ABSTRACT
Carbapenem-resistant Pseudomonas aeruginosa (CR-PA) is a major healthcare-associated pathogen worldwide. In the United States, 10–30% of P. aeruginosa isolates are carbapenem-resistant, while globally the percentage varies considerably. A subset of carbapenem-resistant P. aeruginosa isolates harbour carbapenemases, although due in part to limited screening for these enzymes in clinical laboratories, the actual percentage is unknown. Carbapenemase-mediated carbapenem resistance in P. aeruginosa is a significant concern as it greatly limits the choice of anti-infective strategies, although detecting carbapenemase-producing P. aeruginosa in the clinical laboratory can be challenging. Such organisms also have been associated with nosocomial spread requiring infection prevention interventions. The carbapenemases present in P. aeruginosa vary widely by region but include the Class A beta-lactamases, KPC and GES; metallo-beta-lactamases IMP, NDM, SPM, and VIM; and the Class D, OXA-48 enzymes. Rapid confirmation and differentiation among the various classes of carbapenemases is key to the initiation of early effective therapy. This may be accomplished using either molecular genotypic methods or phenotypic methods, although both have their limitations. Prompt evidence that rules out carbapenemases guides clinicians to more optimal therapeutic selections based on local phenotypic profiling of non-carbapenemase-producing, carbapenem-resistant P. aeruginosa. This article will review the testing strategies available for optimizing therapy of P. aeruginosa infections.
Introduction
Carbapenem-resistant Pseudomonas aeruginosa (CR-PA) is a major healthcare-associated pathogen worldwide [Citation1–3]. In the United States, P. aeruginosa is the primary cause of ventilator-associated pneumonia (VAP) in long-term acute care hospitals and on hospital wards and second most common cause of VAP in intensive care units. It also is the third most common cause of catheter-related urinary tract infections [Citation4]. Overall, in the United States, 10–30% of P. aeruginosa isolates are carbapenem-resistant [Citation5,Citation6] while globally the percentage varies considerably. There are several key mechanisms of carbapenem resistance in P. aeruginosa. The first mechanism is efflux of the drug, which is mediated by overexpression of the MexAB-OprM efflux pump [Citation7]. This results in resistance to most beta-lactam drugs with the exception of imipenem. The second mechanism is the overproduction of AmpC beta-lactamase and inactivation of the OprD outer membrane protein. This combination of mechanisms can cause resistance to essentially all antipseudomonal beta-lactams. A less common mechanism of carbapenem resistance among P. aeruginosa isolates, but one that appears to be increasing in frequency, is the production of carbapenemases [Citation2,Citation8,Citation9]. This mechanism of carbapenem resistance is important because it significantly alters the efficacy of commonly used antipseudomonal agents, including ceftazidime, cefepime, piperacillin-tazobactam, as well as the newly introduced beta-lactam/beta-lactamase inhibitor combinations such as ceftolozane-tazobactam, imipenem-relebactam and ceftazidime-avibactam. The carbapenem resistance determinants carried by P. aeruginosa are often encoded on plasmids, such as IncP type; class I integrons, such as those carrying the blaVIM gene; and other mobile genetics elements, such as those associated with insertion sequences with a common region (ISCRs), which enhance the organism's ability to disseminate resistance among multiple species [Citation10]. In addition, these isolates frequently carry additional resistance determinants that diminish the clinical utility of the fluoroquinolones and aminoglycosides. Carbapenemase-producing P. aeruginosa (CP-PA) are often resistant to all of these therapeutic options, thus making treatment failure a likely outcome. CP-PA has also been associated with nosocomial spread prompting infection prevention interventions [Citation11].
