vaccine, followed by the US and France with 48.7% and 44.5%, respectively. While Indian or Chinese produced vaccines were the least trusted vaccines, the bulk of the respondents stated that they will not take a locally produced one.
The trust in German or American produced vaccines are likely because of the producers’ sound scientific reputations, as well as their transparency through the development processes. Although the reason for not taking a local vaccine was not investigated, we assume that the existing distrust in the local vaccine production is influenced by the sources of vaccine information that led to such inclination of rejecting local vaccines. This assumption could be supported by the finding that 10% of the participants believe or source their vaccine information from family and friends, which stems from people’s tendency in believing or trusting their peers or those close to them.
Another influencing factor is the participants’ trust in the health care system, which can be defined as a “set of expectations that patients have from the health care system to help in the healing process including appropriate diagnosis, correct treatment, nonexploitation, interest in the welfare of the patient and transparent disclosure of information”.Citation12 While evaluating the trust in the health care system is beyond the scope of the current study, participants were asked to evaluate their trust in the health care system through their attitude toward the MOH recommended vaccination schedule. Several studies characterized the measurement of trust in the health care system as a relative measure proportional to individual set of services, in this instance vaccine schedule.Citation13
Interestingly, there is a significant trust in the MOH vaccination plan with more than 80% of the respondents stated that they do follow the recommended vaccination schedule, including those for children, with two-thirds agreed with the compulsory plan, indicating a high degree of trust in the health care system. Interestingly, trust in the health care system is a crucial factor in boosting medication adherence and acceptance.Citation14 Furthermore, most of the participants, (88%) either believed or strongly believed the results of evidence-based research. Therefore, the trust in the health care system and the believe in the results of evidence-based research are likely to be influenced by the participants’ level of education as all of the respondents stated that they have received at least 12 years of formal education. The relationship between the level of education and health is well established and that the level of education is strongly linked to health behaviors.Citation15 Also, participants’ fluency and knowledge have been identified among the most important determinants that influenced individuals’ belief in research-based evidence.Citation16
Also, the results showed a minority of less than 20% of the participants might be discouraged from vaccination following a negative media post regarding vaccines. However, a study by Walter and colleagues who examined certain social media accounts between 2015 and 2017 and their spread of vaccine misinformation; suggested that the social media platform could exacerbate vaccine misinformation that could potentially reduce vaccination rates and magnify health disparities.Citation17 Other factors that could reduce vaccine confidence include the rapid and widespread of vaccine misinformation online and the anti-vaccine campaigns that prey on the less-knowledgeable individuals and the divisive public.Citation17,Citation18 However, the participants’ above-average educational status and their trust in evidence-based research may explain the observed relatively low influence of social media.
Based on the analyses using the multivariable regression model, the characteristics of a COVID-19 vaccine hesitant, is likely to be a parent with one child or more, easily discouraged from vaccination following a negative post on social media, and will not pay for a vaccine. To the contrary, respondents who are willing to participate in COVID-19 vaccination trial, and rely on evidence-based scientific research were identified as vaccine acceptance. However, another decisive factor that seems to significantly impact the rate of COVID-19 vaccine hesitancy in Kazakhstan is the vaccine country of origin, with hesitant individuals have high degree of hesitancy toward locally produced vaccines and vaccine acceptant individuals appear to favor vaccines that are developed outside of Kazakhstan. This is important information for governmental institutions and policymakers and should be accounted for in their trust-building communication strategy that aims to boost vaccine acceptance in the Republic of Kazakhstan.
Limitations and bias
There are a number of drawbacks in the study. For example, first drawback is the use of convenience sampling as the method of sample recruitment, which has an inherited bias of under or over presenting the target group. Another drawback is the sample recruitment method. The participants were recruited via mass e-mails to local universities, hospitals and other institutions, which explains the high percentage of university graduates. This could also test the impact of education on vaccine hesitancy. Also, the studied sample size does not represent the whole country; however, the participants were from all major cities in the country, which represent wide geographic areas. Hence, the study can be considered as a representative sample. Additionally, the sample size was large enough to obtain sufficient statistical power (100%) in accurately detecting prevalence of COVID vaccine hesitancy.
Conclusion
In spite of the significant economical and mortality impact of the current COVID-19 pandemic, a number of people are either reluctant or not willing to vaccinate against the potentially deadly disease. Mass-vaccination is one of the most effective interventions capable of controlling widespread infections such as the current pandemic, but their outcome is based on the public’s trust in, and willingness to receive the vaccine (WHO). There are several reported reasons that impacted the respondent’s attitude toward COVID-19 vaccines, most importantly, vaccines country of origin and its side effects. Thus, addressing valid reason of concerns such vaccines’ production, side effects or adverse outcomes, responding to vaccine misinformation, and emphasizing on the benefits of vaccination will improve vaccine acceptance. This requires a multidimensional approach from different parties including policymakers, hhealth care providers, and all media platforms. Therefore, it is imperative to increase people’s trust in vaccine in order to improve its coverage and potentially control the current pandemic.
Disclosure of potential conflicts of interest
No potential conflicts of interest were disclosed.