Epidemiology of carbapenemase-producing P. aeruginosa
P. aeruginosa isolates have been reported to contain a wide variety of carbapenemases globally. For example, in Latin America, this includes KPC, GES, IMP, VIM, NDM, and SPM [Citation9]. In the Arabian Peninsula, carbapenemases in P. aeruginosa include VIM, IMP, and NDM [Citation8]. In the United States, carbapenemases in P. aeruginosa include KPC, NDM, VIM, and IMP [Citation12,Citation13]. Unfortunately, several of the phenotypic methods for detecting carbapenemases that have been used worldwide, such as the Modified Hodge test, show either poor sensitivity or specificity, which confounds the epidemiology of these organisms [Citation14]. This and the lack of testing specifically for carbapenemase production among CR-PA globally suggest that the prevalence of CP-PA may be much higher than is perceived. The diversity and emerging prevalence of carbapenemase producers among CR-PA has been recently highlighted in the multi-national ERACE-PA Surveillance Program [Citation14a]. Of the 807 CR-PA collected over 2019–2021 from 17 centres in 12 countries, 33% tested carbapenemase-positive phenotypically (using the mCIM method) and of these, 86% were genotypically positive with the most common being VIM followed by GES. While carbapenemase producers were anticipated in the Middle East centres based on previously published epidemiology data, a high prevalence and diversity was also observed in the European, South American, and African centres. Moreover, in the United States centres (n = 5), a region not known to be of high prevalence, CP-PA were identified in 3–30% of the CR-PA. These contemporary global data suggest that carbapenemase testing in CR-PA is warranted. The key question is whether laboratories should be testing either for carbapenemase production phenotypically or genotypically via PCR for the presence of specific carbapenem-resistance genes among CR-PA isolates to assist antimicrobial stewardship programmes in selecting appropriate therapy for pseudomonal infections.
Testing carbapenemase-producing P. aeruginosa to aid antimicrobial stewardship
At present, when a CR-PA isolate is identified in a clinical laboratory during the first round of antimicrobial susceptibility testing, many institutions will perform additional susceptibility tests for ceftolozane/tazobactam, ceftazidime/avibactam, and/or imipenem/relebactam using automated antimicrobial susceptibility testing (AST) methods, agar diffusion methods (i.e. Gradient diffusion strips), or disk diffusion to guide therapy. These antimicrobial agents are highly active against a wide variety, although not all, CR-PA [Citation15–17]. However, not all laboratories have access to susceptibility testing panels, strips or disks for these beta-lactam/beta-lactamase inhibitor combinations. In addition, gradient diffusion strips and disk diffusion testing for these novel antimicrobials require another 16–20 h of incubation after the initial susceptibility test results become available, which may slow the decision-making process for guiding therapy. More timely data, such as that provided by commercial PCR or immunochromatographic tests to exclude the most common carbapenemases, can guide clinicians to early use of ceftolozane/tazobactam, ceftazidime/avibactam, and imipenem/relebactam as they are highly active against non-carbapenemase-producing, CR-PA. On the other hand, detection of carbapenemases, especially metallo-beta-lactamases (e.g. IMP, NDM, and VIM) will indicate the need to consider cefiderocol [Citation1,Citation18] or combination therapy inclusive of aztreonam [Citation19–21].
How should laboratories test for CP-PA? There are two approaches to testing that could guide antimicrobial stewardship efforts and thus improve therapeutic outcomes (, options 1 and 2). The first approach (Option 1) is to test colonies of P. aeruginosa with a broad phenotypic carbapenemase test, such as the modified carbapenem inactivated method (mCIM) or CarbaNP test [Citation22,Citation23]. These tests indicate whether or not a carbapenemase is present in the isolate, but not which type of carbapenemases it is (i.e. a serine versus metallo-beta-lactamase). However, performing a second mCIM test in the presence of EDTA (i.e. the eCIM test) can differentiate serine carbapenemases (which are not inhibited by EDTA) from metallo-carbapenemases, which is key information for selecting anti-pseudomonal therapy. Alternatively, if only the mCIM test is performed and it is positive, a genotypic test, either PCR or an immunochromatographic test, can be used to identify the specific classes of carbapenemases present (i.e. KPC, VIM, IMP, VIM, and OXA-48) [Citation24–26]. Several commercial tests have been shown to be accurate for detection of carbapenemases or carbapenem resistance genes, although the costs of the tests may vary from country to country. It should be noted that the mCIM test can have difficulty detecting some carbapenemases, such as IMP [Citation27,Citation28], although a recent study showed the combination of mCIM and eCIM testing to be effective for detecting most other carbapenemases in P. aeruginosa isolates [Citation29].
Figure 1. Laboratory-based screening options for the detection of carbapenemase-producers among carbapenem-resistant P. aeruginosa.
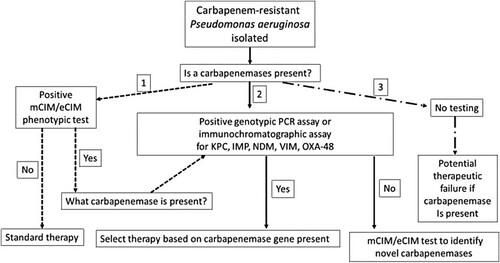
The second testing option is to begin with a commercial PCR or immunochromatographic test that detects KPC, VIM, IMP, VIM, and OXA-48, and if that test is negative to follow up with the mCIM or mCIM/eCIM combination tests. The advantage of the latter approach is that both the PCR and immunochromatographic test can often be completed in under 1 h, which although potentially more expensive for the laboratory, can lead to more precise therapeutic interventions in a time frame that may be 48 h sooner than Option 1. This makes the up-front use of these methods cost-effective. While whole genome sequencing (WGS) of isolated colonies can provide much greater information about the mechanisms of antimicrobial resistance in a P. aeruginosa isolate compared with the phenotypic and genotypic methods mentioned above, the slow turn-around time of results, the technical expertise required for nucleic acid extraction, library preparation, sequencing, and finally the lack of standardized databases to translate genotypes into phenotypes that can be readily understand by clinicians, currently limit the use of WGS to research facilities rather than hospital laboratories.
Option 3 is to perform to no additional testing and treat empirically, but this is not recommended as the number of treatments failures will surely increase as CP-PA isolates continue to spread globally. While the argument to forgo additional phenotypic and genotypic assessments for carbapenemase-production in CR-PA often focuses on the perceived value of the test relative to percent positive, staff and testing resources, the clinical implementation of a recently developed algorithm will aid in streamlining carbapenemase detection workflow in the laboratory [Citation27]. Importantly, test availability, whether positive or negative, translates to actionable results in the form of enhanced therapeutic and / or infection control interventions that are central to safeguarding good clinical outcomes while minimizing the dissemination of CP-PA.
Summary
There is an increasing probability of treatment failures with infections caused by carbapenem-resistant P. aeruginosa due to the unrecognized presence of carbapenemases. Combining both phenotypic and genotypic methods can significantly shorten the time to effective therapy and enhance patient outcomes. Yet, these tests often are not performed on P. aeruginosa isolates in many clinical laboratories. Thus, optimizing therapy for P. aeruginosa infections remains a challenge.
Disclosure statement
No potential conflict of interest was reported by the author(s).
Correction Statement
This article has been republished with minor changes. These changes do not impact the academic content of the article.
Additional information
Funding
References
- Horcajada JP, Montero M, Oliver A, et al. Epidemiology and treatment of multidrug-resistant and extensively drug-resistant Pseudomonas aeruginosa infections. Clin Microbiol Rev. 2019 Sep 18;32(4):e00031–19.
- Wang M-G, Liu Z-Y, Liao X-P, et al. Retrospective data insight into the global distribution of carbapenemase-producing Pseudomonas aeruginosa. Antibiotics (Basel). 2021 May 9;10(5):548.
- Al-Orphaly M, Hadi HA, Eltayeb FK, et al. Epidemiology of multidrug-resistant Pseudomonas aeruginosa in the Middle East and North Africa region. mSphere. 2021 May 19;6(3):e00202–21.