Acknowledgments
This work is part of the Longitudinal Scholarly Project for Zylkiya Akhmetzhanova at the Nazarbayev University School of Medicine (NUSOM). The Authors are grateful for the diligent administrative support provided by the School.
References
- De Gregorio E, Rappuoli R. From empiricism to rational design: a personal perspective of the evolution of vaccine development. Nat Rev Immunol. 2014;14:505–14. doi:10.1038/nri3694.
- Zimmer C, Corum J, Wee S-L. Coronavirus vaccine tracker. 2021 Jan 26 [accessed 2021 Feb 15]. https://www.nytimes.com/interactive/2020/science/coronavirus-vaccine-tracker.html.
- Pew Research Center. Intent to get a COVID-19 vaccine rises to 60% as confidence in research and development process increases. 2020 Dec 3 [accessed 2021 Feb 16]. https://www.pewresearch.org/science/2020/12/03/intentto-get-a-covid-19-vaccine-rises-to-60-as-confidence-in-research-and-development-process-increases/.
- Akhmetzhanova Z, Sazonov V, Riethmacher D, Aljofan M. Vaccine adherence: the rate of hesitancy toward childhood immunization in Kazakhstan. Expert Rev Vaccines. 2020 Jun;19(6):579–84. Epub 2020 Jun 4. PMID: 32498567. doi:10.1080/14760584.2020.1775080.
- Aljofan M, Altebainawi A, Alrashidi MN. Public knowledge, attitude and practice toward diabetes mellitus in Hail region, Saudi Arabia. Int J Gen Med. 2019;12:255–62. PubMed PMID: WOS: 000477634800001. doi:10.2147/Ijgm.S214441.
- Opel DJ, Taylor JA, Mangione-Smith R, Solomon C, Zhao C, Catz S, Martin D. Validity and reliability of a survey to identify vaccine-hesitant parents. Vaccine. 2011;29(38):6598–605. Epub 2011/ 07/19. PubMed PMID: 21763384. doi:10.1016/j.vaccine.2011.06.115.
- Nowak GJ, Gellin BG, MacDonald NE, Butler R, SAGE Working Group on Vaccine Hesitancy. Addressing vaccine hesitancy: the potential value of commercial and social marketing principles and practices. Vaccine. 2015 Aug 14;33(34):4204–11. Epub 2015 Apr 18. PMID: 25900132. doi:10.1016/j.vaccine.2015.04.039.
- COVID Collaborative. COVID collaborative survey: coronavirus vaccination hesitancy in the black and latinx communities. 2020 Nov 23 [accessed 2021 Jan 26]. https://www.covidcollaborative.us/content/vaccine-treatments/coronavirusvaccine-hesitancy-in-black-and-latinx-communities.
- Kaiser Family Foundation. KFF health tracking poll – September 2020. 2020 Sept 10 [accessed 2021 Feb 15]. http://files.kff.org/attachment/Topline-KFF-Health-Tracking-Poll-September-2020.pdf.
- Chou WY, Burgdorf CE, Gaysynsky A, Hunter CM. Applying behavioral and social science to address vaccine hesitancy and foster vaccine confidence. USA: National Institute of Health; 2020 Dec.
- Fowler GL, Kennedy A, Leidel L, Kohl KS, Khromava A, Bizhanova G, Shui I, Gust D. Vaccine safety perceptions and experience with adverse events following immunization in Kazakhstan and Uzbekistan: a summary of key informant interviews and focus groups. Vaccine. 2007 May 4;25(18):3536–43. doi:10.1016/j.vaccine.2007.01.082.
- Caterinicchio R. Testing plausible path models of interpersonal trust in patient physician treatment relationships. Soc Sci Med. 1979;13:81–99.
- Sabate E, editor. Adherence to long-term therapies: evidence for action. Geneva (Switzerland): World Health Organization; 2003. http://www.who.int/chronic_conditions/adherencereport/en/.
- Kirst M, Shankardass K, Bomze S, Lofters A, Quiñonez C. Sociodemographic data collection for health equity measurement: a mixed methods study examining public opinions. Int J Equity Health. 2013;12(75):75. doi:10.1186/1475-9276-12-75.
- Feinstein L, Sabates R, Anderson TM, Sorhaindo A, Hammond C. What are the effects of education on health?. Measuring the Effects of Education on Health and Civic Engagement: Proceedings of the Copenhagen Symposium; 2006; Copenhagen, Denmark. OECD.
- Black AT, Balneaves LG, Garossino C, Puyat JH, Qian H. Promoting evidence-based practice through a research training program for point-of-care clinicians. JONA. 2015 Jan;45(1):14–20. doi:10.1097/NNA.0000000000000151.
- Walter D, Ophir Y, Jamieson KH. Russian Twitter accounts and the partisan polarization of vaccine discourse, 2015–2017. Am J Public Health. 2020;110(5):718–24. doi:10.2105/AJPH.2019.305564.
- Broniatowski DA, Jamison AM, Qi S, AlKulaib L, Chen T, Benton A, Quinn SC, Dredze M. Weaponized health communication: Twitter bots and Russian trolls amplify the vaccine debate. Am J Public Health. 2018;108(10):1378–84. doi:10.2105/AJPH.2018.304567.