- Weiner-Lastinger LM, Abner S, Edwards JR, et al. Antimicrobial-resistant pathogens associated with adult healthcare-associated infections: summary of data reported to the National Healthcare Safety Network, 2015–2017. Infect Control Hosp Epidemiol. 2020 Jan;41(1):1–18.
- Woodworth KR, Walters MS, Weiner LM, et al. Vital signs: containment of novel multidrug-resistant organisms and resistance mechanisms – United States, 2006–2017. MMWR Morb Mortal Wkly Rep. 2018 Apr 6;67(13):396–401.
- Almarzoky Abuhussain SS, Sutherland CA, Nicolau DP. In vitro potency of antipseudomonal β-lactams against blood and respiratory isolates of P. aeruginosa collected from US hospitals. J Thorac Dis. 2019 May;11(5):1896–1902.
- Livermore DM. Multiple mechanisms of antimicrobial resistance in Pseudomonas aeruginosa: our worst nightmare? Clin Infect Dis. 2002 Mar 1;34(5):634–640.
- Zowawi HM, Balkhy HH, Walsh TR, et al. β-Lactamase production in key gram-negative pathogen isolates from the Arabian peninsula. Clin Microbiol Rev. 2013 Jul;26(3):361–380.
- Escandón-Vargas K, Reyes S, Gutiérrez S, et al. The epidemiology of carbapenemases in Latin America and the Caribbean. Expert Rev Anti Infect Ther. 2017 Mar;15(3):277–297.
- Yoon E-J, Jeong SH. Mobile carbapenemase genes in Pseudomonas aeruginosa. Front Microbiol. 2021 Feb 18;12:614058.
- Center for Disease Control and Prevention. 2019 AR Threats Report [Internet]. [cited 2019 May 8]. Available from: https://www.cdc.gov/drugresistance/biggest-threats.html.
- Poirel L, Nordmann P, Lagrutta E, et al. Emergence of KPC-producing Pseudomonas aeruginosa in the United States. Antimicrob Agents Chemother. 2010 Jul;54(7):3072.
- Walters MS, Grass JE, Bulens SN, et al. Carbapenem-resistant Pseudomonas aeruginosa at US Emerging infections program sites, 2015. Emerging Infect Dis. 2019 Jul;25(7):1281–1288.
- Simner PJ, Opene BNA, Chambers KK, et al. Carbapenemase detection among carbapenem-resistant glucose-nonfermenting gram-negative bacilli. J Clin Microbiol. 2017 Sep;55(9):2858–2864.
- Gill CM, Aktaþ E, Alfouzan W, Bourassa L, Brink A, Burnham CD, Canton R, Carmeli Y, Falcone M, Kiffer C, Marchese A, Martinez O, Pournaras S, Satlin M, Seifert H, Thabit AK, Thomson KS, Villegas MV, Nicolau DP, ERACE-PA Global Study Group. The ERACE-PA Global Surveillance Program: Ceftolozane/tazobactam and Ceftazidime/avibactam in vitro Activity against a Global Collection of Carbapenem-resistant Pseudomonas aeruginosa. Eur J Clin Microbiol Infect Dis. 2021 Dec;40(12):2533–2541.
- Grupper M, Sutherland C, Nicolau DP. Multicenter evaluation of ceftazidime-avibactam and ceftolozane-Tazobactam inhibitory activity against meropenem-nonsusceptible Pseudomonas aeruginosa from blood, respiratory tract, and wounds. Antimicrob Agents Chemother. 2017 Oct;61(10):e00875–17.
- Humphries RM, Hindler JA, Wong-Beringer A, et al. Activity of ceftolozane-tazobactam and ceftazidime-avibactam against beta-lactam-resistant Pseudomonas aeruginosa isolates. Antimicrob Agents Chemother. 2017 Dec;61(12):e01858–17.
- Pogue JM, Kaye KS, Veve MP, et al. Ceftolozane/tazobactam vs polymyxin or aminoglycoside-based regimens for the treatment of drug-resistant Pseudomonas aeruginosa. Clin Infect Dis. 2020 Jul 11;71(2):304–310.
- Hackel MA, Tsuji M, Yamano Y, et al. In vitro activity of the siderophore cephalosporin, cefiderocol, against carbapenem-nonsusceptible and multidrug-resistant isolates of gram-negative Bacilli collected worldwide in 2014 to 2016. Antimicrob Agents Chemother. 2018 Jan 25;62(2):e01968–17.
- Vasoo S, Cunningham SA, Cole NC, et al. In vitro activities of Ceftazidime-Avibactam, aztreonam-avibactam, and a panel of older and contemporary antimicrobial agents against carbapenemase-producing gram-negative bacilli. Antimicrob Agents Chemother. 2015 Dec;59(12):7842–7846.
- Brown AC, Malik S, Huang J, et al. 484. Metallo-β-Lactamase-Positive carbapenem-resistant enterobacteriaceae and Pseudomonas aeruginosa in the antibiotic resistance laboratory network, 2017–2018. Open Forum Infect Dis. 2019 Oct 23;6(Supplement_2):S237–S237.
- Li H, Estabrook M, Jacoby GA, et al. In vitro susceptibility of characterized β-lactamase-producing strains tested with avibactam combinations. Antimicrob Agents Chemother. 2015 Mar;59(3):1789–1793.
- Simner PJ, Johnson JK, Brasso WB, et al. Multicenter evaluation of the modified Carbapenem inactivation method and the carba NP for detection of Carbapenemase-producing Pseudomonas aeruginosa and acinetobacter baumannii. J Clin Microbiol. 2018;56(1):e01369–17.
- Gill CM, Lasko MJ, Asempa TE, et al. Evaluation of the EDTA-modified Carbapenem inactivation method (eCIM) for detecting metallo-β-lactamase-producing Pseudomonas aeruginosa. J Clin Microbiol. 2020 Apr 1;e02015–19.
- Traczewski MM, Carretto E, Canton R, Moore NM, Carba-R Study Team. Multicenter Evaluation of the Xpert Carba-R Assay for Detection of Carbapenemase Genes in Gram-Negative Isolates. J Clin Microbiol. 2018 Jul 26;58(8):e00272–18.
- Kanahashi T, Matsumura Y, Yamamoto M, et al. Comparison of the Xpert Carba-R and NG-test CARBA5 for the detection of carbapenemases in an IMP-type carbapenemase endemic region in Japan. J Infect Chemother. 2021 Mar;27(3):503–506.
- Gill CM, Asempa TE, Tickler IA, et al. Evaluation of the Xpert Carba-R NxG assay for detection of carbapenemase genes in a global challenge set of Pseudomonas aeruginosa isolates. J Clin Microbiol. 2020 Nov 18;58(12):e01098–20.
- Gill CM, Asempa TE, Nicolau DP. Development and application of a pragmatic algorithm to guide definitive carbapenemase testing to identify carbapenemase-producing Pseudomonas aeruginosa. Antibiotics (Basel). 2020 Oct 27;9(11):738.
- Lasko MJ, Gill CM, Asempa TE, et al. EDTA-modified carbapenem inactivation method (eCIM) for detecting IMP metallo-β-lactamase-producing Pseudomonas aeruginosa: an assessment of increasing EDTA concentrations. BMC Microbiol. 2020 Jul 20;20(1):220.
- Tenover FC, Dela Cruz CM, Dewell S, et al. Does the presence of multiple β-lactamases in gram-negative bacilli impact the results of antimicrobial susceptibility tests and extended-spectrum β-lactamase and carbapenemase confirmation methods? J Glob Antimicrob Resist. 2020 Sep 2;23:87–93.
